AZD8797_COA_24760_MedChemExpress
超高效液相色谱-串联质谱法测定人血浆中精氨酸及衍生物含量

超高效液相色谱-串联质谱法测定人血浆中精氨酸及衍生物含量田晔;江骥;胡蓓;薛金萍;王洪允【摘要】建立了超高效液相色谱-串联质谱(UPLC-MS/MS)法同时测定使用艾普拉唑后人血浆中二甲基精氨酸(ADMA)、对称二甲基精氨酸(SDMA)、单甲基精氨酸(NMMA)、瓜氨酸(Cit)和L-精氨酸(L-Arg)的浓度.采用HILIC亲水相互作用色谱和非衍生化的蛋白沉淀法进行分离分析,色谱柱选取Waters Atlantic HILIC柱(2.1 mm×50 mm×3μm),流动相由乙腈(含0.5%乙酸和0.025%三氟乙酸)-水(含0.5%乙酸和0.025%三氟乙酸)(85:15,v/V)组成,流速0.25 mL/min.采用多反应离子监测(MRM)模式,以电喷雾离子源(ESI)正离子方式检测.结果显示,ADMA、SDMA、NMMA、L-Arg和Cit的线性关系良好,相关系数r均大于0.994 0;ADMA、SDMA和NMMA的线性范围为0.1~5 mmol/L,L-Arg和Cit的线性范围为10~250 mmol/L;5种氨基酸的日内、日间精密度均小于15%,准确度在85%~115%之间.该方法快速、简便、灵敏,可为相关疾病的临床诊断提供一种高效的检测手段.【期刊名称】《质谱学报》【年(卷),期】2016(037)005【总页数】7页(P446-452)【关键词】超高效液相色谱-串联质谱(UPLC-MS/MS);艾普拉唑;蛋白沉淀法;亲水性色谱【作者】田晔;江骥;胡蓓;薛金萍;王洪允【作者单位】福州大学化学学院,福建省功能材料工程研究中心,福建省光动力治疗药物与诊疗工程技术研究中心,福建福州350108;中国医学科学院北京协和医院临床药理中心,北京100730;中国医学科学院北京协和医院临床药理中心,北京100730;中国医学科学院北京协和医院临床药理中心,北京100730;福州大学化学学院,福建省功能材料工程研究中心,福建省光动力治疗药物与诊疗工程技术研究中心,福建福州350108;中国医学科学院北京协和医院临床药理中心,北京100730【正文语种】中文【中图分类】O657.63一氧化氮是人体重要的信使分子,L-精氨酸(L-Arg)在一氧化氮全酶(NOS)的催化下,产生一氧化氮(NO)和瓜氨酸(Cit)[1-2]。
Gelucire-14-44-SDS-MedChemExpress

Inhibitors, Agonists, Screening LibrariesSafety Data Sheet Revision Date:Nov.-23-2018Print Date:Nov.-23-20181. PRODUCT AND COMPANY IDENTIFICATION1.1 Product identifierProduct name :Gelucire 14/44Catalog No. :HY-Y1892CAS No. :121548-04-71.2 Relevant identified uses of the substance or mixture and uses advised againstIdentified uses :Laboratory chemicals, manufacture of substances.1.3 Details of the supplier of the safety data sheetCompany:MedChemExpress USATel:609-228-6898Fax:609-228-5909E-mail:sales@1.4 Emergency telephone numberEmergency Phone #:609-228-68982. HAZARDS IDENTIFICATION2.1 Classification of the substance or mixtureNot a hazardous substance or mixture.2.2 GHS Label elements, including precautionary statementsNot a hazardous substance or mixture.2.3 Other hazardsNone.3. COMPOSITION/INFORMATION ON INGREDIENTS3.1 SubstancesSynonyms:NoneFormula:N/AMolecular Weight:N/ACAS No. :121548-04-74. FIRST AID MEASURES4.1 Description of first aid measuresEye contactRemove any contact lenses, locate eye-wash station, and flush eyes immediately with large amounts of water. Separate eyelids with fingers to ensure adequate flushing. Promptly call a physician.Skin contactRinse skin thoroughly with large amounts of water. Remove contaminated clothing and shoes and call a physician.InhalationImmediately relocate self or casualty to fresh air. If breathing is difficult, give cardiopulmonary resuscitation (CPR). Avoid mouth-to-mouth resuscitation.IngestionWash out mouth with water; Do NOT induce vomiting; call a physician.4.2 Most important symptoms and effects, both acute and delayedThe most important known symptoms and effects are described in the labelling (see section 2.2).4.3 Indication of any immediate medical attention and special treatment neededTreat symptomatically.5. FIRE FIGHTING MEASURES5.1 Extinguishing mediaSuitable extinguishing mediaUse water spray, dry chemical, foam, and carbon dioxide fire extinguisher.5.2 Special hazards arising from the substance or mixtureDuring combustion, may emit irritant fumes.5.3 Advice for firefightersWear self-contained breathing apparatus and protective clothing.6. ACCIDENTAL RELEASE MEASURES6.1 Personal precautions, protective equipment and emergency proceduresUse full personal protective equipment. Avoid breathing vapors, mist, dust or gas. Ensure adequate ventilation. Evacuate personnel to safe areas.Refer to protective measures listed in sections 8.6.2 Environmental precautionsTry to prevent further leakage or spillage. Keep the product away from drains or water courses.6.3 Methods and materials for containment and cleaning upAbsorb solutions with finely-powdered liquid-binding material (diatomite, universal binders); Decontaminate surfaces and equipment by