Lemborexant_LCMS_16649_MedChemExpress
果胶酶活性检测试剂盒说明书 微量法

果胶酶活性检测试剂盒说明书微量法注意:正式测定之前选择2-3个预期差异大的样本做预测定。
货号:BC2635规格:100T/48S产品内容:提取液:液体100mL×1瓶,4℃保存。
试剂一:液体25mL×1瓶,4℃保存。
若溶液中有不溶解物质,可以50℃水浴溶解。
试剂二:液体20mL×1瓶,4℃避光保存。
标准品:粉剂×1支,10mg半乳糖醛酸。
临用前加入0.943mL蒸馏水,配成50μmol/mL的标准液产品说明:果胶酶(pectinase)是分解果胶的酶类,包括原果胶酶,果胶酯酶,多聚半乳糖醛酸酶和果胶裂解酶四大类,广泛存在于高等植物果实和微生物中,是水果加工中最重要的酶。
果胶酶水解果胶生成半乳糖醛酸,半乳糖醛酸与DNS试剂反应生成在540nm有特征吸收峰的棕红色物质,测定540nm处吸光值变化可计算得果胶酶活性。
试验中所需的仪器和试剂:可见分光光度计/酶标仪、台式离心机、水浴锅、微量玻璃比色皿/96孔板、可调式移液枪、研钵/匀浆器、冰和蒸馏水。
操作步骤:一、粗酶液提取:组织:按照组织质量(g):提取液体积(mL)为1:5~10的比例(建议称取约0.1g组织,加入1mL提取液)进行冰浴匀浆,然后10000g,4℃,离心10min,取上清置于冰上待测。
菌类:按照细胞数量(104个):提取液体积(mL)为500~1000:1的比例(建议500万细胞加入1mL 提取液),冰浴超声波破碎细胞(功率300w,超声3秒,间隔7秒,总时间3min);然后10000g,4℃,离心10min,取上清置于冰上待测。
液体:直接检测。
二、测定步骤:1、分光光度计/酶标仪预热30min以上,调节波长至540nm,蒸馏水调零。
2、将50μmol/mL标准液用蒸馏水稀释为10、8、6、4、2、1μmol/mL的标准溶液备用。
3、取40μL样本沸水浴10min备用。
4、操作表:(在1.5mL离心管中)对照管测定管标准管空白管试剂一(µL)20020020020050℃水浴温育5min标准溶液(µL)--40-样本(µL)-40--蒸馏水(µL)---40煮沸样本(µL)40---混匀,50℃水浴反应30min,马上沸水浴5min,冷却后8000g,常温离心10min,取上清。
Gelucire-14-44-SDS-MedChemExpress

Inhibitors, Agonists, Screening LibrariesSafety Data Sheet Revision Date:Nov.-23-2018Print Date:Nov.-23-20181. PRODUCT AND COMPANY IDENTIFICATION1.1 Product identifierProduct name :Gelucire 14/44Catalog No. :HY-Y1892CAS No. :121548-04-71.2 Relevant identified uses of the substance or mixture and uses advised againstIdentified uses :Laboratory chemicals, manufacture of substances.1.3 Details of the supplier of the safety data sheetCompany:MedChemExpress USATel:609-228-6898Fax:609-228-5909E-mail:sales@1.4 Emergency telephone numberEmergency Phone #:609-228-68982. HAZARDS IDENTIFICATION2.1 Classification of the substance or mixtureNot a hazardous substance or mixture.2.2 GHS Label elements, including precautionary statementsNot a hazardous substance or mixture.2.3 Other hazardsNone.3. COMPOSITION/INFORMATION ON INGREDIENTS3.1 SubstancesSynonyms:NoneFormula:N/AMolecular Weight:N/ACAS No. :121548-04-74. FIRST AID MEASURES4.1 Description of first aid measuresEye contactRemove any contact lenses, locate eye-wash station, and flush eyes immediately with large amounts of water. Separate eyelids with fingers to ensure adequate flushing. Promptly call a physician.Skin contactRinse skin thoroughly with large amounts of water. Remove contaminated clothing and shoes and call a physician.InhalationImmediately relocate self or casualty to fresh air. If breathing is difficult, give cardiopulmonary resuscitation (CPR). Avoid mouth-to-mouth resuscitation.IngestionWash out mouth with water; Do NOT induce vomiting; call a physician.4.2 Most important symptoms and effects, both acute and delayedThe most important known symptoms and effects are described in the labelling (see section 2.2).4.3 Indication of any immediate medical attention and special treatment neededTreat symptomatically.5. FIRE FIGHTING MEASURES5.1 Extinguishing mediaSuitable extinguishing mediaUse water spray, dry chemical, foam, and carbon dioxide fire extinguisher.5.2 Special hazards arising from the substance or mixtureDuring combustion, may emit irritant fumes.5.3 Advice for firefightersWear self-contained breathing apparatus and protective clothing.6. ACCIDENTAL RELEASE MEASURES6.1 Personal precautions, protective equipment and emergency proceduresUse full personal protective equipment. Avoid breathing vapors, mist, dust or gas. Ensure adequate ventilation. Evacuate personnel to safe areas.Refer to protective measures listed in sections 8.6.2 Environmental precautionsTry to prevent further leakage or spillage. Keep the product away from drains or water courses.6.3 Methods and materials for containment and cleaning upAbsorb solutions with finely-powdered liquid-binding material (diatomite, universal binders); Decontaminate surfaces and equipment by scrubbing with alcohol; Dispose of contaminated material according to Section 13.7. HANDLING AND STORAGE7.1 Precautions for safe handlingAvoid inhalation, contact with eyes and skin. Avoid dust and aerosol formation. Use only in areas with appropriate exhaust ventilation.7.2 Conditions for safe storage, including any incompatibilitiesKeep container tightly sealed in cool, well-ventilated area. Keep away from direct sunlight and sources of ignition.Recommended storage temperature:Pure form-20°C 3 years4°C 2 yearsIn solvent-80°C 6 months-20°C 1 monthShipping at room temperature if less than 2 weeks.7.3 Specific end use(s)No data available.8. EXPOSURE CONTROLS/PERSONAL PROTECTION8.1 Control parametersComponents with workplace control parametersThis product contains no substances with occupational exposure limit values.8.2 Exposure controlsEngineering controlsEnsure adequate ventilation. Provide accessible safety shower and eye wash station.Personal protective equipmentEye protection Safety goggles with side-shields.Hand protection Protective gloves.Skin and body protection Impervious clothing.Respiratory protection Suitable respirator.Environmental exposure controls Keep the product away from drains, water courses or the soil. Cleanspillages in a safe way as soon as possible.9. PHYSICAL AND CHEMICAL PROPERTIES9.1 Information on basic physical and chemical propertiesAppearance White to off-white (Oil)Odor No data availableOdor threshold No data availablepH No data availableMelting/freezing point No data availableBoiling point/range No data availableFlash point No data availableEvaporation rate No data availableFlammability (solid, gas)No data availableUpper/lower flammability or explosive limits No data availableVapor pressure No data availableVapor density No data availableRelative density No data availableWater Solubility No data availablePartition coefficient No data availableAuto-ignition temperature No data availableDecomposition temperature No data availableViscosity No data availableExplosive properties No data availableOxidizing properties No data available9.2 Other safety informationNo data available.10. STABILITY AND REACTIVITY10.1 ReactivityNo data available.10.2 Chemical stabilityStable under recommended storage conditions.10.3 Possibility of hazardous reactionsNo data available.10.4 Conditions to avoidNo data available.10.5 Incompatible materialsStrong acids/alkalis, strong oxidising/reducing agents.10.6 Hazardous decomposition productsUnder fire conditions, may decompose and emit toxic fumes.Other decomposition products - no data available.11.TOXICOLOGICAL INFORMATION11.1 Information on toxicological effectsAcute toxicityClassified based on available data. For more details, see section 2Skin corrosion/irritationClassified based on available data. For more details, see section 2Serious eye damage/irritationClassified based on available data. For more details, see section 2Respiratory or skin sensitizationClassified based on available data. For more details, see section 2Germ cell mutagenicityClassified based on available data. For more details, see section 2CarcinogenicityIARC: No component of this product present at a level equal to or greater than 0.1% is identified as probable, possible or confirmed human carcinogen by IARC.ACGIH: No component of this product present at a level equal to or greater than 0.1% is identified as a potential or confirmed carcinogen by ACGIH.NTP: No component of this product present at a level equal to or greater than 0.1% is identified as a anticipated or confirmed carcinogen by NTP.OSHA: No component of this product present at a level equal to or greater than 0.1% is identified as a potential or confirmed carcinogen by OSHA.Reproductive toxicityClassified based on available data. For more details, see section 2Specific target organ toxicity - single exposureClassified based on available data. For more details, see section 2Specific target organ toxicity - repeated exposureClassified based on available data. For more details, see section 2Aspiration hazardClassified based on available data. For more details, see section 212. ECOLOGICAL INFORMATION12.1 ToxicityNo data available.12.2 Persistence