BMS-833923_COA_09099_MedChemExpress
碧云天生物技术产品说明书.pdf_1694034956.947723
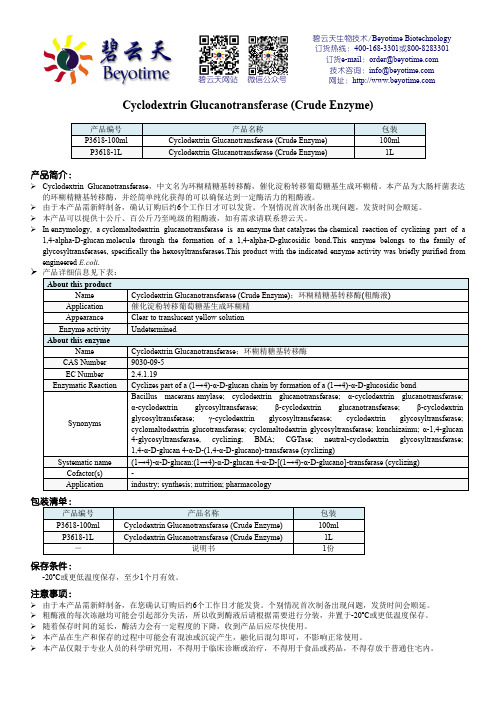
碧云天生物技术/Beyotime Biotechnology订货热线:400-168-3301或800-8283301订货e-mail:******************技术咨询:*****************网址:碧云天网站微信公众号Cyclodextrin Glucanotransferase (Crude Enzyme)产品编号产品名称包装P3618-100ml Cyclodextrin Glucanotransferase (Crude Enzyme) 100mlP3618-1L Cyclodextrin Glucanotransferase (Crude Enzyme) 1L产品简介:Cyclodextrin Glucanotransferase,中文名为环糊精糖基转移酶,催化淀粉转移葡萄糖基生成环糊精。
本产品为大肠杆菌表达的环糊精糖基转移酶,并经简单纯化获得的可以确保达到一定酶活力的粗酶液。
由于本产品需新鲜制备,确认订购后约6个工作日才可以发货。
个别情况首次制备出现问题,发货时间会顺延。
本产品可以提供十公斤、百公斤乃至吨级的粗酶液,如有需求请联系碧云天。
In enzymology, a cyclomaltodextrin glucanotransferase is an enzyme that catalyzes the chemical reaction of cyclizing part of a 1,4-alpha-D-glucan molecule through the formation of a 1,4-alpha-D-glucosidic bond.This enzyme belongs to the family of glycosyltransferases, specifically the hexosyltransferases.This product with the indicated enzyme activity was briefly purified from engineered E.coli.产品详细信息见下表:About this productName Cyclodextrin Glucanotransferase (Crude Enzyme);环糊精糖基转移酶(粗酶液)Application 催化淀粉转移葡萄糖基生成环糊精Appearance Clear to translucent yellow solutionEnzyme activity UndeterminedAbout this enzymeName Cyclodextrin Glucanotransferase;环糊精糖基转移酶CAS Number 9030-09-5EC Number 2.4.1.19Enzymatic Reaction Cyclizes part of a (1→4)-α-D-glucan chain by formation of a (1→4)-α-D-glucosidic bondSynonyms Bacillus macerans amylase; cyclodextrin glucanotransferase; α-cyclodextrin glucanotransferase; α-cyclodextrin glycosyltransferase; β-cyclodextrin glucanotransferase; β-cyclodextrin glycosyltransferase; γ-cyclodextrin glycosyltransferase; cyclodextrin glycosyltransferase; cyclomaltodextrin glucotransferase; cyclomaltodextrin glycosyltransferase; konchizaimu; α-1,4-glucan 4-glycosyltransferase, cyclizing; BMA; CGTase; neutral-cyclodextrin glycosyltransferase; 1,4-α-D-glucan 4-α-D-(1,4-α-D-glucano)-transferase (cyclizing)Systematic name (1→4)-α-D-glucan:(1→4)-α-D-glucan 4-α-D-[(1→4)-α-D-glucano]-transferase (cyclizing)Cofactor(s) -Application industry; synthesis; nutrition; pharmacology包装清单:产品编号产品名称包装P3618-100ml Cyclodextrin Glucanotransferase (Crude Enzyme) 100mlP3618-1L Cyclodextrin Glucanotransferase (Crude Enzyme) 1L-说明书1份保存条件:-20ºC或更低温度保存,至少1个月有效。
四种烷基咪唑磷酸酯离子液体的热力学性质

化工进展Chemical Industry and Engineering Progress2024 年第 43 卷第 3 期四种烷基咪唑磷酸酯离子液体的热力学性质刘泽鹏,曾纪珺,唐晓博,赵波,韩升,廖袁淏,张伟(西安近代化学研究所氟氮化工资源高效开发与利用国家重点实验室,陕西 西安 710065)摘要:针对烷基咪唑磷酸酯离子液体的热物性数据较少的问题,本文在常压下测定了1-乙基-3-甲基咪唑磷酸二氢盐([EMIM][DHP])、1-乙基-3-甲基咪唑磷酸二甲酯盐([EMIM][DMP])、1-乙基-3-甲基咪唑磷酸二乙酯盐([EMIM][DEP])、1-丁基-3-甲基咪唑磷酸二丁酯盐([BMIM][DBP])四种烷基咪唑磷酸酯离子液体的密度、黏度(293.15~353.15K )和电导率(293.15~343.15K ),并且测定了四种离子液体的热稳定性。
结果表明,离子液体的密度、黏度随温度的升高而减小,而电导率随温度的升高而增大。
采用自然对数方程关联四种离子液体的密度,根据实验值计算到了离子液体体积性质;采用VFT 方程关联离子液体黏度和电导率,其中密度与电导率的实验值与模型相关系数R 2达到0.9999,黏度相关系数R 2达到0.99999,实验测定的数据与模型一致;四种离子液体的热稳定性相近,分解温度均在271.9~278.6℃范围内;瓦尔登规则分析表明,四种烷基咪唑磷酸酯离子液体符合Walden 规则,而[EMIM][DMP]和[EMIM][DEP]被归类为“good ionic liquids ”。
关键词:烷基咪唑磷酸酯离子液体;密度;黏度;电导率;热稳定性中图分类号:TQ013.1 文献标志码:A 文章编号:1000-6613(2024)03-1484-08Thermodynamic properties of four alkyl imidazolium phosphate ionicliquidsLIU Zepeng ,ZENG Jijun ,TANG Xiaobo ,ZHAO Bo ,HAN Sheng ,LIAO Yuanhao ,ZHANG Wei(State Key Laboratory of Fluorine & Nitrogen Chemicals, Xi ’an Modern Chemistry Research Institute, Xi ’an 710065,Shaanxi, China)Abstract: The density, viscosity, and conductivity of 1-ethyl-3-methylimidazolium dihydrogen-phosphate ([EMIM][DHP]), 1-ethyl-3-methylimidazolium dimethylphosphate ([EMIM][DMP]), 1-ethyl-3-methylimidazolium diethylphosphate ([EMIM][DEP]) and 1-butyl-3-methylimidazolium dibutyl-phosphate ([BMIM][DBP]) ionic liquids were measured in the temperature range of 293.15K to 353.15K under ambient conditions. Some important volumetric properties, including the isobaric thermal expansion coefficients, molecular volume, standard entropy and lattice potential energy were calculated from the experimental density values. The thermal gravimetric analysis was performed in the temperature range of 35℃ to 700℃, resulting in thermal decomposition temperatures up to 271.9—278.6℃. The Walden rule analysis demonstrated that four phosphate ionic liquids complied with the Walden rule well, while [EMIM][DMP] and [EMIM][DEP] were classified as “good ionic liquids ”.Keywords: alkyl imidazolium phosphate ionic liquids; density; viscosity; conductivity; thermal stability研究开发DOI :10.16085/j.issn.1000-6613.2023-1722收稿日期:2023-09-28;修改稿日期:2023-12-05。