scrubbing with alcohol; Dispose of contaminated material according to Section 13.7. HANDLING AND STORAGE7.1 Precautions for safe handlingAvoid inhalation, contact with eyes and skin. Avoid dust and aerosol formation. Use only in areas with appropriate exhaust ventilation.7.2 Conditions for safe storage, including any incompatibilitiesKeep container tightly sealed in cool, well-ventilated area. Keep away from direct sunlight and sources of ignition.Recommended storage temperature:Pure form-20°C 3 years4°C 2 yearsIn solvent-80°C 6 months-20°C 1 monthShipping at room temperature if less than 2 weeks.7.3 Specific end use(s)No data available.8. EXPOSURE CONTROLS/PERSONAL PROTECTION8.1 Control parametersComponents with workplace control parametersThis product contains no substances with occupational exposure limit values.8.2 Exposure controlsEngineering controlsEnsure adequate ventilation. Provide accessible safety shower and eye wash station.Personal protective equipmentEye protection Safety goggles with side-shields.Hand protection Protective gloves.Skin and body protection Impervious clothing.Respiratory protection Suitable respirator.Environmental exposure controls Keep the product away from drains, water courses or the soil. Cleanspillages in a safe way as soon as possible.9. PHYSICAL AND CHEMICAL PROPERTIES9.1 Information on basic physical and chemical propertiesAppearance White to off-white (Oil)Odor No data availableOdor threshold No data availablepH No data availableMelting/freezing point No data availableBoiling point/range No data availableFlash point No data availableEvaporation rate No data availableFlammability (solid, gas)No data availableUpper/lower flammability or explosive limits No data availableVapor pressure No data availableVapor density No data availableRelative density No data availableWater Solubility No data availablePartition coefficient No data availableAuto-ignition temperature No data availableDecomposition temperature No data availableViscosity No data availableExplosive properties No data availableOxidizing properties No data available9.2 Other safety informationNo data available.10. STABILITY AND REACTIVITY10.1 ReactivityNo data available.10.2 Chemical stabilityStable under recommended storage conditions.10.3 Possibility of hazardous reactionsNo data available.10.4 Conditions to avoidNo data available.10.5 Incompatible materialsStrong acids/alkalis, strong oxidising/reducing agents.10.6 Hazardous decomposition productsUnder fire conditions, may decompose and emit toxic fumes.Other decomposition products - no data available.11.TOXICOLOGICAL INFORMATION11.1 Information on toxicological effectsAcute toxicityClassified based on available data. For more details, see section 2Skin corrosion/irritationClassified based on available data. For more details, see section 2Serious eye damage/irritationClassified based on available data. For more details, see section 2Respiratory or skin sensitizationClassified based on available data. For more details, see section 2Germ cell mutagenicityClassified based on available data. For more details, see section 2CarcinogenicityIARC: No component of this product present at a level equal to or greater than 0.1% is identified as probable, possible or confirmed human carcinogen by IARC.ACGIH: No component of this product present at a level equal to or greater than 0.1% is identified as a potential or confirmed carcinogen by ACGIH.NTP: No component of this product present at a level equal to or greater than 0.1% is identified as a anticipated or confirmed carcinogen by NTP.OSHA: No component of this product present at a level equal to or greater than 0.1% is identified as a potential or confirmed carcinogen by OSHA.Reproductive toxicityClassified based on available data. For more details, see section 