and degradabilityNo data available.12.3 Bioaccumlative potentialNo data available.12.4 Mobility in soilNo data available.12.5 Results of PBT and vPvB assessmentPBT/vPvB assessment unavailable as chemical safety assessment not required or not conducted.12.6 Other adverse effectsNo data available.13. DISPOSAL CONSIDERATIONS13.1 Waste treatment methodsProductDispose substance in accordance with prevailing country, federal, state and local regulations.Contaminated packagingConduct recycling or disposal in accordance with prevailing country, federal, state and local regulations.14. TRANSPORT INFORMATIONDOT (US)This substance is considered to be non-hazardous for transport.IMDGThis substance is considered to be non-hazardous for transport.IATAThis substance is considered to be non-hazardous for transport.15. REGULATORY INFORMATIONSARA 302 Components:No chemicals in this material are subject to the reporting requirements of SARA Title III, Section 302.SARA 313 Components:This material does not contain any chemical components with known CAS numbers that exceed the threshold (De Minimis) reporting levels established by SARA Title III, Section 313.SARA 311/312 Hazards:No SARA Hazards.Massachusetts Right To Know Components:No components are subject to the Massachusetts Right to Know Act.Pennsylvania Right To Know Components:No components are subject to the Pennsylvania Right to Know Act.New Jersey Right To Know Components:No components are subject to the New Jersey Right to Know Act.California Prop. 65 Components:This product does not contain any chemicals known to State of California to cause cancer, birth defects, or anyother reproductive harm.16. OTHER INFORMATIONCopyright 2018 MedChemExpress. The above information is correct to the best of our present knowledge but does not purport to be all inclusive and should be used only as a guide. The product is for research use only and for experienced personnel. It must only be handled by suitably qualified experienced scientists in appropriately equipped and authorized facilities. The burden of safe use of this material rests entirely with the user. MedChemExpress disclaims all liability for any damage resulting from handling or from contact with this product.Caution: Product has not been fully validated for medical applications. For research use only.Tel: 609-228-6898 Fax: 609-228-5909 E-mail: tech@Address: 1 Deer Park Dr, Suite Q, Monmouth Junction, NJ 08852, USA。
上市新药Lemborexant(莱博雷生)合成检索总结报告

合成方法二
NaHMDS (85 mL, 2.0 M) was added dropwise to a solution of 2-phenylacetonitrile1(7.96g) in THF (300 mL) under cooling in an ice-salt bath. The mixture was stirred for 2 h, then R-(−)-epichlorohydrin2(7.548 g) was added dropwise (3 h, 0 °C). Stirring was continued for 2h at 0°C then overnight at room temperature. The reaction solution was then cooled on ice, and a small amount of water was added. The resulting mixture was concentrated under reduced pressure, then ethanol (100 mL) and 1.5 N/KOH aqueous solution (100 mL) were added to the residue. The obtained mixture was heated to reflflux for 8h. The reaction solution was allowed to cool to room temperature, and concentrated hydrochloric acid was added to adjust the pH to <2. This mixture was stirred at 0°C for 2h then was concentrated under reduced pressure. Ethyl acetate and water were added to the concentrated solution to carry out liquid separation. The organic layer was washed successively with a saturated sodium bicarbonate aqueous solution and a saturated sodium chloride aqueous solution. The organic layer was dried over magnesium sulfate, the drying agent was removed by filtration, and the filtrate was concentrated under reduced pressure. . The residue was purifified by silica gel column chromatography, giving the title compound3(9.96 g, 49.8% yield) as a white solid
羟自由基清除能力检测试剂盒说明书 微量法

北京索莱宝科技有限公司羟自由基清除能力检测试剂盒说明书微量法注意:正式测定前务必取2-3个预期差异较大的样本做预测定。
货号:BC1325规格:100T/96S 产品内容:提取液:液体110mL×1瓶,4℃保存。
试剂一:液体8mL×1瓶,4℃避光保存。
试剂二:液体15mL×1瓶,4℃保存。
试剂三:液体15mL×1瓶,4℃保存。
试剂四:液体0.16mL×1瓶,4℃避光保存。
临用前加入9.84mL 蒸馏水混匀,也可按比例现用现配,配好的试剂4℃保存一周。
产品说明:羟自由基是人体在新陈代谢过程中产生的对生物体毒性强、危害大的一种自由基。
它可以使组织中的糖类、氨基酸、蛋白质、核酸等物质发生氧化,遭受氧化性损伤和破坏,导致细胞坏死或突变。
羟自由基清除能力是样品抗氧化能力的重要指标之一,在抗氧化类保健品和药品研究中得到广泛应用。
H 2O 2/Fe 2+通过Fenton 反应产生羟自由基,将邻二氮菲-Fe 2+水溶液中Fe 2+氧化为Fe 3+,导致536nm 的吸光度下降,样品536nm 吸光度下降速率的抑制程度,反映了样品清除羟自由基的能力。
自备实验用品:恒温水浴锅、可见分光光度计/酶标仪、微量玻璃比色皿/96孔板、低温离心机、研钵/匀浆器和蒸馏水。
操作步骤:一、样品的制备:(1)组织样品的制备:称取约0.1g 组织,加入1mL 提取液进行冰浴匀浆;10000g 4℃离心10min,取上清,置冰上待测。
(2)血清、果汁等液体样品可直接测定。
(3)提取物(或者药物)可配制成一定浓度,如5mg/mL。
二、测定步骤:1、分光光度计或酶标仪预热30min以上,调节波长至536nm,蒸馏水调零。
2、操作表:在1.5或0.5mLEP管中分别加入下列试剂空白管对照管测定管试剂一(µL)505050试剂二(µL)100100100试剂三(µL)100100100充分混匀,防止颜色不均一。
BP2015英国药典索引
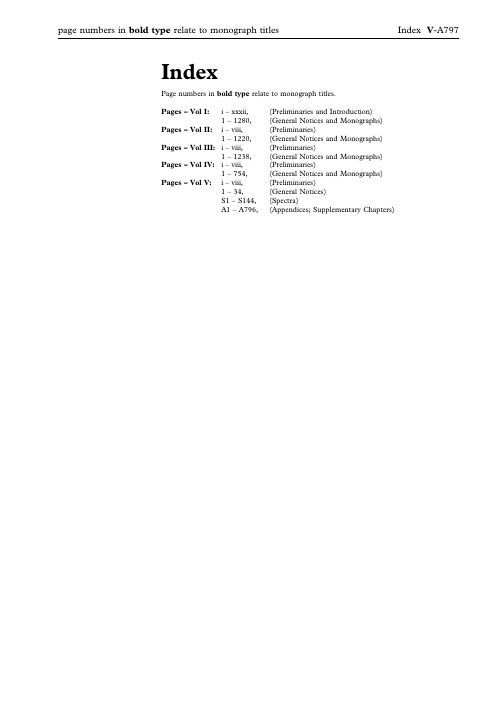
page numbers in bold type relate to monograph titles Index V-A797IndexPage numbers in bold type relate to monograph titles.Pages–Vol I:i–xxxii,(Preliminaries and Introduction)1–1280,(General Notices and Monographs)Pages–Vol II:i–viii,(Preliminaries)1–1220,(General Notices and Monographs)Pages–Vol III:i–viii,(Preliminaries)1–1238,(General Notices and Monographs)Pages–Vol IV:i–viii,(Preliminaries)1–754,(General Notices and Monographs)Pages–Vol V:i–viii,(Preliminaries)1–34,(General Notices)S1–S144,(Spectra)A1–A796,(Appendices;Supplementary Chapters)AAbacavir,V-S4Abacavir Oral Solution,III-85 Abacavir Sulfate,I-39Abacavir Tablets,III-86 Abbreviated,V-598Adjectives,V-598Anions,V-598Cations,V-598Preparations,V-598Titles of Monographs,V-598 Abbreviated Titles,Status of,I-7,II-7, III-7,IV-7,V-7Abbreviations and symbols,I-30,II-30, III-30,IV-30,V-30Abnormal Toxicity,Test for,V-409 About,definition of,I-5,II-5,III-5,IV-5,V-5Absence of Mycoplasmas,Test forV-487Absolute Ethanol,V-A61Absolute Ethanol R1,V-A62 Absorbent Cotton,IV-743Absorbent Viscose Wadding,IV-744 Absorption spectrophotometry,infrared, V-162Absorption Spectrophotometry, Ultraviolet and Visible,V-169 Acacia,I-41,V-A19Acacia Solution,V-A19Acacia Spray-dried,I-42 Acamprosate Calcium,I-43 Acanthopanax Bark,IV-49 Acarbose,I-44Accuracy,V-674Acebutolol Capsules,III-87 Acebutolol Hydrochloride,I-46,V-S5, V-A19Acebutolol Tablets,III-88 Aceclofenac,I-48Acemetacin,I-50Acenocoumarol,I-52,V-S5 Acenocoumarol Tablets,III-88 Acesulfame Potassium,I-52Acetal,V-A19Acetaldehyde,V-A19Acetaldehyde Ammonia Trimer Trihydrate,V-A20Acetaldehyde Standard Solution(100ppm C2H4O),V-A148 Acetaldehyde Standard Solution(100ppm C2H4O)R1,V-A148 Acetamide,V-A20Acetate Buffer pH2.8,V-A152 Acetate Buffer pH2.45,V-A152 Acetate Buffer pH3.4,V-A152 Acetate Buffer pH3.5,V-A152 Acetate Buffer pH3.7,V-A152 Acetate Buffer pH4.4,V-A152 Acetate Buffer pH4.6,V-A152 Acetate Buffer pH5.0,V-A152 Acetate Buffer pH6.0,V-A152 Acetate Buffer Solution pH4.7R1,V-A153Acetate Buffer Solution pH4.4,see Acetate Buffer pH4.4Acetate Buffer Solution pH4.6,see Acetate Buffer pH4.6Acetate Buffer Solution pH6.0,seeAcetate Buffer pH6.0Acetate Buffer Solution pH4.4,V-A152Acetate Buffer Solution pH4.5,V-A152Acetate Buffer Solution pH4.7,V-A152Acetate Buffer Solution pH5.0,V-A153Acetate Buffer Solution pH6.0,V-A153Acetate–edetate Buffer Solution pH5.5,V-A153Acetates,Reactions of,V-266Acetazolamide,I-54,V-S5Acetazolamide Oral Suspension,III-89Acetazolamide Tablets,III-90Acetic Acid,V-A20Acetic Acid(6per cent),I-56Acetic Acid(33per cent),I-56Acetic Acid,Anhydrous,V-A20Acetic Acid,Deuterated,V-A50Acetic Acid,Dilute,V-A20Acetic Acid,Dilute,see Acetic Acid(6per cent)Acetic Acid,Glacial,I-55,V-A20Acetic Acid in Synthetic Peptides,Determination of,V-299Acetic Acid VS,V-A142Acetic Acid,see Acetic Acid(33per cent)Acetic Anhydride,V-A20Acetic Anhydride Solution R1,V-A20Acetic Anhydride–Dioxan Solution,V-A20Acetic Anhydride–Sulfuric Acid Solution,V-A20Acetic Anhydride–Sulphuric AcidSolution,see Acetic Anhydride–SulfuricAcid SolutionAcetic Bromine Solution,V-A34Acetone,I-57,V-A20Acetone,Deuterated,V-A50Acetone Solution,Buffered,V-A153Acetone-dried Ox Brain,V-A98Acetonitrile,V-A20Acetonitrile for Chromatography,V-A20Acetonitrile R1,V-A20Acetoxyvalerenic