安徽省环保厅关于赛乐谱制药有限公司国家一类抗肿瘤新药紫衫肽原料药生产项目环保预审的复函

安徽省环保厅关于赛乐谱制药有限公司国家一类抗肿瘤新药紫衫肽原料药生产项目环保预审的复函
文章属性
•【制定机关】安徽省环境保护厅
•【公布日期】2016.02.04
•【字号】
•【施行日期】2016.02.04
•【效力等级】地方规范性文件
•【时效性】现行有效
•【主题分类】环境保护其他规定
正文
安徽省环保厅关于赛乐谱制药有限公司国家一类抗肿瘤新药紫衫肽原料药生产项目环保预审的复函
淮南市环保局:
《关于赛乐谱制药有限公司国家一类抗肿瘤新药紫衫肽原料药生产项目进行预审的请示》(淮环秘〔2016〕5号)收悉。
经研究,现函复如下:
赛乐谱制药有限公司国家一类抗肿瘤新药紫衫肽原料药生产项目拟选址于安徽凤台经济开发区。
根据《关于安徽凤台经济开发区总体发展规划环境影响报告书审查意见的函》(皖环函〔2013〕368号),凤台经济开发区主导产业为煤化工、矿山机械、纺织服装、农副产品加工。
该项目属于医药化工行业,与凤台经济开发区产业发展规划要求不符,且凤台经济开发区未按要求配套建设集中污水处理设施。
因此,不同意该项目开展环评等前期工作。
高效液相色谱在过氧化氢异丙苯合成产物分析中的应用

高效液相色谱在过氧化氢异丙苯合成产物分析中的应用近年来,随着精确分析技术的发展,高效液相色谱在过氧化氢异丙苯合成产物分析中得到了广泛应用。
高效液相色谱(HPLC)是一种涤模柔性、灵活性和立体选择性强的分子筛选技术,目前已成为最流行的分析方法之一,在药物合成、分析、研究和检测等领域中受到了极大的重视。
过氧化氢异丙苯合成产物是一种重要的有机结构物,它可以用作药物的原料或中间体,用于制备复杂的仿生药物、抗病毒药物等。
由于产品结构经常复杂且易变,高效液相色谱技术正在发挥重要作用,以确定合成的每一步的复杂产物的结构和纯度。
高效液相色谱技术是在过氧化氢异丙苯合成产物分析中的首选技术。
高效液相色谱可以快速准确地对样品进行分离,并可以进行高灵敏度的测量。
相比其他分析技术,高效液相色谱技术更加简单、快速、高灵敏,而且只需使用少量的样品。
在过氧化氢异丙苯合成产物分析中,使用高效液相色谱技术可以快速准确地确定样品的结构;它能有效分离混合物,可以在短时间内完成分离,从而节省时间和金钱。
此外,高效液相色谱还可以鉴定合成过程中产生的异构体。
另外,高效液相色谱还可以检测样品中的有机物、水溶性物质和微量元素。
高效液相色谱技术可以检测样品中成分的组成,包括它们的量和种类,从而为研究者提供完整的信息。
高效液相色谱技术在过氧化氢异丙苯合成产物分析中的应用受到了极大的重视。
高效液相色谱技术可以准确测定各种复杂的产物,为药物合成、分析、研究和检测等领域的研究提供了有力的支持。
总而言之,高效液相色谱技术在过氧化氢异丙苯合成产物分析中不仅可以得到准确的结果,而且可以节省更多时间和金钱。
只有这种先进的技术可以满足药物合成和分析的需求,才能更好地满足科学研究和临床应用的需要。
BMS-986205-SDS-MedChemExpress

Inhibitors, Agonists, Screening LibrariesSafety Data Sheet Revision Date:Oct.-10-2018Print Date:Oct.-10-20181. PRODUCT AND COMPANY IDENTIFICATION1.1 Product identifierProduct name :BMS-986205Catalog No. :HY-101560CAS No. :1923833-60-61.2 Relevant identified uses of the substance or mixture and uses advised againstIdentified uses :Laboratory chemicals, manufacture of substances.1.3 Details of the supplier of the safety data sheetCompany:MedChemExpress USATel:609-228-6898Fax:609-228-5909E-mail:sales@1.4 Emergency telephone numberEmergency Phone #:609-228-68982. HAZARDS IDENTIFICATION2.1 Classification of the substance or mixtureGHS Classification in accordance with 29 CFR 1910 (OSHA HCS)Skin irritation (Category 2), H315Serious eye damage (Category 1), H318Respiratory sensitisation (Category 1), H334Skin sensitisation (Category 1), H317Germ cell mutagenicity (Category 2), H341Reproductive toxicity (Category 2), H361Specific target organ toxicity - single exposure (Category 1), H370Specific target organ toxicity - single exposure (Category 3), Respiratory system, H335Chronic aquatic toxicity (Category 4), H4132.2 GHS Label elements, including precautionary statementsPictogramSignal word DangerHazard statement(s)H315: Causes skin irritation.H317: May cause an allergic skin reactionH318: Causes serious eye damage.H334: May cause allergy or asthma symptoms or breathing difficulties if inhaled.H335: May cause respiratory irritation.H341: Suspected of causing genetic defects.H361: Suspected of damaging fertility or the unborn child.H370: Causes damage to organs.H413: May cause long lasting harmful effects to aquatic life.Precautionary statement(s)P201: Obtain special instructions before use.P202: Do not handle until all safety precautions have been read and understood.P260: Do not breathe dust ⁄ fume ⁄ gas ⁄ mist ⁄ vapors ⁄ spray.P264: Wash skin thoroughly after handling.P270: Do not eat, drink or smoke when using this product.P271: Use only outdoors or in a well-ventilated area.P272: Contaminated work clothing should not be allowed out of the workplace.P273: Avoid