2Specific target organ toxicity - single exposureClassified based on available data. For more details, see section 2Specific target organ toxicity - repeated exposureClassified based on available data. For more details, see section 2Aspiration hazardClassified based on available data. For more details, see section 212. ECOLOGICAL INFORMATION12.1 ToxicityNo data available.12.2 Persistence and degradabilityNo data available.12.3 Bioaccumlative potentialNo data available.12.4 Mobility in soilNo data available.12.5 Results of PBT and vPvB assessmentPBT/vPvB assessment unavailable as chemical safety assessment not required or not conducted.12.6 Other adverse effectsNo data available.13. DISPOSAL CONSIDERATIONS13.1 Waste treatment methodsProductDispose substance in accordance with prevailing country, federal, state and local regulations.Contaminated packagingConduct recycling or disposal in accordance with prevailing country, federal, state and local regulations.14. TRANSPORT INFORMATIONDOT (US)This substance is considered to be non-hazardous for transport.IMDGThis substance is considered to be non-hazardous for transport.IATAThis substance is considered to be non-hazardous for transport.15. REGULATORY INFORMATIONSARA 302 Components:No chemicals in this material are subject to the reporting requirements of SARA Title III, Section 302.SARA 313 Components:This material does not contain any chemical components with known CAS numbers that exceed the threshold (De Minimis) reporting levels established by SARA Title III, Section 313.SARA 311/312 Hazards:No SARA Hazards.Massachusetts Right To Know Components:No components are subject to the Massachusetts Right to Know Act.Pennsylvania Right To Know Components:No components are subject to the Pennsylvania Right to Know Act.New Jersey Right To Know Components:No components are subject to the New Jersey Right to Know Act.California Prop. 65 Components:This product does not contain any chemicals known to State of California to cause cancer, birth defects, or anyother reproductive harm.16. OTHER INFORMATIONCopyright 2018 MedChemExpress. The above information is correct to the best of our present knowledge but does not purport to be all inclusive and should be used only as a guide. The product is for research use only and for experienced personnel. It must only be handled by suitably qualified experienced scientists in appropriately equipped and authorized facilities. The burden of safe use of this material rests entirely with the user. MedChemExpress disclaims all liability for any damage resulting from handling or from contact with this product.Caution: Product has not been fully validated for medical applications. For research use only.Tel: 609-228-6898 Fax: 609-228-5909 E-mail: tech@Address: 1 Deer Park Dr, Suite Q, Monmouth Junction, NJ 08852, USA。
气相色谱法测定那格列奈原药中二氯甲烷和吡啶残留量

气相色谱法测定那格列奈原药中二氯甲烷和吡啶残留量刘振莲【摘要】建立气相色谱法测定那格列奈中二氯甲烷和吡啶的残留量.采用6%氰丙基苯基-94%二甲基聚硅氧烷为固定液的毛细管柱,以氮气为载气,FID检测,直接进样,测定残留溶剂的含量.二氯甲烷质量浓度在0.75~36.0 μg/mL范围内线性关系良好,相关系数r=0.999 9,回收率为97.45%,最低检测限为0.24 μg/mL;吡啶质量浓度在0.27~12.30 μg/mL范围内线性关系良好,相关系数r=0.999 6,回收率为93.49%,最低检测限为0.08 μg/mL.【期刊名称】《淮海工学院学报(自然科学版)》【年(卷),期】2013(022)001【总页数】3页(P35-37)【关键词】那格列奈;二氯甲烷;吡啶;气相色谱法【作者】刘振莲【作者单位】江苏正大天晴药业股份有限公司连云港研究院,江苏连云港222062【正文语种】中文【中图分类】R9170 引言那格列奈即Nateglinide(商品名Starsis),化学名为 N-(反-4-异丙基环己羰基)-D-苯丙氨酸,是新一代具有氨基酸结构的降糖药物[1-3]。
其能有效控制餐时血糖,效果优于磺脉类降糖药,并且能降低低血糖的发生率。
据美国糖尿病学会会议上公布的临床资料显示,在用于Ⅱ型糖尿病治疗时,该药与同样具有刺激胰岛素分泌作用的其他产品相比,具有作用效果好、起效快、作用时间短和给药灵活等优点。
药品中的残留溶剂是指在合成原料药、辅料或制剂生产过程中使用或产生的挥发性有机化学物质,它们在实际生产中未能被完全清除。
近年来,药品中残存的有机溶剂的毒性和致癌作用日益引起各方面的重视[4-8]。
由于在那格列奈原药合成过程中使用了有机溶剂二氯甲烷和吡啶,因此在成品中可能会有一定量的残留。
按《中华人民共和国药典2005年版》(附录Ⅷ)规定,该2种有机溶剂均有残留限量,因此对那格列奈原药中二氯甲烷和吡啶的残留量检测具有一定意义。
阿苯达唑亚砜盐酸盐对照品候选物纯度检测方法的建立