Acid,V-A20Acetyl Chloride,V-A20Acetyl Groups,Reactions of,V-266Acetyl Salicylic Acid see AspirinAcetyl Value,Determination of,V-317Acetylacetamide,V-A20Acetylacetone,V-A20Acetylacetone Reagent R1,V-A20Acetylacetone Reagent R2,V-A204-Acetylbiphenyl,V-A20O-Acetyl Groups in PolysaccharideVaccines,V-467N-Acetyl-e-caprolactam,V-A20Acetylcholine Chloride,I-58,V-A20Acetylcysteine,I-59,V-S6Acetylcysteine Eye Drops,III-90Acetylcysteine Injection,III-91Acetyldigoxin,I-61b-Acetyldigoxin see AcetyldigoxinAcetyleugenol,V-A20N-Acetylglucosamine,V-A21Acetyl-11-keto-b-boswellic Acid,V-A21N-Acetyl-L-cysteine,V-A20N-Acetylneuraminic Acid,V-A21Acetylsalicylic Acid Tablets,see AspirinTabletsN-Acetyltryptophan,V-A21N-Acetyltryptophan see AcetyltryptophanAcetyltryptophan,I-63Acetyltyrosine,I-65N-Acetyltyrosine see AcetyltyrosineAcetyltyrosine Ethyl Ester,V-A21Acetyltyrosine Ethyl Ester,0.2M,V-A21Aciclovir,I-67Aciclovir Cream,III-93Aciclovir Eye Ointment,III-94Aciclovir Infusion,III-95Aciclovir Intravenous Infusion,seeAciclovir Infusion,Aciclovir Oral Suspension,III-97Aciclovir Sodium for Infusion,III-95Aciclovir Sodium for IntravenousInfusion,see Aciclovir Sodium forInfusion,Aciclovir Tablets,III-98Aciclovir Tablets,Dispersible,III-99Acid Blue92,V-A21Acid Blue92Solution,V-A21Acid Blue83,V-A21Acid Blue93Solution,V-A21Acid Blue90,V-A21Acid Gentian Mixture,IV-197Acid Gentian Oral Solution,IV-197Acid Potassium IodobismuthateSolution,V-A108Acid Value,V-317Acid/base Indicators,V-789Acid-base titrations,V-788Acidified Chloroform,V-A41Acidified Dichloromethane,V-A52Acidified Methanol,V-A85Acidified Methylene Chloride,seeAcidified DichloromethaneAcid-insoluble Ash,Determination of,V-336Acid-washed Diatomaceous Support,V-A51Acitretin,I-69Acitretin Capsules,III-100Acknowledgements,I-xxviiAcrylamide,V-A21Acrylamide/bisacrylamide(29:1)Solution,30per cent,V-A21Acrylamide/bisacrylamide(36.5:1)Solution,30per cent,V-A21Acrylic Acid,V-A21Actein,V-A21Acteoside,V-A21Action and Use Statement,Status of,I-17,II-17,III-17,IV-17,V-17Activated Acid Aluminium Oxide,V-A23Activated Attapulgite,I-220Activated Charcoal,I-496,V-A40Activated Zinc,V-A140Active Moiety,V-651Adamantane,V-A21Adapalene,I-71Adapalene Cream,III-101Adapalene Gel,III-103Additions,List of,I-xxviiiAdditions,List of Monographs,I-xxiiAdditives,Plastic,V-592Adenine,I-72,V-A21Adenosine,I-73,V-A21Adipic Acid,I-75,V-A21Adrenaline,V-A21Adrenaline/Epinephrine,I-76page numbers in bold type relate to monograph titles Index V-A799Adrenaline Acid Tartrate,V-A21 Adrenaline Acid Tartrate/Epinephrine Acid Tartrate,I-77Adrenaline and Cocaine Intranasal Solution,III-107Adrenaline(Epinephrine),V-S6 Adrenaline Eye Drops,Epinephrine Eye Drops,Neutral,III-104Adrenaline Eye Drops/Epinephrine Eye Drops,III-104Adrenaline Injection,Bupivacaine and, III-220Adrenaline Injection,Dilute(1in10,000),III-106Adrenaline Injection,Lidocaine and,III-751Adrenaline Injection/Epinephrine Injection,III-105Adrenaline Solution/Epinephrine Solution,III-106Adrenaline Tartrate see Adrenaline Acid TartrateAdrenaline TartrateInjection/Epinephrine Tartrate Injection,III-105Adrenaline TartrateSolution/Epinephrine Tartrate Solution,III-106Adrenalone Hydrochloride,V-A21 Adsorbed Diphtheria and Tetanus Vaccine,IV-537Adsorbed Diphtheria and Tetanus Vaccine for Adults and Adolescents, see Adsorbed Diphtheria and Tetanus Vaccine(adsorbed,Reduced Antigen(s) Content)Adsorbed Diphtheria,Tetanus and Pertussis(Acellular Component) Vaccine,IV-541Adsorbed Diphtheria,Tetanus,Pertussis (Acellular Component)and Haemophilus Type b Conjugate Vaccine,IV-545Adsorbed Diphtheria,Tetanus,Pertussis (Acellular Component)and Hepatitis B(rDNA)Vaccine,IV-547 Adsorbed Diphtheria,Tetanus,Pertussis (Acellular Component)and Inactivated Poliomyelitis Vaccine,IV-548Adsorbed Diphtheria Vaccine,IV-534 Adsorbed Diphtheria Vaccine for Adults and Adolescents,see Diphtheria Vaccine (Adsorbed,Reduced Antigen Content) Adsorbed Pertussis Vaccine(Acellular Component),IV-604Adsorbed Pertussis Vaccine(Acellular, Co-purified),IV-605Adsorbed Tetanus Vaccine,IV-633 Adsorbed Vaccines,Aluminium in,V-463Adsorbed Vaccines,Calcium in,V-464 Adsorption,Gas,Specific Surface Area By(2.9.26.)(5.8.),V-701Aescin,V-A22Aflatoxin B1,V-A22Aflatoxin B,in Herbal Drugs, Determination of,V-341Agar,I-79,V-A22Agarose for Chromatography,V-A22Agarose for Chromatography,Cross-linked,V-A22Agarose for Chromatography R1,Cross-linked,V-A22Agarose for Electrophoresis,V-A22Agarose/Cross-linked Polyacrylamide,V-A22Agarose-DEAE for Ion ExchangeChromatography,V-A22Agnus Castus Fruit,IV-50Agrimony,IV-52Air,Medical,I-78Air,Medicinal see Medical AirAir Permeability,Specific Surface Areaby,V-505Air,Synthetic,I-81Air,Synthetic Medicinal see Synthetic AirAlanine,I-83,V-A22ß-Alanine,see3-Aminopropionic AcidAlbendazole,I-84Albumin,Bovine,V-A22Albumin,Bovine R1,V-A22Albumin,Human,V-A22Albumin Solution,IV-467Albumin Solution,Human,V-A22Albumin Solution R1,Human,V-A22Alchemilla,IV-53Alcohol(20per cent),I-900Alcohol(25per cent),I-900Alcohol(45per cent),I-900Alcohol(50per cent),I-900Alcohol(60per cent),I-900Alcohol(70per cent),I-900Alcohol(80per cent),I-900Alcohol(90per cent),I-900Alcohol,Aldehyde-free,see Ethanol(96%),Aldehyde-freeAlcoholic Calcium Standard Solution(100ppm Ca),V-A149Alcoholic DimethylaminobenzaldehydeSolution,V-A55Alcoholic Hydroxylamine Solution,V-A74Alcoholic Iodine Solution,III-696,V-A75Alcoholic Potassium Hydroxide,2M,V-A108Alcoholic Potassium Hydroxide,seePotassium Hydroxide VS,EthanolicAlcoholic Potassium Hydroxide Solution,V-A108Alcoholic Potassium Hydroxide SolutionR1,V-A108Alcoholic Solution of Sulfuric Acid,V-A128Alcoholic Sulfuric Acid,0.25M,V-A128Alcoholimetric Tables,V-687Alcohol,see Ethanol(96%)Alcuronium Chloride,I-85Aldehyde Dehydrogenase,V-A22Aldehyde Dehydrogenase Solution,V-A22Aldehyde-free alcohol,see Ethanol(96%),Aldehyde-freeAldehyde-free Ethanol(96%),V-A62Aldehyde-free Methanol,V-A85Aldehydes,Determination of,V-321Aldrin,V-A22Alendronate Sodium Tablets,seeAlendronic Acid TabletsAlendronic Acid Tablets,III-109Aleuritic Acid,V-A22Alexandrian Senna Fruit,IV-362Alfacalcidol,I-87Alfadex,I-88Alfentanil Hydrochloride,I-89Alfuzosin,V-S7Alfuzosin Hydrochloride,I-91Alfuzosin Tablets,III-111Alfuzosin Tablets,Prolonged-release,III-112Alginate Antacid Oral Suspension,Compound,III-113Alginate Oral Suspension,Raft-forming,III-114Alginate Raft-forming Oral Suspension,III-114Alginic Acid,I-92Alimemazine,V-S7Alimemazine Oral Solution,Paediatric,III-115Alimemazine Tablets,III-116Alimemazine Tartrate,I-93Alizarin S,V-A22Alizarin S Solution,V-A22Alkaline Corallin Solution,V-A46Alkaline Eye Drops,see Hypromellose EyeDropsAlkaline Gentian Mixture,IV-198Alkaline Gentian Oral Solution,IV-198Alkaline Hydroxylamine Solution,V-A74Alkaline Hydroxylamine Solution R1,V-A74Alkaline Potassium Mercuri-iodideSolution,V-A109Alkaline Potassium TetraiodomercurateSolution,V-A109Alkaline Pyrogallol Solution,V-A111Alkaline Sodium Picrate Solution,V-A124Alkaline Tetrazolium Blue Solution,V-A132Alkali-washed Diatomaceous Support,V-A51Alkaloids,Complete Extraction of,V-335Alkaloids,Reactions of,V-266all--Alpha-Tocopherol,II-1051Allantoin,I-94,V-A23Allergen Products,I-95Allium Sativum for HomoeopathicPreparations,IV-427Allopurinol,I-98Allopurinol Oral Suspension,III-117Allopurinol Tablets,III-118all-rac-Alpha-Tocopheryl Acetate,II-1054all-rac-a-Tocopheryl see all-rac-Alpha-Tocopherylall-rac-Tocopheryl Acetate see all-rac-Alpha-Tocopheryl AcetateAlmagate,I-100Almond Oil Ear Drops,III-119Almond Oil,Refined,I-100Almond Oil,Virgin,I-99Almond Oil see Virgin Almond OilAloes,Barbados,IV-53Aloes,Cape,IV-54Alovudine,V-A23Alovudine(18F)Injection,IV-669V-A800IndexAloxiprin,I-102Aloxiprin Tablets,III-119Alpha Tocopheryl Acetate Concentrate (Powder Form),II-1057Alpha Tocopheryl Hydrogen Succinate, II-1058Alpha Tocopheryl Succinate Tablets,III-1164Alphacyclodextrin see Alfadex Alprazolam,I-103Alprenolol Hydrochloride,I-105 Alprostadil,I-107Alteplase for Injection,I-109 Alternative