release to the environment.P280: Wear protective gloves ⁄ eye protection ⁄ face protection.P302 + P352: IF ON SKIN: Wash with plenty of soap and water.P304 + P340: IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for breathing.P305 + P351 + P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do.Continue rinsing.P310: Immediately call a POISON CENTER ⁄doctor.P321: Specific treatment (see supplemental first aid instructions on this label).P333 + P313: If skin irritation or rash occurs: Get medical advice ⁄ attention.P362: Take off contaminated clothing and wash before reuse.P403 + P233: Store in a well-ventilated place. Keep container tightly closed.P405: Store locked up.P501: Dispose of contents ⁄ container to an approved waste disposal plant.2.3 Other hazardsNone.3. COMPOSITION/INFORMATION ON INGREDIENTS3.1 SubstancesSynonyms:NoneFormula:C24H24ClFN2OMolecular Weight:410.91CAS No. :1923833-60-64. FIRST AID MEASURES4.1 Description of first aid measuresEye contactRemove any contact lenses, locate eye-wash station, and flush eyes immediately with large amounts of water. Separate eyelids with fingers to ensure adequate flushing. Promptly call a physician.Skin contactRinse skin thoroughly with large amounts of water. Remove contaminated clothing and shoes and call a physician.InhalationImmediately relocate self or casualty to fresh air. If breathing is difficult, give cardiopulmonary resuscitation (CPR). Avoid mouth-to-mouth resuscitation.IngestionWash out mouth with water; Do NOT induce vomiting; call a physician.4.2 Most important symptoms and effects, both acute and delayedThe most important known symptoms and effects are described in the labelling (see section 2.2).4.3 Indication of any immediate medical attention and special treatment neededTreat symptomatically.5. FIRE FIGHTING MEASURES5.1 Extinguishing mediaSuitable extinguishing mediaUse water spray, dry chemical, foam, and carbon dioxide fire extinguisher.5.2 Special hazards arising from the substance or mixtureDuring combustion, may emit irritant fumes.5.3 Advice for firefightersWear self-contained breathing apparatus and protective clothing.6. ACCIDENTAL RELEASE MEASURES6.1 Personal precautions, protective equipment and emergency proceduresUse full personal protective equipment. Avoid breathing vapors, mist, dust or gas. Ensure adequate ventilation. Evacuate personnel to safe areas.Refer to protective measures listed in sections 8.6.2 Environmental precautionsTry to prevent further leakage or spillage. Keep the product away from drains or water courses.6.3 Methods and materials for containment and cleaning upAbsorb solutions with finely-powdered liquid-binding material (diatomite, universal binders); Decontaminate surfaces and equipment by scrubbing with alcohol; Dispose of contaminated material according to Section 13.7. HANDLING AND STORAGE7.1 Precautions for safe handlingAvoid inhalation, contact with eyes and skin. Avoid dust and aerosol formation. Use only in areas with appropriate exhaust ventilation.7.2 Conditions for safe storage, including any incompatibilitiesKeep container tightly sealed in cool, well-ventilated area. Keep away from direct sunlight and sources of ignition.Recommended storage temperature:Powder-20°C 3 years4°C 2 yearsIn solvent-80°C 6 months-20°C 1 monthShipping at room temperature if less than 2 weeks.7.3 Specific end use(s)No data available.8. EXPOSURE CONTROLS/PERSONAL PROTECTION8.1 Control parametersComponents with workplace control parametersThis product contains no substances with occupational exposure limit values.8.2 Exposure controlsEngineering controlsEnsure adequate ventilation. Provide accessible safety shower and eye wash station.Personal protective equipmentEye protection Safety goggles with side-shields.Hand protection Protective gloves.Skin and body protection Impervious clothing.Respiratory protection Suitable respirator.Environmental exposure controls Keep the