[ 摘 要 】 以盐酸 阿苯达 唑亚砜 原料 为基 础 , 过反 复提纯 , 到盐 酸 阿苯达 唑亚砜 对 照 品候 选 通 得 物, 该候选 物 经过熔 点 , 薄层色谱 法 ( L 和 高 效液 相 色谱 法 检测 , 步 确认 符 合对 照 品的要 求 , T C) 初 经 过高 效液相 色谱 检测 , 用 面积 归一化 法 计算 其 纯度 不 小 于 9 .% 。该 方法 的建 立 为 盐酸 阿 采 95 苯 达 唑对 照 品进 一步研 究提 供 了帮助 。 [ 关键 词 ] 阿苯 达 唑亚砜 盐酸 盐 ; 薄层 色谱 法 ; 效液相 色谱 法 ; 积 归一化 高 面 Esa ihm e o tc i t bl s ntf rDe e tng Purt fAl n z l ulo i iy o be da o e S f xde Hy o hl rde Ree e e S s n e Ca d da e dr c o i fr nc ub t c n i t s a
2 1 , ( ) 1 5 刘 志亮 , 0 04 7 :3—1/ 4 等
中国兽药 杂志
・ 3・ 1
阿苯达 唑亚砜 盐 酸盐 对 照 品候 选 物 纯 度 检 测方 法 的建 立
刘 志亮 徐光科 裴香玲 乔彦 良 , , , , 闫祥华
( .山东信得科技股份有限公司 , 1 山东青岛 26 6 ; . 阳农业高等专科学 校 , 60 1 2 信 河南信 阳 4 40 ) 6 00 [ 收稿 日 ]0 0 0 1 [ 期 2 1 — 4— 2 文献标识码] [ A 文章编号] 02— 2 0 2 1 )7- 0 3 0 【 10 18 (0 0 0 0 1 - 3 中图分类号] Q 6 .2 T 4 0 7
GeXP简介

•Alignment
•Call scores
•Heterozygote Detection
2013/11/12
6
GeXP荧光系统
•GeXP更适合检测突变/杂合子: •波长越长,干扰越少 ,背景噪音低;
•650nm •laser •750nm •laser
•无10%的cut off把噪音,不会把10%以上杂合子去掉;
•NO Interference •from biological materials
7
个体化用药检测
KIT-Exon9
PDGFRA-exon12
EGFR突变检测
肿瘤药物对应相关基因的检测
药物名称 易瑞沙/特罗凯类 检测基因
EGFR-Exon18 突变 EGFR-Exon19 突变 EGFR-Exon21 突变 EGFR-Exon20突变 C-KIT-Exon9 突变 C-KIT-Exon11 突变 C-KIT-Exon13 突变 C-KIT-Exon17 突变 PDGFRα-Exon12 PDGFRα-Exon18 CYP2D6*10 多态性 XRCC1-Exon6 多态性 XRCC1-Exon10 多态性 ERCC1-codon118 多态性 MRP2-Exon10 多态性 BRCA1-Exon2 (女)多态性 BRCA1-Exon20 (女)多态性 XPD基因多态性 UGT1A1 *6 多态性 UGT1A1*28 多态性 DPYD*2A 多态性
伊马替尼 他莫昔芬
铂类
伊立替康 氟脲嘧啶类
HBV分型、耐药突变检测
2、片段分析
• 只需要研究长度,不需要知道具体序列 • 分别率为1bp
片段分析应用
STR/SSR
融合基因,可变剪切体
GC测定盐酸普拉克索中三乙胺残留量

一种国产胃泌素释放肽前体化学发光免疫试剂的性能验证

一种国产胃泌素释放肽前体化学发光 免疫试剂的性能验证
欧赛英1,徐海伟1,陈秀发1,沙利烽1,梁 辰2,冯 杰2,薛 辉2,颜光涛2
(1.苏州长光华医生物试剂有限公司,江苏 苏州 215163; 2.中国人民解放军总医院医学检验中心,北京 100853)
ThePerformanceVerificationofaDomesticGastrinreleasing PeptideforChemiluminescenceImmunoreagent
OU Saiying1,XU Haiwei1,CHEN Xiufa1,SA Lifeng1,LIANG Chen2, FENG Jie2,XUEHui2,YAN Guangtao2
(1.SuzhouChangguanghuaMedicalReagentCo.LTD,Suzhou215163,China; 2.MedicalLaboratoryCenterofPLA GeneralHospital,Beijing100853,China)
Abstract:ObjectiveProGRPisapromisingeffectivetumormarkerindiscriminatingSCLCfrom NSCLCand otherbenignlungdiseases.ProGRPChemiluminescentimmunoassay(CLIA)wasdevelopedandmanufactured byHybiome.Themanufacturerdeclaredtheperformanceparametersasfollowing:theprecisionofthereagent waslessthan10%,thelimitofdetectionwas5pg/mL,therecoverywasbetween <±10%,thereference rangewas<65pg/mL,thelinearrangewas153000pg/mL,theserum waswellcorrelatedwithplasma,and theserum stabilitycouldreach96hours,andthecorrelationwithRocheelectrochemiluminescenceproGRP reagentwasgood.Theperformanceofthereagentwasverifiedinthisstudy.MethodsAccordingtotheCLSI andCAP,theprecision,limitofquantitation,recoveryrate,linearrange,referenceinterval,serum plasma consistency,serum stabilityandcomplementinterferenceofthedomesticproGRPassayanditssupporting instrument,AE180detectionsystem,wereverified.199casesamplesfrom healthycontrol,smallcelllung cancer,nonsmallcelllungcancerandbenignlungdiseasepatients,weredetectedinparallelwithRocheE601 system tocomparetheconsistencyoftheresultsbetweenthetwodetectionsystems.ResultsThenewProGRP
氘可来昔替尼结构式 -回复

氘可来昔替尼结构式-回复氘可来昔替尼(Dacomitinib)是一种口服酪氨酸激酶抑制剂(EGFR 抑制剂),用于治疗非小细胞肺癌(NSCLC)。
它被用作一线治疗药物,可以延长患者的生存期和改善症状。
以下是关于氘可来昔替尼的结构式及其相关信息的详细解释。
氘可来昔替尼的结构式如下:化学名称:N-[2-[(3-Chloro-4-fluorophenyl)amino]-4-methoxy-6-quinolinyl]-1 ,3-thiazole-5-carboxamide结构式:
Inhibitors, Agonists, Screening LibrariesSafety Data Sheet Revision Date:May-24-2017Print Date:May-24-20171. PRODUCT AND COMPANY IDENTIFICATION1.1 Product identifierProduct name :AZ876Catalog No. :HY-18282CAS No. :898800-26-51.2 Relevant identified uses of the substance or mixture and uses advised againstIdentified uses :Laboratory chemicals, manufacture of substances.1.3 Details of the supplier of the safety data sheetCompany:MedChemExpress USATel:609-228-6898Fax:609-228-5909E-mail:sales@1.4 Emergency telephone numberEmergency Phone #:609-228-68982. HAZARDS IDENTIFICATION2.1 Classification of the substance