methods,I-20,II-20,III-20, IV-20,V-20Alternative Methods for Control of Microbiological Quality,V-745 Altizide,I-113Alum,I-114Aluminium,V-A23Aluminium Acetate Ear Drops,III-120 Aluminium Chloride,V-A23 Aluminium Chloride Hexahydrate,I-114 Aluminium Chloride Reagent,V-A23 Aluminium Chloride Solution,III-121, V-A23Aluminium Glycinate,I-115 Aluminium Hydroxide and Magnesium Trisilicate Tablets,Chewable,III-776 Aluminium Hydroxide,Dried,I-115 Aluminium Hydroxide Gel,V-A23 Aluminium Hydroxide,Hydrated for Adsorption,I-114Aluminium Hydroxide Oral Suspension, III-121Aluminium Hydroxide Oral Suspension, Magnesium Hydroxide and,III-401 Aluminium Hydroxide Tablets, Chewable,III-122Aluminium Hydroxide Tablets, Magnesium Hydroxide and,III-402 Aluminium Hydroxide Tablets see Chewable Aluminium Hydroxide Tablets,III-122Aluminium in Adsorbed Vaccines,V-463 Aluminium Magnesium Silicate,I-118 Aluminium Nitrate,V-A23Aluminium Oxide,Activated Acid,V-A23Aluminium Oxide,Anhydrous,V-A23 Aluminium Oxide,Basic,V-A23 Aluminium Oxide,Deactivated,V-A23 Aluminium Oxide G,V-A23 Aluminium Oxide,Neutral,V-A23 Aluminium Paste,Compound,III-120 Aluminium Phosphate Gel,I-120 Aluminium Phosphate,Hydrated see Dried Aluminium Phosphate Aluminium Potassium Sulfate,V-A23 Aluminium Potassium Sulphate,see Aluminium Potassium Sulfate Aluminium Powder,I-121Aluminium Salts,Reactions of,V-266 Aluminium Sodium Silicate,I-122 Aluminium Standard Solution(2ppm Al),V-A148Aluminium Standard Solution(10ppm Al),V-A148Aluminium Standard Solution(100ppm Al),V-A148Aluminium Standard Solution(200ppmAl),V-A148Aluminium Stearate,I-123Aluminium Sulfate,I-125,V-A23Aluminium Sulphate,see AluminiumSulfateAlverine Capsules,III-122Alverine Citrate,I-126,V-S7Amantadine Capsules,III-123Amantadine Hydrochloride,I-127Amantadine Oral Solution,III-124Amantidine,V-S8Amaranth S,V-A23Amaranth Solution,V-A23Ambroxol Hydrochloride,I-128Americium-243Spiking Solution,V-A23Amethocaine Eye Drops,see TetracaineEye DropsAmfetamine Sulfate,I-130Amfetamine Sulphote,see AmfetamineSulfate,I-130Amido Black10B Solution,V-A23Amidohexadecylsilyl Silica Gel forchromatography,V-A115Amidotrizoic Acid Dihydrate,I-130Amikacin,I-132Amikacin Injection,III-124Amikacin Sulfate,I-135Amiloride and Furosemide Tablets,seeCo-amilofruse TabletsAmiloride and Hydrochlorothiazide OralSolution,see Co-amilozide Oral SolutionAmiloride and HydrochlorothiazideTablets,see Co-amilozide TabletsAmiloride Hydrochloride,I-138Amiloride Tablets,III-125Amines,Primary Aromatic,Reactions of,V-266Amino Acid Analysis,V-221Amino Acid Analysis(2.2.56.)(5.8.),V-700Amino Acids,Use of Codes for,I-8,II-8,III-8,IV-8,V-8Aminoazobenzene,V-A23Aminobenzoic Acid,I-139,V-A23,V-A244-Aminobenzoic Acid Solution,V-A24(4-Aminobenzoyl)-L-glutamic Acid,V-A244-Aminobutanoic acid,see4-Amino-n-butyric Acid2-Aminobutan-1-ol,V-A24Aminocaproic Acid,I-1402-Amino-5-chlorobenzophenone,V-A24Aminochlorobenzophenone,see2-Amino-5-chlorbenzophenone4-Aminofolic Acid,V-A24Aminoglutethimide,I-141,V-S8Aminoglutethimide Tablets,III-126Aminohexadecylsilyl Silica Gel forChromatography,V-A1156-Aminohexanoic Acid,V-A24p-Aminohippuric Acid,V-A24Aminohippuric Acid Reagent,V-A244-Amino-3-hydroxynaphthalene-1-sulfonic Acid,V-A24Aminohydroxynaphthalenesulfonic AcidSolution,V-A24Aminohydroxynaphthalenesulfonic AcidSolution,Strong,V-A24AminohydroxynaphthalenesulphonicAcid Solution,Strong,seeAminohydroxynaphthalenesulfonic AcidSolution,StrongAminohydroxynaphthalenesulphonicAcid Solution,seeAminohydroxynaphthalenesulfonic AcidSolution4-Amino-3-hydroxynaphthalene-1-sulphonic Acid,see4-Amino-3-hydroxynaphthalene-1-sulfonic AcidAminohydroxynaphthalenesulphonic,Acid,see Aminonaphthalenesulfonic AcidSolution5-Aminoimidazole-4-carboxamideHydrochloride,V-A24cis-Aminoindanol,V-A24Aminomethylalizarindiacetic AcidReagent,V-A24Aminomethylalizarindiacetic AcidSolution,V-A253-Aminomethylalizarin-N,N-diaceticAcid,V-A244-Aminomethylbenzoic acid,V-A253-(Aminomethyl)pyridine,V-A258-Aminonaphthalene-2-sulfonic Acid,V-A25Aminonaphthalenesulfonic AcidSolution,V-A25Aminonaphthalenesulphonic AcidSolution,see AminonaphthalenesulfonicAcid Solution8-Aminonaphthalene-2-sulphonic Acid,see8-Aminonaphthalene-2-sulfonic Acid4-Amino-n-butyric Acid,V-A242-Amino-5-nitrobenzophenone,V-A25Aminonitrobenzophenone,see2-Amino-5-nitrobenzophenone4-Aminophenazone,V-A25Aminophenazone Solution,V-A253-Aminophenol,V-A254-Aminophenol-free Paracetamol,V-A99Aminophylline,I-143Aminophylline Hydrate,I-145Aminophylline Injection,III-128Aminophylline Tablets,III-128Aminophylline Tablets,Prolonged-release,III-129Aminopolyether,V-A253-Aminopropanol,V-A253-Aminopropionic Acid,V-A25Aminopropylmethylsilyl Silica Gel forChromatography,V-A115Aminopropylsilyl Silica Gel forChromatography,V-A115Aminopropylsilyl Silica Gel forChromatography R1,V-A115Aminopyrazolone,see4-AminophenazoneAminopyrazolone Solution,seeAminophenazone Solution3-Aminosalicylic Acid,V-A25Amiodarone,V-S8Amiodarone Concentrate,Sterile,III-130Amiodarone Hydrochloride,I-147Amiodarone Infusion,III-129Amiodarone Intravenous Infusion,seeAmiodarone Infusion,Amiodarone Oral Suspension,III-131Amiodarone Sterile Concentrate,III-130page numbers in bold type relate to monograph titles Index V-A801Amiodarone Tablets,III-132 Amisulpride,I-149,V-S9Amisulpride Oral Solution,III-133 Amisulpride Tablets,III-134 Amitriptyline Embonate,I-150 Amitriptyline Hydrochloride,I-151 Amitriptyline Tablets,III-135 Amlodipine Besilate,I-153 Ammonia,V-A25Ammonia(13N)Injection,IV-672 Ammonia Buffer pH9.5,see Ammonium Chloride Buffer Solution pH9.5 Ammonia Buffer pH10.9,V-A153 Ammonia Buffer pH10.9,Dilute,V-A153Ammonia Buffer pH10.0,V-A153 Ammonia,Chloride-free,V-A25 Ammonia,Concentrated,V-A25 Ammonia,Lead-free,V-A25 Ammonia,Methanolic,V-A25 Ammonia R1,Concentrated,V-A26 Ammonia R1,Dilute,V-A26 Ammonia R2,Dilute,V-A26 Ammonia R3,Dilute,V-A26 Ammonia Solution,Aromatic,III-136 Ammonia Solution,Concentrated see Strong Ammonia SolutionAmmonia Solution,Dilute,III-137 Ammonia Spirit,Aromatic,III-137 Ammoniacal Copper Oxide Solution,V-A46Ammoniacal Silver Nitrate Solution,V-A120Ammoniacal Solution of Copper Tetrammine,V-A46Ammonia-free Water,V-A139 Ammonio Methacrylate Copolymer (Type A),I-155Ammonio Methacrylate Copolymer (Type B),I-1560.5M Ammonium acetate buffer solution pH4.5,see Ammonium acetate buffer pH4.5,0.5M0.01M Ammonium and Cerium Nitrate, see Ammonium Cerium(IV)Nitrate VS 0.1M Ammonium and Cerium Sulfate, see Ammonium Cerium(IV)Sulfate VS Ammonium Acetate,V-A26 Ammonium acetate buffer pH4.5,0.5M, V-A153Ammonium Acetate Solution,V-A26 Ammonium Acetate Solution,Strong,III-137Ammonium and Cerium Nitrate,see Ammonium Cerium(IV)Nitrate Ammonium and Cerium Sulfate,see Ammonium Cerium(IV)Sulfate Ammonium and Cerium Sulphate,see Ammonium and Cerium Sulfate Ammonium Bicarbonate,I-157 Ammonium Bromide,I-158 Ammonium Carbamate,V-A26 Ammonium Carbonate,V-A26 Ammonium Carbonate Buffer Solution pH10.3,0.1M,V-A153Ammonium Carbonate Solution,V-A26 Ammonium Carbonate Solution,Dilute, V-A26Ammonium carbonate solution R1,V-A26Ammonium Cerium(IV)Nitrate,V-A26Ammonium Cerium(IV)Nitrate VS,V-A142Ammonium Cerium(IV)Sulfate,V-A26Ammonium Cerium(IV)Sulfate VS,V-A142Ammonium Cerium(IV)Sulphate VS,seeAmmonium Cerium(IV)Sulfate VSAmmonium Cerium(IV)Sulphate,seeAmmonium Cerium(iv)SulfateAmmonium Chloride,I-159,V-A26Ammonium Chloride Buffer SolutionpH10.0,see Ammonia Buffer pH10.0Ammonium Chloride Buffer SolutionpH10.4,V-A153Ammonium Chloride Buffer SolutionpH10.7,V-A153Ammonium Chloride Buffer SolutionpH9.5,V-A153Ammonium Chloride Buffer SolutionpH10.0,V-A153Ammonium Chloride Mixture,III-137Ammonium Chloride Oral Solution,III-137Ammonium Chloride Solution,V-A26Ammonium Citrate,V-A26Ammonium Citrate Solution,V-A26Ammonium CobaltothiocyanateSolution,V-A26Ammonium DihydrogenOrthophosphate,V-A26Ammonium Formate,V-A26Ammonium Glycyrrhizinate,I-160Ammonium Hexafluorogermanate,V-A26Ammonium Hydrogen Carbonate,V-A26Ammonium Hydrogen Carbonate seeAmmonium BicarbonateAmmonium