product away from drains, water courses or the soil. Cleanspillages in a safe way as soon as possible.9. PHYSICAL AND CHEMICAL PROPERTIES9.1 Information on basic physical and chemical propertiesAppearance White to off-white (Solid)Odor No data availableOdor threshold No data availablepH No data availableMelting/freezing point No data availableBoiling point/range No data availableFlash point No data availableEvaporation rate No data availableFlammability (solid, gas)No data availableUpper/lower flammability or explosive limits No data availableVapor pressure No data availableVapor density No data availableRelative density No data availableWater Solubility No data availablePartition coefficient No data availableAuto-ignition temperature No data availableDecomposition temperature No data availableViscosity No data availableExplosive properties No data availableOxidizing properties No data available9.2 Other safety informationNo data available.10. STABILITY AND REACTIVITY10.1 ReactivityNo data available.10.2 Chemical stabilityStable under recommended storage conditions.10.3 Possibility of hazardous reactionsNo data available.10.4 Conditions to avoidNo data available.10.5 Incompatible materialsStrong acids/alkalis, strong oxidising/reducing agents.10.6 Hazardous decomposition productsUnder fire conditions, may decompose and emit toxic fumes.Other decomposition products - no data available.11.TOXICOLOGICAL INFORMATION11.1 Information on toxicological effectsAcute toxicityClassified based on available data. For more details, see section 2Skin corrosion/irritationClassified based on available data. For more details, see section 2Serious eye damage/irritationClassified based on available data. For more details, see section 2Respiratory or skin sensitizationClassified based on available data. For more details, see section 2Germ cell mutagenicityClassified based on available data. For more details, see section 2CarcinogenicityIARC: No component of this product present at a level equal to or greater than 0.1% is identified as probable, possible or confirmed human carcinogen by IARC.ACGIH: No component of this product present at a level equal to or greater than 0.1% is identified as a potential or confirmed carcinogen by ACGIH.NTP: No component of this product present at a level equal to or greater than 0.1% is identified as a anticipated or confirmed carcinogen by NTP.OSHA: No component of this product present at a level equal to or greater than 0.1% is identified as a potential or confirmed carcinogen by OSHA.Reproductive toxicityClassified based on available data. For more details, see section 2Specific target organ toxicity - single exposureClassified based on available data. For more details, see section 2Specific target organ toxicity - repeated exposureClassified based on available data. For more details, see section 2Aspiration hazardClassified based on available data. For more details, see section 2Additional informationRTECS No.: DA8340700Alopecia., Liver injury may occur., Kidney injury may occur., Nausea, Headache, Vomiting, bone marrow depressionThis information is based on our current knowledge. However the chemical, physical, and toxicological properties have not been completely investigated.12. ECOLOGICAL INFORMATION12.1 ToxicityNo data available.12.2 Persistence and degradabilityNo data available.12.3 Bioaccumlative potentialNo data available.12.4 Mobility in soilNo data available.12.5 Results of PBT and vPvB assessmentPBT/vPvB assessment unavailable as chemical safety assessment not required or not conducted.12.6 Other adverse effectsNo data available.13. DISPOSAL CONSIDERATIONS13.1 Waste treatment methodsProductDispose substance in accordance with prevailing country, federal, state and local regulations.Contaminated packagingConduct recycling or disposal in accordance with prevailing country, federal, state and local regulations.14. TRANSPORT INFORMATIONDOT (US)This substance is considered to be non-hazardous for transport.IMDGThis substance is considered to be non-hazardous for transport.IATAThis substance is considered to be non-hazardous for transport.15. REGULATORY INFORMATIONSARA 302 ComponentsNo chemicals in this material are subject to the reporting requirements of SARA Title III, Section 302.SARA 313 ComponentsNo chemicals in this material are subject to the reporting requirements of SARA Title III, Section 313.SARA 311/312 HazardsAcute Health Hazard, Chronic Health Hazard16. OTHER INFORMATIONCopyright 2018 MedChemExpress. The above information is correct to the best of our present knowledge but does not purport to be all inclusive and should be used only as a guide. The product is for research use only and for experienced personnel. It must only be handled by suitably qualified experienced scientists in appropriately equipped and authorized facilities. The burden of safe use of this material rests entirely with the user. MedChemExpress disclaims all liability for any damage resulting from handling or from contact with this product.Caution: Product has not been fully validated for medical applications. For research use only.Tel: 609-228-6898 Fax: 609-228-5909 E-mail: tech@Address: 1 Deer Park Dr, Suite Q, Monmouth Junction, NJ 08852, USA。
一种由环氧乙烷制备3-羟基丙酸酯及1,3-丙二醇的方法[发明专利]
![一种由环氧乙烷制备3-羟基丙酸酯及1,3-丙二醇的方法[发明专利]](https://img.taocdn.com/s3/m/fc5538f659eef8c75ebfb325.png)
专利名称:一种由环氧乙烷制备3-羟基丙酸酯及1,3-丙二醇的方法
专利类型:发明专利
发明人:吕志果,郭振美,王恒生
申请号:CN201010501535.9
申请日:20101004
公开号:CN101973881A
公开日:
20110216
专利内容由知识产权出版社提供
摘要:本发明公开了一种以环氧乙烷为原料制备3-羟基丙酸酯及1,3-丙二醇的方法。
该方法选用适当的羰基钴功能化离子液体催化剂及相应的溶剂和促进剂,在同一催化体系下实现环氧乙烷氢酯基化制备3-羟基丙酸酯及其加氢合成1,3-丙二醇。
该羰基钴功能化离子液体催化体系具有催化活性好、选择性高、产物易分离、易循环等优点。
申请人:青岛科技大学
地址:266061 山东省青岛市崂山区松岭路69号
国籍:CN
更多信息请下载全文后查看。
胶束电动毛细管色谱安培检测中药马齿苋中多巴胺和去甲肾上腺素

!"#"$%&’(#&)’ )* !)+(%&’" (’, -)$"+&’"+.$&’" &’ !"#$%&’(’ "&)#’()’ /0 12 3&4"55($ 65"4#$)7&’"#&4 8(+&55($2 8.$)%(#)9$(+.2 :&#. ;%+"$)%"#$&4 !"#"4#&)’
56%7 89:;1<;= ,>?3% @:9#";= ,A(3%7 (";=B9;= ,CD2%7 E9:;FG9 , 53%7 HF:;= ,C? 7G"I:;=
( #$%&’($) *+,’-$%’.,$/+&0 12 3 4 52 *6(%6, ,7$)&$( 8(9%&%:%6 ’; *+6-&0$) <+=9&09 , >+6 *+&(696 50$?6-= ’; !0&6(069 ,7$)&$( !!"#!! ,*+&($ )
;1<#$(4# :3 -<JF"K L:M<K "; -9N<##:O <#<NJO"P9;<J9N N:’9##:OB NFO"-:J"=O:’FB I9JF <#<NJO"4 NF<-9N:# K<J<NJ9";( Q6@H46H ) I:M K<R<#"’<K 1"O JF< M9-G#J:;<"GM K<J<O-9;:J9"; "1 ;"O<’94 ;<’FO9;<( %6 ):;K K"’:-9;<( *3 ) 9; HF9;<M< F<OL:# ’#:;J <SJO:NJM "1 <’,%:)$0$ ’)6,$06$ /$ TF< <11<NJM "1 M<R<O:# 1:NJ"OM MGNF :M JF< :N9K9JB :;K N";N<;JO:J9"; "1 OG;;9;= LG11<O ,M"K9GK"K<NB#MG#1:J<( )*) )N";N<;JO:J9"; :;K K<J<NJ9"; ’"J<;J9:# I<O< 9;R<MJ9=:J<K J" :NUG9O< JF< "’4 J9-G- N";K9J9";M$ TF< JI" :;:#BJ<M N"G#K L< I<## M<’:O:J<K I9JF9; $! -9; 9; : 5. N- N:’9##:OB :J JF< M<’:O:J9"; R"#J:=< "1 !# P! 9; : $" --"# . / ’F"M’F:J< LG11<O( ’( 5G !+ ) N";J:9;9;= $" --"# . / )*)$ TF< <SN<##<;J #9;<:O9JB I:M "LJ:9;<K 9; JF< N";N<;JO:J9"; O:;=< 1O"- $G " F $" + 5 J" #G " F $" + . -"# . /$ TF< K<J<NJ9"; #9-9JM "1 N";N<;JO:J9"; I<O< . $ ! F $" + 0 -"# . / 1"O %6 :;K ,G 0 F $" + 0 -"# . / 1"O *3 :;K JF"M< "1 UG:;J9JB I<O< "G .$ 1-"# 1"O %6 :;K $G .# 1-"# 1"O *3 :J : M9=4 ;:#4J"4;"9M< O:J9" "1 + $ TF9M -<JF"K I:M MGNN<MM1G##B GM<K 9; JF< :;:#BM9M "1 <’,%:)$0$ ’)6,$06$ /$ I9JF"GJ K<O9R:J9V:J9"; ’O"N<KGO< ,:;K O<:# :R<O:=< N";J<;JM "1 %6 :;K *3 9; <’,%:)$0$ ’)6,@ $06$ /$ I<O< " $ "$# W :;K " $ !" W ,O<M’<NJ9R<#B$ ="2 :)$,< :-9N<##:O <#<NJO"P9;<J9N N:’9##:OB NFO"-:J"=O:’FB ;:-’<O"-<JO9N K<J<NJ9"; ;K"’:4 -9;< ;;"O<’9;<’FO9;< ;<’,%:)$0$ ’)6,$06$ /$ & & 马齿苋在中国 不 仅 是 一 种 可 食 用 的 野 菜, 而且 还是应用广泛的传 统 中 草 药, 可用于减轻疼痛和消 肿。马 齿 苋 还 有 抗 菌、 抗 病 毒、 抗 衰 老、 治疗糖尿病 及增强免 疫 力 的 作 用
德谷门冬双胰岛素注射液治疗2_型糖尿病临床效果及安全性探讨

DOI:10.16658/ki.1672-4062.2023.17.098德谷门冬双胰岛素注射液治疗2型糖尿病临床效果及安全性探讨林生,谢平,陈予福州市长乐区人民医院内分泌科,福建福州350200[摘要]目的研究德谷门冬双胰岛素注射液治疗2型糖尿病的临床效果及安全性。
方法选取于2022年7月—2023年4月福州市长乐区人民医院收治的2型糖尿病患者98例为研究对象,采用随机抓阄法分为两组,每组49例。
两组均联用常规降糖药物治疗,对照组采用甘精胰岛素注射液治疗,观察组采用德谷门冬双胰岛素注射液治疗。