or mixtureNot a hazardous substance or mixture.2.2 GHS Label elements, including precautionary statementsNot a hazardous substance or mixture.2.3 Other hazardsNone.3. COMPOSITION/INFORMATION ON INGREDIENTS3.1 SubstancesSynonyms:AZ⁻876; AZ 876Formula:C24H29N3O3SMolecular Weight:439.57CAS No. :898800-26-54. FIRST AID MEASURES4.1 Description of first aid measuresEye contactRemove any contact lenses, locate eye-wash station, and flush eyes immediately with large amounts of water. Separate eyelids with fingers to ensure adequate flushing. Promptly call a physician.Skin contactRinse skin thoroughly with large amounts of water. Remove contaminated clothing and shoes and call a physician.InhalationImmediately relocate self or casualty to fresh air. If breathing is difficult, give cardiopulmonary resuscitation (CPR). Avoid mouth-to-mouth resuscitation.IngestionWash out mouth with water; Do NOT induce vomiting; call a physician.4.2 Most important symptoms and effects, both acute and delayedThe most important known symptoms and effects are described in the labelling (see section 2.2).4.3 Indication of any immediate medical attention and special treatment neededTreat symptomatically.5. FIRE FIGHTING MEASURES5.1 Extinguishing mediaSuitable extinguishing mediaUse water spray, dry chemical, foam, and carbon dioxide fire extinguisher.5.2 Special hazards arising from the substance or mixtureDuring combustion, may emit irritant fumes.5.3 Advice for firefightersWear self-contained breathing apparatus and protective clothing.6. ACCIDENTAL RELEASE MEASURES6.1 Personal precautions, protective equipment and emergency proceduresUse full personal protective equipment. Avoid breathing vapors, mist, dust or gas. Ensure adequate ventilation. Evacuate personnel to safe areas.Refer to protective measures listed in sections 8.6.2 Environmental precautionsTry to prevent further leakage or spillage. Keep the product away from drains or water courses.6.3 Methods and materials for containment and cleaning upAbsorb solutions with finely-powdered liquid-binding material (diatomite, universal binders); Decontaminate surfaces and equipment by scrubbing with alcohol; Dispose of contaminated material according to Section 13.7. HANDLING AND STORAGE7.1 Precautions for safe handlingAvoid inhalation, contact with eyes and skin. Avoid dust and aerosol formation. Use only in areas with appropriate exhaust ventilation.7.2 Conditions for safe storage, including any incompatibilitiesKeep container tightly sealed in cool, well-ventilated area. Keep away from direct sunlight and sources of ignition.Recommended storage temperature:Powder-20°C 3 years4°C 2 yearsIn solvent-80°C 6 months-20°C 1 monthShipping at room temperature if less than 2 weeks.7.3 Specific end use(s)No data available.8. EXPOSURE CONTROLS/PERSONAL PROTECTION8.1 Control parametersComponents with workplace control parametersThis product contains no substances with occupational exposure limit values.8.2 Exposure controlsEngineering controlsEnsure adequate ventilation. Provide accessible safety shower and eye wash station.Personal protective equipmentEye protection Safety goggles with side-shields.Hand protection Protective gloves.Skin and body protection Impervious clothing.Respiratory protection Suitable respirator.Environmental exposure controls Keep the product away from drains, water courses or the soil. Cleanspillages in a safe way as soon as possible.9. PHYSICAL AND CHEMICAL PROPERTIES9.1 Information on basic physical and chemical propertiesAppearance Off-white to orange (Solid)Odor No data availableOdor threshold No data availablepH No data availableMelting/freezing point No data availableBoiling point/range No data availableFlash point No data availableEvaporation rate No data availableFlammability (solid, gas)No data