Ichthosulphonate seeIchthammolAmmonium Iron(II)Sulfate,V-A26Ammonium Iron(II)Sulfate VS,V-A142Ammonium Iron(II)Sulphate VS,seeAmmonium Iron(II)Sulfate VSAmmonium Iron(II)Sulphate,seeAmmonium Iron(ii)SulfateAmmonium Iron(III)Citrate,V-A26Ammonium Iron(III)Sulfate,V-A26Ammonium Iron(III)Sulfate Solution R1,V-A26Ammonium Iron(III)Sulfate Solution R2,V-A26Ammonium Iron(III)Sulfate Solution R5,V-A26Ammonium Iron(III)Sulfate Solution R6,V-A27Ammonium Iron(III)Sulfate VS,V-A142Ammonium Iron(III)Sulphate SolutionR1,see Ammonium Iron(iii)SulfateSolution R1Ammonium Iron(III)Sulphate SolutionR2,see Ammonium Iron(iii)SulfateSolution R2Ammonium Iron(III)Sulphate SolutionR5,see Ammonium Iron(iii)SulfateSolution R5Ammonium Iron(III)Sulphate VS,seeAmmonium Iron(III)Sulfate VSAmmonium Iron(III)Sulphate,seeAmmonium Iron(iii)SulfateAmmonium Mercaptoacetate Solution,V-A27Ammonium Mercurithiocyanate Reagent,V-A27Ammonium Metavanadate,V-A27Ammonium Metavanadate Solution,V-A27Ammonium Molybdate,V-A27Ammonium Molybdate Reagent,V-A27Ammonium Molybdate Reagent R1,V-A27Ammonium Molybdate Reagent R2,V-A27Ammonium Molybdate Solution,V-A27Ammonium Molybdate Solution R2,V-A27Ammonium Molybdate Solution R3,V-A27Ammonium Molybdate Solution R4,V-A27Ammonium Molybdate Solution R5,V-A27Ammonium Molybdate Solution R6,V-A27Ammonium Molybdate-Sulfuric AcidSolution,V-A27Ammonium Molybdate-Sulphuric AcidSolution,see Ammonium Molybdate-Sulfuric Acid SolutionAmmonium Muriaticum,V-609Ammonium Nitrate,V-A27Ammonium Nitrate R1,V-A27Ammonium Oxalate,V-A27Ammonium Oxalate Solution,V-A27Ammonium Persulfate,V-A27Ammonium Persulphate,see AmmoniumPersulfateAmmonium Phosphate,see DiammoniumHydrogen OrthophosphateAmmonium Polysulfide Solution,V-A27Ammonium Polysulphide Solution,seeAmmonium Polysulfide SolutionAmmonium Pyrrolidinedithiocarbamate,V-A27Ammonium PyrrolidinedithiocarbamateSolution,V-A27Ammonium Reineckate,V-A27Ammonium Reineckate Solution,V-A28Ammonium Salts and Salts of VolatileBases,Reactions of,V-267Ammonium Salts Reactions of,V-266Ammonium Standard Solution(1ppmNH4),V-A148Ammonium Standard Solution(2.5ppmNH4),V-A148Ammonium Standard Solution(3ppmNH4),V-A148Ammonium Standard Solution(100ppmNH4),V-A148Ammonium Sulfamate,V-A28Ammonium Sulfate,V-A28Ammonium Sulfide Solution,V-A28Ammonium Sulphamate,see AmmoniumSulfamateAmmonium Sulphate,see AmmoniumSulfateAmmonium Sulphide Solution,seeAmmonium Sulfide SolutionV-A802IndexAmmonium Thiocyanate,V-A28 Ammonium Thiocyanate Solution,V-A28Ammonium Thiocyanate VS,V-A142 Ammonium Vanadate Solution,V-A28 Ammonium Vanadate,see Ammonium MetavanadateAmobarbital,I-161Amobarbital Sodium,I-162Amomum fruit,IV-56Amorphous Organosilica Polymer, Octadecylsilyl,V-A98Amoxicillin and Potassium Clavulanate Injection,see Co-amoxiclav Injection Amoxicillin and Potassium Clavulanate Oral Suspension,see Co-amoxiclav Oral SuspensionAmoxicillin and Potassium Clavulanate Tablets,Dispersible,see Dispersible Co-amoxiclav TabletsAmoxicillin and Potassium Clavulanate Tablets,see Co-amoxiclav Tablets Amoxicillin Capsules,III-138 Amoxicillin Injection,III-139 Amoxicillin Oral Suspension,III-141 Amoxicillin Sodium,I-163,V-S9 Amoxicillin Sodium for Injection,III-139Amoxicillin Trihydrate,I-165,V-S9,V-A28Ampere,Definition of,I-32,II-32,III-32,IV-32,V-32 Amperometric,Potentiometric and Voltametric Titrations,V-280 Amperometric Titration,V-280 Amphotericin,I-168Amphotericin B see Amphotericin Amphotericin for Infusion,III-142 Amphotericin Lozenges,I-xxix Amphotericin Oral Suspension,I-xxix Ampicillin,I-170Ampicillin Capsules,III-143Ampicillin Capsules,Flucloxacillin and, see Co-fluampicil CapsulesAmpicillin Injection,III-144Ampicillin Oral Suspension,III-146 Ampicillin Oral Suspension, Flucloxacillin and,see Co-fluampicil Oral SuspensionAmpicillin Sodium,I-172,V-S10 Ampicillin Sodium for Injection,III-144 Ampicillin Trihydrate,I-175,V-S10 Amyl Acetate,V-A28Amyl Alcohol,see Isoamyl Alcohola-Amylase,V-A28a-Amylase Solution,V-A28 Amylmetacresol,I-178,V-S10Amylose-derivative Silica Gel for Chromatography,V-A115b-Amyrin,V-A28Anacardium for Homoeopathic Preparations,IV-428Anaesthetic Ether,I-902Analytical Procedures,Validation of,V-673Analytical Sieving,Particle-size Distribution Estimation By,V-503 Anastrozole,I-179cis-Anethole,V-A28Anethum Graveolens L.Sowa Group,seeAnethum Graveolens Sowa Fruit,Anethum Graveolens Sowa Fruit,IV-58Angelica Archangelica Root,IV-59Angelica Dahurica Root,IV-60Angelica Pubescens Root,IV-62Angelica Sinensis Root,IV-63Angelica Sinensis Root,see ProcessedAngelica Sinensis RootAnhydrous Acetic Acid,V-A20Anhydrous Aluminium Oxide,V-A23Anhydrous Ampicilin see AmpicillinAnhydrous Azapropazone,V-S12Anhydrous Beclometasone Dipropionate,I-239Anhydrous Caffeine,see CaffeineAnhydrous Calcipotriol,I-353Anhydrous Calcium Acetate,see CalciumAcetateAnhydrous Calcium Chloride,V-A37Anhydrous Calcium Gluconate,I-372Anhydrous Calcium HydrogenPhosphate,I-377Anhydrous Calcium Lactate,I-378Anhydrous Chlorobutanol,I-518Anhydrous Citric Acid,I-569,V-A45Anhydrous Copper Sulfate,I-647Anhydrous Disodium HydrogenOrthophosphate,V-A59Anhydrous Disodium HydrogenPhosphate,I-788Anhydrous Docetaxel,I-796Anhydrous Ephedrine,I-849Anhydrous Formic Acid,V-A67Anhydrous Glucose,I-1083Anhydrous Iron(III)Chloride,V-A77Anhydrous Lactose,II-66Anhydrous Lithium Metaborate,V-A81Anhydrous Magnesium Citrate,II-166Anhydrous Methanol,V-A85Anhydrous Morphine,V-A91Anhydrous Nevirapine,II-358Anhydrous Niclosamide,II-362Anhydrous Paroxetine Hydrochloride,II-504Anhydrous Phloroglucinol,II-566Anhydrous Pyridine,V-A110Anhydrous Silica Gel,V-A114Anhydrous Silica,HydrophobicColloidal,II-807Anhydrous Sodium Acetate,V-A120Anhydrous Sodium Carbonate,II-830,V-A121,V-A141Anhydrous Sodium DihydrogenOrthophosphate,V-A122Anhydrous Sodium DihydrogenPhosphate,II-839,V-A122Anhydrous Sodium Sulfate,II-872,V-A124Anhydrous Sodium Sulfite,II-873,V-A124Anhydrous Sodium Sulphate seeAnhydrous Sodium SulfateAnhydrous Sodium Sulphite seeAnhydrous Sodium SulfiteAnhydrous Torasemide,II-1066Anhydrous Valaciclovir Hydrochloride,II-1134Aniline,V-A28Aniline Hydrochloride,V-A28Aniline Hydrochloride Solution,V-A28Animal Spongiform EncephalopathyAgents Via Human and VeterinaryMedicinal Products,Minimising theRisk of Transmitting,V-611Animals,Use of,I-15,II-15,III-15,IV-15,V-15Anion Exchange Resin,V-A28Anion Exchange Resin forChromatography,Strongly Basic,V-A28Anion Exchange Resin R1,V-A28Anion Exchange Resin R2,V-A28Anion exchange resin R3,V-A29Anion Exchange Resin,Strongly Basic,V-A28Anion Exchange Resin,Weak,V-A29Anion-exchange Resin forChromatography,Strongly Basic R1,V-A28Anionic Emulsifying Wax,see EmulsifyingWaxAnisaldehyde,V-A29Anisaldehyde Solution,V-A29Anisaldehyde Solution R1,V-A29Anise Ketone,V-A29Anise Oil,IV-71Anise Water,Concentrated,IV-73Aniseed,IV-66Aniseed Oil,see Anise Oilp-Anisidine,V-A29Anisidine Value,V-326Anolyte for Isoelectric Focusing pH3to5,V-A29Antazoline Hydrochloride,I-181Anthracene,V-A29Anthranilic Acid,see2-Aminobenzoic AcidAnthrax,see Anthrax Vaccine for HumanUse(Adsorbed,Prepared from CultureFiltrates)Anthrax Vaccine for Human Use(Adsorbed,Prepared from CultureFiltrates),IV-527Anthrone,V-A29Anthrone Reagent,V-A29Antibiotics,Microbiological Assay of,V-396,V-655Antibiotics,Potency of,I-14,II-14,III-14,IV-14,V-14Anticoagulant and Preservative Solutionsfor Blood,IV-461Anti-D Immunoglobulin for IntravenousUse,IV-497Anti-D(Rh0)Immunoglobulin,IV-496Antimicrobial Preservation,Efficacy of,V-494,V-653Antithrombin III ConcentrateAnticomplimentary activity ofimmunoglobulin,Test for V-427Anti-D immunoglobulin,human,Assayof V-429Anti-D antibodies in humanimmunoglobulin V-431Anti-A and anti-B haemogglutininsV-432Antimicrobial Preservatives,Definition ofSuitable,I-11,II-11,III-11,IV-11,V-11Antimony Compounds,Reactions of,V-267page numbers in bold type relate to monograph titles Index V-A803。
常用蛋白酶切割位点
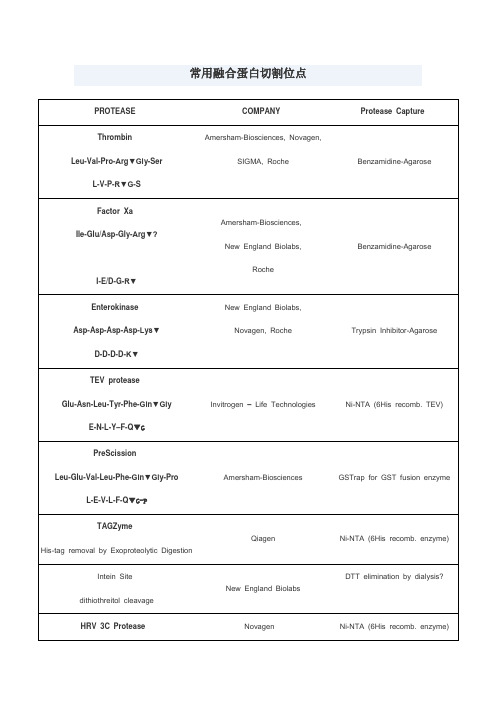
Intein Site
dithiothreitol cleavage
New England Biolabs
DTT elimination by dialysis?