对比两组临床治疗效果、临床症状好转时间和胰岛素用量情况、糖代谢指标、胰岛素功能指标、不良反应发生情况、心血管不良事件发生情况。
结果观察组总有效率高于对照组,差异有统计学意义(P<0.05)。
观察组尿酮体转阴时间、血糖达标时间、胰岛素用量均优于对照组,差异有统计学意义(P< 0.05)。
观察组空腹血糖、餐后2 h血糖、糖化血红蛋白均低于对照组,差异有统计学意义(P<0.05)。
观察组胰岛β细胞功能指数高于对照组,胰岛素抵抗指数、空腹胰岛素低于对照组,差异有统计学意义(P<0.05)。
两组恶心呕吐、倦怠乏力、低血糖总发生率比较,差异无统计学意义(P>0.05)。
两组心绞痛、心力衰竭总发生率比较,差异无统计学意义(P>0.05)。
结论德谷门冬双胰岛素注射液治疗2型糖尿病临床效果显著优于甘精胰岛素注射液,但是治疗安全性无显著变化。
[关键词] 2型糖尿病;德谷门冬双胰岛素注射液;不良反应;心血管不良事件[中图分类号] R59 [文献标识码] A [文章编号] 1672-4062(2023)09(a)-0098-04Discussion on the Clinical Effect and Safety of Insulin Degludec and Insu⁃lin Aspart Injection in the Treatment of Type 2 Diabetes MellitusLIN Sheng, XIE Ping, CHEN YuDepartment of Endocrinology, Changle District People's Hospital, Fuzhou, Fujian Province, 350200 China[Abstract] Objective To study the clinical effect and safety of insulin degludec and insulin aspart injection in the treatment of type 2 diabetes mellitus. Methods A total of 98 patients with type 2 diabetes admitted to Fuzhou Changle District People's Hospital from July 2022 to April 2023 were selected as the study objects and divided into two groups with 49 cases in each group by random lottery method. Both groups were treated with conventional hypoglycemic drugs, the control group was treated with insulin glargine injection, and the observation group was treated with Degu asparton double insulin injection. The clinical therapeutic effect, time of improvement of clinical symptoms, insulin dosage, glucose metabolism index, insulin function index, occurrence of adverse reactions and cardiovascular adverse events were compared between the two groups. Results The total effective rate of the observation group was higher than that of the control group, and the difference was statistically significant (P<0.05). The time of urine ketone body turning negative, blood glucose reaching standard and insulin dosage in observation group were better than those in control group, and the differences were statistically significant (P<0.05). Fasting plasma glucose, 2-hour postprandial blood glucose and glycated hemoglobin in the observation group were lower than those in the control group, and the differences were statistically significant (P<0.05). The function index of islet β cells in observation group was higher than that in control group, the insulin resistance index and fasting insulin was lower than that in control group, the dif⁃ference was statistically significant (P<0.05). There was no statistically significant difference in the total incidence of [作者简介]林生(1981-),男,本科,副主任医师,研究方向为糖尿病及其并发症的相关临床研究。
BMS-833923 1059734-66-5 GlpBio

Product Data SheetProduct Name:BMS-833923Cat. No.:GC13628Chemical PropertiesCas No.1059734-66-5Chemical Name N-[2-methyl-5-(methylaminomethyl)phenyl]-4-[(4-phenylquinazolin-2-yl)amino]benzamideCanonical SMILES CC1=C(C=C(C=C1)CNC)NC(=O)C2=CC=C(C=C2)NC3=NC4=CC=CC=C4C(=N3)C5=CC =CC=C5Formula C30H27N5O M.Wt473.57Solubility ≥47.4 mg/mL in DMSO, ≥5.14mg/mL in EtOH with ultrasonicand warming, <2.6 mg/mL in H2OStorage Store at -20°CGeneral tips For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months.Shipping Condition Evaluation sample solution : ship with blue ice All other available size: ship with RT , or blue ice upon request.Structure实验参考方法Cell experiment [1]:Product Data SheetCell lines OE19 (JROECL19) and OE33 (JROECL33) esophageal adenocarcinoma(EAC) cell linesPreparation method The solubility of this compound in DMSO is >47.4mg/mL. General tips for obtaining a higher concentration: Please warm the tube at 37 ℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months.Reacting condition0 to 100 μM for 24 and 48 hrApplications In OE19 and OE33 cells, BMS-833923 (10 μM) inhibited cell proliferation with the IC50 of 10 μM. BMS-833923 (25 μM) completely inhibited cell proliferation. In OE19 and OE33 cells, treatment with 10 μM BMS-833923 resulted in 82 and 73.4% apoptotic cells, respectively.Animal experiment [2,3]:Animal models