availableUpper/lower flammability or explosive limits No data availableVapor pressure No data availableVapor density No data availableRelative density No data availableWater Solubility No data availablePartition coefficient No data availableAuto-ignition temperature No data availableDecomposition temperature No data availableViscosity No data availableExplosive properties No data availableOxidizing properties No data available9.2 Other safety informationNo data available.10. STABILITY AND REACTIVITY10.1 ReactivityNo data available.10.2 Chemical stabilityStable under recommended storage conditions.10.3 Possibility of hazardous reactionsNo data available.10.4 Conditions to avoidNo data available.10.5 Incompatible materialsStrong acids/alkalis, strong oxidising/reducing agents.10.6 Hazardous decomposition productsUnder fire conditions, may decompose and emit toxic fumes.Other decomposition products - no data available.11.TOXICOLOGICAL INFORMATION11.1 Information on toxicological effectsAcute toxicityClassified based on available data. For more details, see section 2Skin corrosion/irritationClassified based on available data. For more details, see section 2Serious eye damage/irritationClassified based on available data. For more details, see section 2Respiratory or skin sensitizationClassified based on available data. For more details, see section 2Germ cell mutagenicityClassified based on available data. For more details, see section 2CarcinogenicityIARC: No component of this product present at a level equal to or greater than 0.1% is identified as probable, possible or confirmed human carcinogen by IARC.ACGIH: No component of this product present at a level equal to or greater than 0.1% is identified as a potential or confirmed carcinogen by ACGIH.NTP: No component of this product present at a level equal to or greater than 0.1% is identified as a anticipated or confirmed carcinogen by NTP.OSHA: No component of this product present at a level equal to or greater than 0.1% is identified as a potential or confirmed carcinogen by OSHA.Reproductive toxicityClassified based on available data. For more details, see section 2Specific target organ toxicity - single exposureClassified based on available data. For more details, see section 2Specific target organ toxicity - repeated exposureClassified based on available data. For more details, see section 2Aspiration hazardClassified based on available data. For more details, see section 212. ECOLOGICAL INFORMATION12.1 ToxicityNo data available.12.2 Persistence and degradabilityNo data available.12.3 Bioaccumlative potentialNo data available.12.4 Mobility in soilNo data available.12.5 Results of PBT and vPvB assessmentPBT/vPvB assessment unavailable as chemical safety assessment not required or not conducted.12.6 Other adverse effectsNo data available.13. DISPOSAL CONSIDERATIONS13.1 Waste treatment methodsProductDispose substance in accordance with prevailing country, federal, state and local regulations.Contaminated packagingConduct recycling or disposal in accordance with prevailing country, federal, state and local regulations.14. TRANSPORT INFORMATIONDOT (US)This substance is considered to be non-hazardous for transport.IMDGThis substance is considered to be non-hazardous for transport.IATAThis substance is considered to be non-hazardous for transport.15. REGULATORY INFORMATIONSARA 302 Components:No chemicals in this material are subject to the reporting requirements of SARA Title III, Section 302.SARA 313 Components:This material does not contain any chemical components with known CAS numbers that exceed the threshold (De Minimis) reporting levels established by SARA Title III, Section 313.SARA 311/312 Hazards:No SARA Hazards.Massachusetts Right To Know Components:No components are subject to the Massachusetts Right to