HRV 3C Protease
Leu-Glu-Val-Leu-Phe-Gln▼Gly-Pro
3.羧肽酶(分A和B型),一般的题目中没有特别指明的话就是两种类型的功能都具备,可以从羧基端切除氨基酸(若羧基端的第1个或第2个氨基酸为Pro的则不能切除)。
4.溴化氰处理,专一性的切割甲硫氨酸羧基端的肽键。
Leu-Glu-Val-Leu-Phe-Gln▼Gly-Pro
L-E-V-L-F-Q▼G-P
Amersham-Biosciences
GSTrap for GST fusion enzyme
TAGZyme
His-tag removal by Exoproteolytic Digestion
Qiagen
常用融合蛋白切割位点
PROTEASE
COMPANY
Protease Capture
Thrombin
Leu-Val-Pro-Arg▼Gly-Ser
L-V-P-R▼G-S
Amersham-Biosciences, Novagen,
SIGMA, Roche
Benzamidine-Agarose
Factor Xa
LifeSensors
Ni-NTA (6His recomb. enzyme)
-2
-Arg-X-Lys/Arg-Arg▼
Invitrogen – Life Technologies,
Ni-NTA (6His recomb. enzyme)
高效液相色谱法测定注射用美罗培南的有关物质

第35卷第3期 长治医学院学报2021 年 6 月JOURNAL OF CHANGZHI MEDICAI COLLEGE167Vol. 35 No. 3Jun. 2021高效液相色谱法测定注射用美罗培南的有关物质李金格禹玉洪**作者单位山西医科大学药学院药剂教研室(030001)* 通信作者(E-mail :3024546064@ qq. com)摘要目的:探讨优化注射用美罗培南杂质A 、B 的测定方法。
方法:运用高效液相色谱法(HPLC) 进行检测,色谱柱以十八烷基硅烷键合硅胶为填充剂;流动相A :20. 0 mmol-L'1磷酸二氢钠-甲醇(89 :11, V/V),流动相B :甲醇,流速1.0 mL-min 1,检测波长220 nm,柱温30 P 。
结果:主成分峰与杂质峰可实现基线分离,杂质A 检测限和定量限分别为1. 62,5.15 ng,杂质B 检测限和定量限分别为0. 85,2. 51 ng ;1.2~24.0 ixg-mL -1的杂质A 具有良好的线性关系(r=0. 999 9) ,0.7-14.0 ixg-mL 1的杂质B 具有良好的线性 关系(r=0. 999 9);杂质A 平均加样回收率为101.2%(RSD= 1.38%,“ = 9),杂质B 平均加样回收率为100.2%(RSD=1.29%,n = 9)o 经破坏性试验,美罗培南可能的降解杂质A 、B 均不干扰美罗培南主峰的测定。
结论:检测限及定量限、精密度、稳定性、耐用性试验结果均符合HPLC 有关物质测定的方法学验证要求。
本HPLC 法专属性良好,可用于美罗培南的主要杂质A 、B 的定量控制。
关键词美罗培南;有关物质;高效液相色谱法中图分类号R97&1文献标识码 A 文章编号1006(2021)03-167-05Determination of Related Substances of Meropenem for Injection by High Performance Liquid ChromatographyLI Jinge , YU YuhongDepartment of Pharmacy , School of Pharmacy , Shanxi Medical UniversityAbstract Objective : To explore and optimize the determination method of impulity A andB of meropenem for injection. Meth ods :Using the high performance liquid chromatography ( HPLC ) to detection , Octadecylsilane-bonded silica gel was used as the fi ler ; The mobile phase A : 20. 0 mmol * L -1 sodium dihydrogen phosphate-methanol ( 89 : 11, V/V) . The mobile phase B : methanol ,the flow rate was 1. 0 mL *m in _1 and the detection wavelength was set at 220 nm. The column temperature was set at 30 % . Re sults :The principal component peak and impurity peak could achieve baseline separation. The detection limit and quantitative limit of impurity A were 1. 62 ng and 5. 15 ng respectively , and the detection limit and quantitative lim 让 of impurity B were 0. 85 ng and 2. 51 ng respectively. There was A good linear relationship between impurity A (r = 0. 999 9) and impurity B ( r= 0. 999 9 ) in therange of 1. 2-24. 0 |xg *m L _1 and 0. 7 ~ 14. 0 jig * mL -1. The average recovery of impurity A was 101. 2% ( RSD = 1. 38% , n = 9),and that of impurity B was 100. 2% ( RSD = 1. 29% , n= 9). After stressing test, both of impurities A and B of meropenem didn * tinterfere w 让h the determination of meropenem main peak. Conclusion : The test results of detection lim 让 and quant N ative lim 让,pre cision ,stabil 让y and durability all meet the methodological verification requirements of HPLC related substance determination. The HPLC method has good specificity and can be used for the quant N ative control of major impurities A and B.Key words meropenem ; related substances ; HPLC注射用美罗培南(Meropenem, C ”H25弘0申) 是由日本住友制药公司与英国ICI 制药公司共同 开发的第二代碳青霉烯抗生素,通过干扰细菌细胞壁的合成发挥杀菌作用,具有广谱耐酶的特 点[1_4]o 在美罗培南原料中常检测出杂质A 及杂质B,杂质A(C 17H 27N 3O 6S)为美罗培南四元内酰 胺环结构发生水解反应而形成,系美罗培南的降 解产物;杂质B(C 34H 50N 6O 10S 2)为美罗培南与杂质A 发生聚合反应而形成,系美罗培南的二聚体X 。
重组贻贝粘蛋白的表征及功效评价

生物技术进展 2023 年 第 13 卷 第 4 期 596 ~ 603Current Biotechnology ISSN 2095‑2341研究论文Articles重组贻贝粘蛋白的表征及功效评价李敏 , 魏文培 , 乔莎 , 郝东 , 周浩 , 赵硕文 , 张立峰 , 侯增淼 *西安德诺海思医疗科技有限公司,西安 710000摘要:为了推进重组贻贝粘蛋白在医疗、化妆品领域的应用,对大肠杆菌规模化发酵及纯化生产获得的重组贻贝粘蛋白进行了表征及功效评价。
经Edman 降解法、基质辅助激光解吸电离飞行时间质谱、PITC 法、非还原型SDS -聚丙烯酰胺凝胶电泳法、凝胶法、改良的Arnow 法对重组贻贝粘蛋白进行氨基酸N 端测序、相对分子量分析、氨基酸组成分析、蛋白纯度分析、内毒素含量测定、多巴含量测定;通过细胞迁移、斑马鱼尾鳍修复效果对重组贻贝粘蛋白进行功效评价。
结果显示,获得的重组贻贝粘蛋白与理论的一级结构一致,蛋白纯度达95%以上,内毒素<10 EU ·mg -1,多巴含量大于5%;重组贻贝粘蛋白浓度为60 μg ·mL -1时能够显著促进细胞增殖的活性(P <0.01);斑马鱼尾鳍面积样品组与模型对照组相比极显著增加(P <0.001)。
研究结果表明,重组贻贝粘蛋白具有显著的促细胞迁移和修复愈合的功效,具备作为生物医学材料的潜质。
关键词:贻贝粘蛋白;基因重组;生物材料;表征;功效评价DOI :10.19586/j.20952341.2023.0021 中图分类号:S985.3+1 文献标志码:ACharacterization and Efficacy Evaluation of Recombinant Mussel Adhesive ProteinLI Min , WEI Wenpei , QIAO Sha , HAO Dong , ZHOU Hao , ZHAO Shuowen , ZHANG Lifeng ,HOU Zengmiao *Xi'an DeNovo Hith Medical Technology Co., Ltd , Xi'an 710000, ChinaAbstract :In order to promote the application of recombinant mussel adhesive protein in the medical and cosmetics field , the recombi⁃nant mussel adhesive protein obtained from scale fermentation and purification of Escherichia coli was characterized and its efficacy was evaluated. Amino acid N -terminal sequencing , relative molecular weight analysis , amino acid composition analysis , protein purityanalysis , endotoxin content , dihydroxyphenylalanine (DOPA ) content of recombinant mussel adhesive protein were determined by the following methods : Edman degradation , matrix -assisted laser desorption ionization time -of -flight mass spectrometry (MALDI -TOF -MS ), phenyl -isothiocyanate (PITC ), nonreductive SDS -polyacrylamide gel electrophoresis (SDS -PAGE ), gel method , modified Ar⁃now. The efficacy of recombinant mussel adhesive protein was evaluated by cell migration and repairing effect of zebrafish tail fin. Re⁃sults showed that the obtained recombinant mussel adhesive protein was confirmed to be consistent with the theoretical primary structure , protein purity of more than 95%, endotoxin <10 EU ·mg -1, DOPA content above 5%. When the recombinant mussel adhesive protein concentration was 60 μg ·mL -1, the effect of promoting cell proliferation was the most obvious , and it had very significant activity (P <0.01). The caudal fin area of zebrafish in sample group was significantly increased compared with model control group (P <0.001). The results indicated that recombinant mussel adhesive protein can promote cell migration and repair healing and has the potential to be used as biomedical materials.Key words :mussel adhesive protein ; gene recombination ; biological materials ; representation ; efficacy evaluation贻贝粘蛋白(mussel adhesive protein , MAP )也称作贻贝足丝蛋白(mussel foot protein ,Mfps ),收稿日期:2023⁃02⁃24; 接受日期:2023⁃03⁃31联系方式:李敏 E -mail:*******************;*通信作者 侯增淼 E -mail:***********************.cn李敏,等:重组贻贝粘蛋白的表征及功效评价是海洋贝类——紫贻贝(Mytilus galloprovincalis)、厚壳贻贝(Mytilus coruscus)、翡翠贻贝(Perna viri⁃dis)等分泌的一种特殊的蛋白质,贻贝中含有多种贻贝粘蛋白,包括贻贝粘蛋白(Mfp 1~6)、前胶原蛋白(precollagens)和基质蛋白(matrix proteins)等[1]。
菠萝蛋白酶检测试剂盒(福林酚比色法)
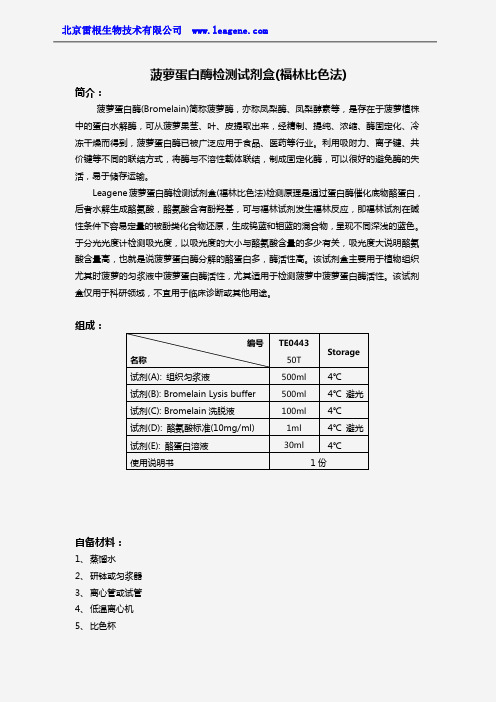
菠萝蛋白酶检测试剂盒(福林比色法)简介:菠萝蛋白酶(Bromelain)简称菠萝酶,亦称凤梨酶、凤梨酵素等,是存在于菠萝植株中的蛋白水解酶,可从菠萝果茎、叶、皮提取出来,经精制、提纯、浓缩、酶固定化、冷冻干燥而得到,菠萝蛋白酶已被广泛应用于食品、医药等行业。
利用吸附力、离子键、共价键等不同的联结方式,将酶与不溶性载体联结,制成固定化酶,可以很好的避免酶的失活,易于储存运输。
Leagene菠萝蛋白酶检测试剂盒(福林比色法)检测原理是通过蛋白酶催化底物酪蛋白,后者水解生成酪氨酸,酪氨酸含有酚羟基,可与福林试剂发生福林反应,即福林试剂在碱性条件下容易定量的被酚类化合物还原,生成钨蓝和钼蓝的混合物,呈现不同深浅的蓝色。
于分光光度计检测吸光度,以吸光度的大小与酪氨酸含量的多少有关,吸光度大说明酪氨酸含量高,也就是说菠萝蛋白酶分解的酪蛋白多,酶活性高。
该试剂盒主要用于植物组织尤其时菠萝的匀浆液中菠萝蛋白酶活性,尤其适用于检测菠萝中菠萝蛋白酶活性。
该试剂盒仅用于科研领域,不宜用于临床诊断或其他用途。
组成:自备材料:1、蒸馏水2、研钵或匀浆器3、离心管或试管4、低温离心机5、比色杯编号名称TE044350TStorage试剂(A):组织匀浆液500ml4℃试剂(B):Bromelain Lysis buffer500ml4℃避光试剂(C):Bromelain洗脱液100ml4℃试剂(D):酪氨酸标准(10mg/ml)1ml4℃避光试剂(E):酪蛋白溶液30ml4℃使用说明书1份6、分光光度计操作步骤(仅供参考):1、准备样品:①植物样品:取菠萝肉或植物组织,切碎后在研钵或匀浆器中加入等体积组织匀浆液进行研磨或匀浆,静置,滤纸过滤或纱布过滤。
留取滤液,边搅拌边加入Bromelain Lysis buffer,搅拌约,静置。
离心,弃上清液,收集沉淀,加入Bromelain 洗脱液,搅拌洗脱。
,离心,取上清液,冻存,用于菠萝蛋白酶的检测。
高密度脂蛋白胆固醇测定试剂盒(磷钨酸镁沉淀法)产品技术要求zhongshengbeikong

高密度脂蛋白胆固醇测定试剂盒(磷钨酸镁沉淀法)适用范围:本产品用于体外定量测定人血清中高密度脂蛋白胆固醇的浓度。
1.1规格干粉型试剂1(R1):10mL×7,试剂2(R2):70mL×1,试剂3(R3):10mL×3,校准品:1mL×11.2规格划分说明根据净含量和复溶体积划分规格。
1.3主要组成成分试剂盒由试剂1(R1)干粉、试剂2(R2)液体、试剂3(R3)液体及校准品液体组成。
1.3.1 试剂1(R1)干粉胆固醇脂酶(CEH)≥1140U/L胆固醇氧化酶(CHOD)≥800U/L过氧化物酶(POD)≥6000U/L1.3.2 试剂2(R2)液体缓冲液 100mmol/L 4-氯酚 3.5mmol/L 4-氨基安替吡啉 0.5mmol/L1.3.3 试剂3(R3)液体磷钨酸氯化镁1.3.4 校准品:牛血清基质(1个浓度)HDL-C校准品定值范围 1.03 mmol/L ~2.07 mmol/L(每批定值)2.1 外观试剂盒中各组件的外观应满足:a) 试剂1(R1)应为白色干粉,外包装完整无破损;b) 试剂2(R2)应为无色透明溶液,无杂质、无絮状物,外包装完整无破损;c) 试剂3(R3)应为无色透明溶液,无杂质、无絮状物,外包装完整无破损;d) 校准品应为浅黄色溶液,无杂质、无絮状物,外包装完整无破损。