Medulloblastoma and pancreatic carcinoma xenografts mouse models, Male Sprague-Dawley rats with gastroesophageal reflux diseaseDosage form Oral administration, 10 mg/kgApplication In medulloblastoma and pancreatic carcinoma xenografts animal models, administration of BMS-833923 at single oral dose showed robust inhibition of Hh pathway. In a rat model with gastroesophageal reflux disease, administration of BMS-833923 (10 mg/kg/day) resulted in the decreased development of both Barrett esophagus and esophageal adenocarcinoma by 35.7%.Other notes Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal.References:[1]. Zaidi A H, Komatsu Y, Kelly L A, et al. Smoothened inhibition leads to decreased proliferation and induces apoptosis in esophageal adenocarcinoma cells. Cancer investigation, 2013, 31(7): 480-489. [2]. Gendreau S B, Hawkins D, Ho C P, et al. Abstract B192: Preclinical characterization of BMS‐833923 (XL139), a hedgehog (HH) pathway inhibitor in early clinical development. Molecular Cancer Therapeutics, 2009, 8(12 Supplement): B192-B192.[3].Gibson M K, Zaidi A H, Davison J M, et al. Prevention of Barrett esophagus and esophageal adenocarcinoma by smoothened inhibitor in a rat model of gastroesophageal reflux disease. Annals of surgery, 2013, 258(1): 82-88.BackgroundBMS-833923 is an orally bioavailable and selective antagonist of smoothened (SMO) with IC50 value of 5.8 nM in NIH3T3 cell line [1].The Hedgehog (Hh) signaling is a critical pathway involved in embryonic development and in tissue maintenance and repair in adults. It consists of the Hh ligands, the transmembrane receptors Patched 1 and 2, the G-protein- coupled receptor-like protein SMO and the glioma-associated oncogeneProduct Data Sheettranscription factors GLI1 to 3. The aberrant activation of Hh pathway, both mutational and epigenetic, is found to be associated with multiple aspects of tumorigenesis in various tumor cells. As a smoothened inhibitor, BMS-833923 can block the binding of cyclopamine (a naturally occurring SMO inhibitor) to SMO. It showed potent Hh pathway inhibitory activity with IC50 values at nanomolar in multiple cell-based assays. BMS-833923 also potently inhibited Hh pathway in medulloblastoma and pancreatic carcinoma xenograft models [1, 2 and 3].In vitro, BMS-833923 inhibited the expression of GLI1 and PTCH1 in cell lines expressing wild-type SMO or activated mutant SMO with IC50 values in the range from 6 to 35 nM. In the FACS-based binding assays, it does-dependently suppressed cyclopamine binding to SMO with IC50 value of 21 nM. In the esophageal adenocarcinoma cell lines OE19 and OE33, treatment of BMS-833923significantly reduced cell proliferation with IC50 values of both 10 μM. Besides that, BMS-833923 was found to inhibit the growth of multiple myeloma cells and the proportion of ALDH+ cancer stem cells. It also inhibited the growth of many other tumor cells derived from patients with hematological malignancies including ALL, AML and CML [3 and 4].In animal models with medulloblastoma and pancreatic carcinoma xenografts, administration of BMS-833923 at single oral dose showed robust inhibition of Hh pathway. In a rat model with gastroesophageal reflux disease, the administration of BMS-833923 at dose of 10 mg/kg/day resulted in the decreased development of both Barrett esophagus and esophageal adenocarcinoma by 35.7% [3 and 5].References:[1] Siu L L, Papadopoulos K P, Alberts S, et al. A first-in-human phase 1study of an oral hedgehog pathway antagonist, BMS-833923 (XL139), in subjects with advanced or metastatic solid tumors. Mol Cancer Ther, 2009, 8(12 Suppl): A55.[2] Justilien V, Fields A P. Molecular Pathways: Novel Approaches for Improved Therapeutic Targeting of Hedgehog Signaling in Cancer Stem Cells. Clinical Cancer Research, 2015, 21(3): 505-513.[3] Gendreau S B, Hawkins D, Ho C P, et al. Abstract B192: Preclinical characterization of BMS‐833923 (XL139), a hedgehog (HH) pathway inhibitor in early clinical development. Molecular Cancer Therapeutics, 2009, 8(12 Supplement): B192-B192.[4] Zaidi A H, Komatsu Y, Kelly L A, et al. Smoothened inhibition leads to decreased proliferation and induces apoptosis in esophageal adenocarcinoma cells. Cancer investigation, 2013, 31(7): 480-489.[5] Gibson M K, Zaidi A H, Davison J M, et al. Prevention of Barrett esophagus and esophageal adenocarcinoma by smoothened inhibitor in a rat model of gastroesophageal reflux disease. Annals of surgery, 2013, 258(1): 82-88.。