Know Act.Pennsylvania Right To Know Components:No components are subject to the Pennsylvania Right to Know Act.New Jersey Right To Know Components:No components are subject to the New Jersey Right to Know Act.California Prop. 65 Components:This product does not contain any chemicals known to State of California to cause cancer, birth defects, or anyother reproductive harm.16. OTHER INFORMATIONCopyright 2017 MedChemExpress. The above information is correct to the best of our present knowledge but does not purport to be all inclusive and should be used only as a guide. The product is for research use only and for experienced personnel. It must only be handled by suitably qualified experienced scientists in appropriately equipped and authorized facilities. The burden of safe use of this material rests entirely with the user. MedChemExpress disclaims all liability for any damage resulting from handling or from contact with this product.Caution: Product has not been fully validated for medical applications. For research use only.Tel: 609-228-6898 Fax: 609-228-5909 E-mail: tech@Address: 1 Deer Park Dr, Suite Q, Monmouth Junction, NJ 08852, USA。
化学仿制药参比制剂目录(第六十七批)
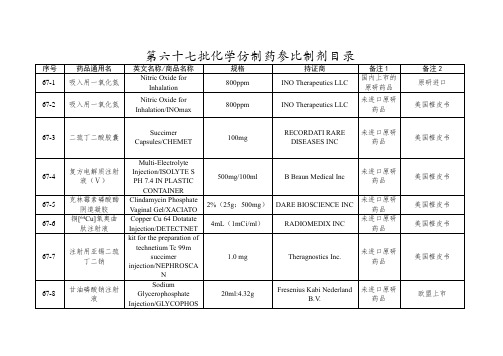
株式会社
未进口原研药品
67-22
普瑞巴林缓释片
Pregabalin Extended release Tablets/Lyrica Cr
165mg
Pf Prism CV/UPJOHN US 2 LLC
未进口原研药品
美国橙皮书
67-23
普瑞巴林缓释片
Pregabalin Extended release Tablets/Lyrica Cr
国际公认的同种药品
美国橙皮书
67-34
头孢地尼干混悬剂
Cefdinir for Oral Suspension
125mg/5mL
Aurobindo Pharma Limited
美国橙皮书
67-35
蔗糖铁注射液
Iron Sucrose Injection/Venofer
5ml:100mg铁和1.6g蔗糖
0.25%
(10ml:25mg)
Hospira Inc
未进口原研药品
美国橙皮书
67-27
左卡尼汀口服溶液
Levocarnitine Oral Solution/CARNITOR SF
10ml:1g
Leadiant Biosciences, Inc.
未进口原研药品
美国橙皮书
67-28
碳酸氢钠血滤置换液(钾4mmol/L)
Hemofiltration Replacement Fluid of Sodium Bicarbonate
(4mmol/L Potassium Calcium free)/PrismaSol
5000ml(250ml/4750ml)
BAXTER HEALTHCARE CORP
哌啶酸检测

迪信泰检测平台
哌啶酸检测
哌啶酸(Pipecolic acid),又称2-哌啶甲酸,是一种重要的刚性环状非蛋白质氨基酸,它既可以限定多肽的构象,还可作为不同化合物合成库中的多功能骨架,所以广泛用于许多手性药物和生物活性物质的制备。
哌啶酸的合成方法主要分为生物催化合成、化学不对称合成和光催化合成。
迪信泰检测平台采用液相质谱联用(LC-MS)的方法,使用Thermo Scientific的
U3000快速液相色谱对样品进行分离,Thermo Scientific™ Q Exactive™对样品进行鉴定,可高效、精准的检测哌啶酸的含量变化。
此外,我们还提供其他极性小分子系列检测服务,以满足您的不同需求。
LC-MS测定哌啶酸样本要求:
1. 请确保样本量大于0.2g或者0.2mL。
周期:2~3周。
项目结束后迪信泰检测平台将会提供详细中英文双语技术报告,报告包括:
1. 实验步骤(中英文)。
2. 相关参数(中英文)。
3.质谱图片。
4. 原始数据。
5. 哌啶酸含量信息。
迪信泰检测平台可根据需求定制其他物质测定方案,具体可免费咨询技术支持。
乙酰胆碱酯酶抑制剂

上海应用技术学院研究生课程《高等天然产物化学》试卷2014 / 2015 学年第1 学期课程代码:NX0702013论文题目:乙酰胆碱酯酶抑制剂的研究进展姓名:芮银146061414康满满146061409专业:制药工程学院:化工学院乙酰胆碱酯酶抑制剂的研究进展芮银,陈祎桐,康满满摘要:本文阐述了乙酰胆碱酯酶抑制剂(AChEI)的研究进展,介绍了用于药物治疗的乙酰胆碱酯酶抑制剂的各种来源如植物、微生物等,及其抑制乙酰胆碱的活性物质。
在此基础上,总结了几种现代分析技术,对AChEIs进行筛选,大大加快AD药物资源的开发利用进程。
这些方法主要有基于比色法的Ellman's法及相关的改进方法、薄层显色法、荧光显色法、电喷雾质谱法等。
但是,到目前为止,现代分析技术在AD药物资源中的应用还处在起步阶段。
关键词:乙酰胆碱酯酶抑制剂,筛选方法,薄层显色法,荧光显色法The progress of acetylcholinesteraseinhibitorsRui Yin, Chen Yitong, Kang ManmanAbstract:In this artical, the research elaborates progress of acetylcholinesterase inhibitors (AChEI), and introduces a variety of sources for drug treatment acetylcholinesterase inhibitors such as plants, microorganisms, and its active ingredients. On this basis, the review summarizes several modern analytic techniques such as Ellman's method which based on the colorimetric method, TLC chromogenic method, fluorescent color method, Electrospray ionization mass spectrometry and so on. However, at present, the application of modern analytic techniques in AD drug resources is still in infancy.Key word: Acetylcholinesterase inhibitors, Screening Methods, TLC chromogenic method, Fluorescent color method目录摘要.................................................................................................错误!未定义书签。
Alda-1_DataSheet_MedChemExpress