2.2 净含量液体试剂净含量应不少于标示值。
2.3 试剂空白吸光度在波长500 nm(480nm~520nm)处(光径1cm),试剂空白吸光度(A)应<0.050。
2.4准确度测定GBW09178,相对偏差应不超过±10%。
2.5分析灵敏度对应于浓度为1.00mmol/L的高密度脂蛋白胆固醇所引起的吸光度差值(△A)的绝对值应在>0.04。
2.6批内瓶间差重复测试血清样本,批内瓶间差的变异系数(CV)应≤4%。
2.7批间差测定血清样本,批间差(R)应≤6%。
Relebactam_SDS_MedChemExpress

Inhibitors, Agonists, Screening LibrariesSafety Data Sheet Revision Date:May-24-2017Print Date:May-24-20171. PRODUCT AND COMPANY IDENTIFICATION1.1 Product identifierProduct name :RelebactamCatalog No. :HY-16752CAS No. :1174018-99-51.2 Relevant identified uses of the substance or mixture and uses advised againstIdentified uses :Laboratory chemicals, manufacture of substances.1.3 Details of the supplier of the safety data sheetCompany:MedChemExpress USATel:609-228-6898Fax:609-228-5909E-mail:sales@1.4 Emergency telephone numberEmergency Phone #:609-228-68982. HAZARDS IDENTIFICATION2.1 Classification of the substance or mixtureNot a hazardous substance or mixture.2.2 GHS Label elements, including precautionary statementsNot a hazardous substance or mixture.2.3 Other hazardsNone.3. COMPOSITION/INFORMATION ON INGREDIENTS3.1 SubstancesSynonyms:MK⁻7655; MK7655; MK 7655Formula:C12H20N4O6SMolecular Weight:348.38CAS No. :1174018-99-54. FIRST AID MEASURES4.1 Description of first aid measuresEye contactRemove any contact lenses, locate eye-wash station, and flush eyes immediately with large amounts of water. Separate eyelids with fingers to ensure adequate flushing. Promptly call a physician.Skin contactRinse skin thoroughly with large amounts of water. Remove contaminated clothing and shoes and call a physician.InhalationImmediately relocate self or casualty to fresh air. If breathing is difficult, give cardiopulmonary resuscitation (CPR). Avoid mouth-to-mouth resuscitation.IngestionWash out mouth with water; Do NOT induce vomiting; call a physician.4.2 Most important symptoms and effects, both acute and delayedThe most important known symptoms and effects are described in the labelling (see section 2.2).4.3 Indication of any immediate medical attention and special treatment neededTreat symptomatically.5. FIRE FIGHTING MEASURES5.1 Extinguishing mediaSuitable extinguishing mediaUse water spray, dry chemical, foam, and carbon dioxide fire extinguisher.5.2 Special hazards arising from the substance or mixtureDuring combustion, may emit irritant fumes.5.3 Advice for firefightersWear self-contained breathing apparatus and protective clothing.6. ACCIDENTAL RELEASE MEASURES6.1 Personal precautions, protective equipment and emergency proceduresUse full personal protective equipment. Avoid breathing vapors, mist, dust or gas. Ensure adequate ventilation. Evacuate personnel to safe areas.Refer to protective measures listed in sections 8.6.2 Environmental precautionsTry to prevent further leakage or spillage. Keep the product away from drains or water courses.6.3 Methods and materials for containment and cleaning upAbsorb solutions with finely-powdered liquid-binding material (diatomite, universal binders); Decontaminate surfaces and equipment by scrubbing with alcohol; Dispose of contaminated material according to Section 13.7. HANDLING AND STORAGE7.1 Precautions for safe handlingAvoid inhalation, contact with eyes and skin. Avoid dust and aerosol formation. Use only in areas with appropriate exhaust ventilation.7.2 Conditions for safe storage, including any incompatibilitiesKeep container tightly sealed in cool, well-ventilated area. Keep away from direct sunlight and sources of ignition.Recommended storage temperature:Powder-20°C 3 years4°C 2 yearsIn solvent-80°C 6 months-20°C 1 monthShipping at room temperature if less than 2 weeks.7.3 Specific end use(s)No data available.8. EXPOSURE CONTROLS/PERSONAL PROTECTION8.1 Control parametersComponents with workplace control parametersThis product contains no substances with occupational exposure limit values.8.2 Exposure controlsEngineering controlsEnsure adequate ventilation. Provide accessible safety shower and eye wash station.Personal protective equipmentEye protection Safety goggles with side-shields.Hand protection Protective gloves.Skin and body protection Impervious clothing.Respiratory protection Suitable respirator.Environmental exposure controls Keep the product away from drains, water courses or the soil. Cleanspillages in a safe way as soon as possible.9. PHYSICAL AND CHEMICAL PROPERTIES9.1 Information on basic physical and chemical propertiesAppearance White to off-white (Solid)Odor No data availableOdor threshold No data availablepH No data availableMelting/freezing point No data availableBoiling point/range No data availableFlash point No data availableEvaporation rate No data availableFlammability (solid, gas)No data availableUpper/lower flammability or explosive limits No data availableVapor pressure No data availableVapor density No data availableRelative density No data availableWater Solubility No data availablePartition coefficient No data availableAuto-ignition temperature No data availableDecomposition temperature No data availableViscosity No data availableExplosive properties No data availableOxidizing properties No data available9.2 Other safety informationNo data available.10. STABILITY AND REACTIVITY10.1 ReactivityNo data available.10.2 Chemical stabilityStable under recommended storage conditions.10.3 Possibility of hazardous reactionsNo data available.10.4 Conditions to avoidNo data available.10.5 Incompatible materialsStrong acids/alkalis, strong oxidising/reducing agents.10.6 Hazardous decomposition productsUnder fire conditions, may decompose and emit toxic fumes.Other decomposition products - no data available.11.TOXICOLOGICAL INFORMATION11.1 Information on toxicological effectsAcute toxicityClassified based on available data. For more details, see section 2Skin corrosion/irritationClassified based on available data. For more details, see section 2Serious eye damage/irritationClassified based on available data. For more details, see section 2Respiratory or skin sensitizationClassified based on available data. For more details, see section 2Germ cell mutagenicityClassified based on available data. For more details, see section 2CarcinogenicityIARC: No component of this product present at a level equal to or greater than 0.1% is identified as probable, possible or confirmed human carcinogen by IARC.ACGIH: No component of this product present at a level equal to or greater than 0.1% is identified as a potential or confirmed carcinogen by ACGIH.NTP: No component of this product present at a level equal to or greater than 0.1% is identified as a anticipated or confirmed carcinogen by NTP.OSHA: No component of this product present at a level equal to or greater than 0.1% is identified as a potential or confirmed carcinogen by OSHA.Reproductive toxicityClassified based on available data. For more details, see section 2Specific target organ toxicity - single exposureClassified based on available data. For more details, see section 2Specific target organ toxicity - repeated exposureClassified based on available data. For more details, see section 2Aspiration hazardClassified based on available data. For more details, see section 212. ECOLOGICAL INFORMATION12.1 ToxicityNo data available.12.2 Persistence and degradabilityNo data available.12.3 Bioaccumlative potentialNo data available.12.4 Mobility in soilNo data available.12.5 Results of PBT and vPvB assessmentPBT/vPvB assessment unavailable as chemical safety assessment not required or not conducted.12.6 Other adverse effectsNo data available.13. DISPOSAL CONSIDERATIONS13.1 Waste treatment methodsProductDispose substance in accordance with prevailing country, federal, state and local regulations.Contaminated packagingConduct recycling or disposal in accordance with prevailing country, federal, state and local regulations.14. TRANSPORT INFORMATIONDOT (US)This substance is considered to be non-hazardous for