2‘3’-cgamp分子量

2‘3’-cgamp分子量
2'3'-cGAMP(2'3'-cyclic GMP-AMP)是一种环核苷酸分子,其分子量为约675.4克/摩尔。
这个分子是由两个核苷酸单元组成,包括鸟苷酸和腺苷酸。
在化学结构上,2'3'-cGAMP具有环状的磷酸二酯键,这使得它在免疫系统中扮演着重要的信号传递分子的角色。
这种分子在免疫调节和抗病毒防御中起着关键作用,因此在免疫疗法和药物开发中备受关注。
从化学结构和生物学功能的角度来看,2'3'-cGAMP的分子量是其重要的物理特性之一,对于研究者来说,了解其分子量有助于理解其在生物体内的作用机制和药理学特性。
希望这些信息能够满足你的需求。
9-芴甲基-n-琥珀酰亚胺基碳酸酯生产工艺

1. 介绍在当今社会,化工行业一直是不可或缺的产业之一。
而在化工行业中,有一种重要的有机合成原料——9-芴甲基-n-琥珀酰亚胺基碳酸酯,是一种广泛应用于生物医药、高性能树脂和涂料等领域的重要中间体。
而对于其生产工艺的研究和优化,将对相关行业的发展和生产效率起到重要作用。
2. 概述9-芴甲基-n-琥酰亚胺基碳酸酯9-芴甲基-n-琥酰亚胺基碳酸酯,简称FHMTC,是一种重要的有机合成原料,具有多样的应用领域。
它可以作为大量生物医药领域中的中间体,还可以用于制备高性能树脂和涂料等。
了解其生产工艺并进行优化,对于提升其生产效率和降低生产成本具有重要意义。
3. FHMTC生产工艺的传统方法目前,传统的FHMTC生产工艺一般采用从芴酸出发,经过一系列的反应步骤,最后得到目标产物FHMTC。
这种方法虽然已经比较成熟,但仍存在一些问题,例如反应过程中产生的废物较多,反应条件要求严格,对环境有一定影响等。
4. FHMTC生产工艺的新趋势随着化工技术的不断发展,针对FHMTC生产工艺,新的方法和技术也在不断涌现。
一些新型催化剂的引入、新的反应条件的确定等,都为FHMTC生产工艺的优化提供了新的思路和途径。
这些新趋势的出现,为FHMTC的生产工艺带来了新的希望和机遇。
5. FHMTC生产工艺的深入探讨针对FHMTC的生产工艺,我们可以从不同的角度进行深入探讨。
首先是原料的选择和处理,其次是反应条件的优化,最后是产物的纯化和提纯。
通过对这些环节的深入了解和研究,可以找到更加高效、环保的生产工艺方法。
6. 个人观点和总结在我看来,FHMTC生产工艺的优化是一个复杂而又具有挑战性的课题。
只有不断地进行深入研究和探索,才能找到更好的解决方案。
我相信随着化工技术的不断发展,FHMTC的生产工艺一定会得到更好的改进和优化,为相关行业的发展和生产效率带来新的活力。
7. 结语FHMTC作为重要的有机合成原料,在生产工艺的优化方面具有重要的意义。
Oracle Health Sciences InForm EDC 平台介绍说明书

Oracle Health Sciences InForm EDC PlatformDeliver Higher-Quality Data, Faster and with Less Effort, Cost and Risk INFORM AT A GLANCEDrive effciencies with integratedand automated workfows andself-service, one-click studydeployment Integration with Oracle Argus increases productivity and speeds safety reporting 50%Cut weeks or months off time to database lock with InForm Data ViewerIntegration with Oracle IRT RTSMsolution reduces data entryand data reconciliation acrossapplications by up to 90%#1 in global site satisfaction means faster study startups with less risk #1Integration with Data Management Workbench reduces time and effort to clean and transform data by up to 60%INCREASE EFFICIENCY • Design trials in just a few days and auto-deploy with one click• Assess , review , and lock a site or entire study in minutes• Streamline workfows with fully integrated CDM, IRT , medical coding, safety and moreIMPROVE DATA QUALITY• Identify problems and take corrective action more quickly with real-time visibility to data• Conduct effective risk-based monitoring• Incorporate mid-study revisions without migrating data like other EDC systemsREDUCE COST• Integrated workfows require far less time and manualeffort50% • Streamlined monitoring and faster time to database lockreduces costly and unnecessary steps• Flexible pricing and delivery models bring capabilities ofmarket-leading InForm EDC to virtually any organizationBROADEST PLATFORMInForm offers the industry’s broadest platform of integrated systems and workfows to drive greater efficiency and data quality while reducing costClinical Data ManagementPhysiologic DataIRT ePROCTMS Clinical BiomarkersMedication AdherenceInForm Site Payments SafetyeTMF Medical Coding Risk-based MonitoringORACLE HEALTH SCIENCES INFORM EDC PLATFORMThe market-leading EDC system and the #1 preferred choice of investigator sites worldwide.For more information visit our clinical data management solutionsJoin our communitiesCopyright © 2015, Oracle and/or its affiliates. All rights reserved. Oracle and Java are registered trademarks of Oracle and/or its affiliates. Other names may be trademarks of their respective owners. 150916。