Inhibitors, Agonists, Screening Libraries Data SheetBIOLOGICAL ACTIVITY:Alda–1 is a potent ALDH2 agonist, which significantly improves ALDH2 activity.In Vivo: Alda–1 treatment results in a significant decrease of 4–HNE–protein content in the plasma of apoE -/- mice. Alda–1administration leads to a slight increase in gene expression related to neurogenesis (Nog ), mitochondrial biogenesis (CYTB , ND1),and apoptosis (Bax , Gsk3b ) in the Hp of apoE -/- mice. Alda–1 administration leads to 2 and 10 differentially expressed proteins in theFCx and Hp of apoE -/- mice, respectively [1]. Alda–1 (1.5 mg/kg, b.w., i.p.) administration significantly increases the climbing time,tends to reduce the immobility time and increases the swimming time of the prenatally stressed rats in the forced swim test.Moreover, treatment of prenatally stressed rats with Alda–1 significantly increases number of entries into the open arms of the maze and the time spent therein, as assessed by elevated plus–maze test [2]. Alda–1 (8.5 mg/kg, i.p.) with glucose significantly lowers 4–HNE and FJB–positive cells in the cerebral cortex of Alda–1–treated rats than in DMSO–treated rats 24 h after glucose administration [3]. Alda–1 (10 mg/kg per day) treatment prevents aldehydic overload, mitochondrial dysfunction and improves ventricular function in post–MI cardiomyopathy rats [4].PROTOCOL (Extracted from published papers and Only for reference)Cell Assay:[2]Spleen cells (4×106 cells/mL) are stimulated by optimal concentrations of concanavalin A (Con A; 2.5μg/mL and 0.6 μg/mL) and lipopolysaccharide (LPS, 5 μg/mL) and are incubated in 96–well plates at final volume of 0.2mL for 72 h. Cell proliferation is determined by adding 0.5 μCi of [3H]–thymidine per well at 16 h before the end of the incubation.The cultures are harvested with an automatic cell harvester, and [3H] thymidine incorporation is assessed using a liquid scintillationcounter.Animal Administration: Alda–1 is dissolved in 1 mL/kg b.w. DMSO/water 50/50.[2]After behavioral verification at three months of age,the animals are divided into the following four groups: control, control + Alda–1, prenatally stressed and prenatally stressed + Alda–1(6 animals per group). Alda–1 injections are given intraperitoneally (i.p.) once daily at a dose of 1.5 mg/kg b.w. (dissolved in 1 mL/kg b.w. DMSO/water 50/50) for 14 days. At the same time, the control and prenatally stressed rats receive 1 mL/kg b.w. DMSO/water 50/50. The injections of Alda–1 and vehicle are given between 10 a.m and 11 a.m. In the last five days of Alda–1 treatment the behavioral parameters in the elevated plus maze test and then in the forced swim test are measured.References:[1]. Stachowicz A, et al. Proteomic Analysis of Mitochondria–Enriched Fraction Isolated from the Frontal Cortex and Hippocampus of Apolipoprotein E Knockout Mice Treated with Alda–1, an Activator of Mitochondrial Aldehyde Dehydrogenase (ALDH2). Int J Mol Sci.[2]. Stachowicz A, et al. The impact of mitochondrial aldehyde dehydrogenase (ALDH2) activation by Alda–1 on the behavioral and biochemical disturbances in animal model of depression. Brain Behav Immun. 2016 Jan;51:144–53.Product Name:Alda–1Cat. No.:HY-18936CAS No.:349438-38-6Molecular Formula:C 15H 11Cl 2NO 3Molecular Weight:324.16Target:Aldehyde Dehydrogenase (ALDH)Pathway:Metabolic Enzyme/Protease Solubility:DMSO: ≥ 51 mg/mL[3]. Ikeda T, et al. Effects of Alda–1, an Aldehyde Dehydrogenase–2 Agonist, on Hypoglycemic Neuronal Death. PLoS One. 2015 Jun 17;10(6):e0128844.[4]. Gomes KM, et al. Aldehydic load and aldehyde dehydrogenase 2 profile during the progression of post–myocardial infarction cardiomyopathy: benefits of Alda–1. Int J Cardiol. 2015 Jan 20;179:129–138.Caution: Product has not been fully validated for medical applications. For research use only.Tel: 609-228-6898 Fax: 609-228-5909 E-mail: tech@Address: 1 Deer Park Dr, Suite Q, Monmouth Junction, NJ 08852, USA。