transport.IMDGThis substance is considered to be non-hazardous for transport.IATAThis substance is considered to be non-hazardous for transport.15. REGULATORY INFORMATIONSARA 302 Components:No chemicals in this material are subject to the reporting requirements of SARA Title III, Section 302.SARA 313 Components:This material does not contain any chemical components with known CAS numbers that exceed the threshold (De Minimis) reporting levels established by SARA Title III, Section 313.SARA 311/312 Hazards:No SARA Hazards.Massachusetts Right To Know Components:No components are subject to the Massachusetts Right to Know Act.Pennsylvania Right To Know Components:No components are subject to the Pennsylvania Right to Know Act.New Jersey Right To Know Components:No components are subject to the New Jersey Right to Know Act.California Prop. 65 Components:This product does not contain any chemicals known to State of California to cause cancer, birth defects, or anyother reproductive harm.16. OTHER INFORMATIONCopyright 2017 MedChemExpress. The above information is correct to the best of our present knowledge but does not purport to be all inclusive and should be used only as a guide. The product is for research use only and for experienced personnel. It must only be handled by suitably qualified experienced scientists in appropriately equipped and authorized facilities. The burden of safe use of this material rests entirely with the user. MedChemExpress disclaims all liability for any damage resulting from handling or from contact with this product.Caution: Product has not been fully validated for medical applications. For research use only.Tel: 609-228-6898 Fax: 609-228-5909 E-mail: tech@Address: 1 Deer Park Dr, Suite Q, Monmouth Junction, NJ 08852, USA。
HPLC法测定乐脉软胶囊中芍药苷的含量

HPLC法测定乐脉软胶囊中芍药苷的含量
徐华;高钢
【期刊名称】《北方药学》
【年(卷),期】2015(000)002
【摘要】目的:建立乐脉软胶囊中芍药苷的含量测定方法。
方法:HPLC法,流动相为甲醇—异丙醇—冰醋酸—水(25∶2∶2∶71),测定波长230nm。
结果:芍药苷在21.2~84.8μg/ml范围内呈线性关系,回归方程:Y=-
0.918743604+0.0001140143928X,γ=0.9999。
样品中芍药苷平均含量
8.356mg·粒-1。
平均回收率99.48%。
结论:本法可作为乐脉软胶囊中芍药苷的含量测定方法。
【总页数】1页(P12-12)
【作者】徐华;高钢
【作者单位】内蒙古包头市食品药品检验检测中心包头 014040;内蒙古包头市食品药品检验检测中心包头 014040
【正文语种】中文
【中图分类】R927.2
【相关文献】
1.HPLC法测定丹瓜护脉口服液中芍药苷的含量
2.HPLC测定乐脉软胶囊中芍药苷的含量
3.HPLC法测定畅脉乐胶囊中芍药苷的含量
4.HPLC法测定丹瓜护脉颗粒中
芍药苷和丹酚酸B的含量5.HPLC法测定乐脉颗粒中丹参素、羟基红花黄色素A 和芍药苷
因版权原因,仅展示原文概要,查看原文内容请购买。
高效液相色谱法测定自制苯海拉明乳膏中羟苯乙酯的含量

高效液相色谱法测定自制苯海拉明乳膏中羟苯乙酯的含量湛雯;徐进;孙芳;景霞;许静【摘要】目的:建立测定医院自制制剂苯海拉明乳膏中羟苯乙酯的含量测定方法。
方法采用高效液相色谱法(HPLC)。
色谱柱:Agilent Eclipse Plus-C18(4.6 mm ×250 mm,5μm)色谱柱;流动相:甲醇-0.03 mol·L -1磷酸二氢钾溶液(含0.1%三乙胺,用50%磷酸调 pH 3.0±0.03)=60∶40;检测波长:258 nm;柱温:25℃;流速:0.9 mL·min -1;进样体积:20μL。
结果羟苯乙酯测定质量浓度在0.02~0.10 g·L -1范围内与其相应峰面积积分值呈良好线性关系(r =0.9995);精密度、稳定性、重复性试验相对标准偏差(RSD)分别为1.6%、1.9%、2.0%;平均绝对回收率为92.7%,RSD 为4.3%(n =9);平均相对回收率为102.5%,RSD 为3.6%(n =9)。
结论该方法专属性、重复性、准确性较好,可用于苯海拉明乳膏中羟苯乙酯的含量测定。
%Objective To establish a method of quantitative determination of ethyl hydroxybenzoate in diphenhydramine cream.Meth-ods The HPLC method was adopted with the Agilent Eclipse Plus-C18 column(4.6 mm×250 mm,5 μm)at room temperature(25℃)and methanol-0.03 mol·L -1 phosphoric acid solution(pH adjust ed to 3.0 ±0.03)(60∶40)as the mobile phase at the flow rate of 0.9 mL·min -1 .The detection wavelength for ethyl hydroxybenzoate was 258 nm,and the injection volume was 20 μL.Results The good linear relationship for ethyl hydroxybenzoate was in the range of 0.02 ~0.10 g·L -1 (r =0.999 5).RSDs of ethyl hydroxybenzo-ate for precision,stability and reproducibility tests were 1.6 %,1.9% and2.0 %,respectively.The average absolute recovery of ethyl hydroxybenzoatewas 92.7%(RSD =4.3 %,n =9),while the average relative recovery was102.5 %(RSD =3.6 %,n =9).Con-clusions This established method was specific,accurate and reproducible for the quantitative determination of ethyl hydroxybenzoate in diphenhydramine cream.【期刊名称】《安徽医药》【年(卷),期】2016(020)009【总页数】4页(P1655-1657,1658)【关键词】防腐剂,药物;色谱法,高压液相;苯海拉明;药物调剂;羟苯乙酯/分析【作者】湛雯;徐进;孙芳;景霞;许静【作者单位】南京医科大学附属南京儿童医院药学部,江苏南京 210008;南京医科大学附属南京儿童医院药学部,江苏南京 210008;南京医科大学附属南京儿童医院药学部,江苏南京 210008;南京医科大学附属南京儿童医院药学部,江苏南京 210008;南京医科大学附属南京儿童医院药学部,江苏南京 210008【正文语种】中文抑菌剂是能有效防止或抑制微生物的生长的化学物质。
固相萃取高效液相色谱法测定蜂王浆啤酒中的1O-羟基-2-癸烯酸

固相萃取高效液相色谱法测定蜂王浆啤酒中的1O-羟基-2-癸
烯酸
李蔚;邓六爱;陈金东;韩同升
【期刊名称】《化学分析计量》
【年(卷),期】2005(014)001
【摘要】建立了固相萃取高效液相色谱法测定蜂王浆啤酒中10-羟基-2-癸烯酸含量的方法.采用固相萃取技术富集蜂王浆中的10-羟基-2-癸烯酸,以甲醇-水-磷酸(体积比为50:50:0.5)为流动相,ODS柱分离,用紫外检测器于215 nm处检测.10-羟基-2-癸烯酸的浓度在0.5~100.0μg/mL范围内与其色谱峰峰面积呈良好线性关系(r=0.9999),回收率为98%~108%,RSD为2.1%.
【总页数】2页(P38-39)
【作者】李蔚;邓六爱;陈金东;韩同升
【作者单位】山东省疾病预防控制中心,济南,250014;日照市产品质量监督检验所,日照,276826;山东省疾病预防控制中心,济南,250014;山东省广播电视报社,济南,250011
【正文语种】中文
【相关文献】
1.高效液相色谱法测定蜂王浆中10-羟基-2-癸烯酸的含量 [J], 郭小莉
2.高效液相色谱法测定蜂王浆中特有成分10-羟基-2-癸烯酸的研究 [J], 张燮
3.反相高效液相色谱法测定蜂王浆中的10-羟基-2-癸烯酸 [J], 杨彬;郝莉花;陈欣
欣
4.蜂王浆中10-羟基癸2-烯酸的含量测定 [J], 张莉萍
5.高效液相色谱法测定蜂王浆中10-羟基-2-癸烯酸 [J], 邹力;严烨;范芳芳;吴坤因版权原因,仅展示原文概要,查看原文内容请购买。
柠檬酸裂解酶ELISA试剂盒使用说明书
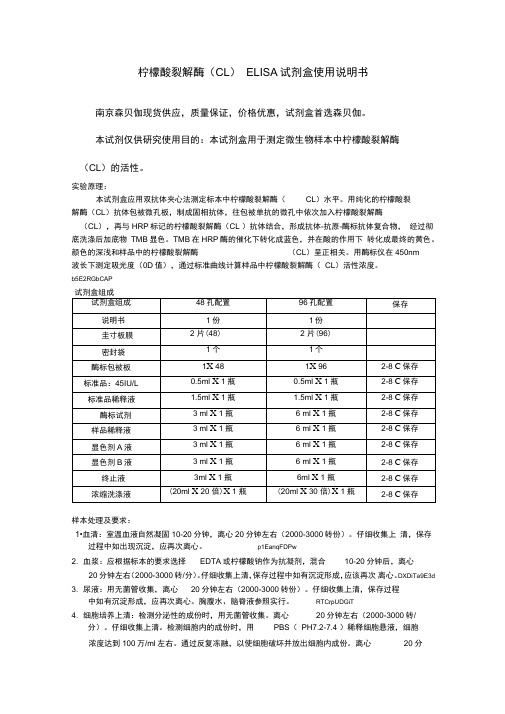
柠檬酸裂解酶(CL)ELISA试剂盒使用说明书南京森贝伽现货供应,质量保证,价格优惠,试剂盒首选森贝伽。
本试剂仅供研究使用目的:本试剂盒用于测定微生物样本中柠檬酸裂解酶(CL)的活性。
实验原理:本试剂盒应用双抗体夹心法测定标本中柠檬酸裂解酶(CL)水平。
用纯化的柠檬酸裂解酶(CL)抗体包被微孔板,制成固相抗体,往包被单抗的微孔中依次加入柠檬酸裂解酶(CL),再与HRP标记的柠檬酸裂解酶(CL )抗体结合,形成抗体-抗原-酶标抗体复合物,经过彻底洗涤后加底物TMB显色。
TMB在HRP酶的催化下转化成蓝色,并在酸的作用下转化成最终的黄色。
颜色的深浅和样品中的柠檬酸裂解酶(CL)呈正相关。
用酶标仪在450nm波长下测定吸光度(0D值),通过标准曲线计算样品中柠檬酸裂解酶(CL)活性浓度。
b5E2RGbCAP样本处理及要求:1•血清:室温血液自然凝固10-20分钟,离心20分钟左右(2000-3000转份)。
仔细收集上清,保存过程中如出现沉淀,应再次离心。
p1EanqFDPw2. 血浆:应根据标本的要求选择EDTA或柠檬酸钠作为抗凝剂,混合10-20分钟后,离心20分钟左右(2000-3000转/分)。
仔细收集上清,保存过程中如有沉淀形成,应该再次离心。
DXDiTa9E3d 3. 尿液:用无菌管收集,离心20分钟左右(2000-3000转份)。
仔细收集上清,保存过程中如有沉淀形成,应再次离心。
胸腹水、脑脊液参照实行。
RTCrpUDGiT4. 细胞培养上清:检测分泌性的成份时,用无菌管收集。
离心20分钟左右(2000-3000转/分)。
仔细收集上清。
检测细胞内的成份时,用PBS(PH7.2-7.4 )稀释细胞悬液,细胞浓度达到100万/ml左右。
通过反复冻融,以使细胞破坏并放出细胞内成份。
离心20分钟左右(2000-3000 转/分)。
仔细收集上清。
保存过程中如有沉淀形成,应再次离心。
5PCzVD7HxA 5. 组织标本:切割标本后,称取重量。
- 1、下载文档前请自行甄别文档内容的完整性,平台不提供额外的编辑、内容补充、找答案等附加服务。
- 2、"仅部分预览"的文档,不可在线预览部分如存在完整性等问题,可反馈申请退款(可完整预览的文档不适用该条件!)。
- 3、如文档侵犯您的权益,请联系客服反馈,我们会尽快为您处理(人工客服工作时间:9:00-18:30)。
=====================================================================Acq. Operator : Li Shan(LCMS-02) Seq. Line : 26
Acq. Instrument : HY-LCMS-02 Location : P1-C-03Injection Date : 6/19/2015 11:29:40 AM Inj : 1
Inj Volume : 3.000 µl
Acq. Method : D:\AGLIENT 1260\DATA\20150619\20150619 2015-06-19 09-39-29\100-1000MS+3MIN- 1.5_(0.02%FA).M
Last changed : 6/19/2015 9:39:29 AM by Li Shan(LCMS-02)
Analysis Method : D:\AGLIENT 1260\METHOD\50-1000MS+3MIN_1.5(0.02%FA)(50-100A).M Last changed : 6/19/2015 12:16:55 PM by Li Shan(LCMS-02) (modified after loading)
Method Info : Postive,MS:50-1000,Column ID:A-RP-132,40℃
Catalog No : HY-16725 Batch#16649 A-RP-132
Additional Info : Peak(s) manually integrated
min
0.5
1
1.5
2 2.53mAU 0
200400
60080010001200 DAD1 B, Sig=214,4 Ref=off (D:\AGLIENT...60\DATA\20150619\20150619 2015-06-19 09-39-29\BIZ2015-619-DJL8.D)
1.600
1.993
2.140 2.257
2.380
2.487
2.800
===================================================================== Area Percent Report =====================================================================
Sorted By : Signal Multiplier : 1.0000Dilution : 1.0000
Do not use Multiplier & Dilution Factor with ISTDs
Signal 1: DAD1 B, Sig=214,4 Ref=off
Peak RetTime Type Width Area Height Area # [min] [min] [mAU*s] [mAU] %
----|-------|----|-------|----------|----------|--------| 1 1.600 MM 0.0433 6.26722e-1 2.41361e-1 0.0175 2 1.993 MM 0.1167 5.60997 8.01149e-1 0.1565 3 2.140 MF 0.0451 3562.13013 1317.69568 99.3744 4 2.257 FM 0.0379 10.25833 4.50551 0.2862 5 2.380 MM 0.0502 2.23005 7.39947e-1 0.0622 6 2.487 MM 0.0467 2.10026 7.49969e-1 0.0586 7 2.800 MM 0.0535 1.59928 4.97823e-1 0.0446
Totals : 3584.55474 1325.23143
===================================================================== *** End of Report ***
=====================================================================Acq. Operator : Li Shan(LCMS-02) Seq. Line : 26
Acq. Instrument : HY-LCMS-02 Location : P1-C-03Injection Date : 6/19/2015 11:29:40 AM Inj : 1
Inj Volume : 3.000 µl
Acq. Method : D:\AGLIENT 1260\DATA\20150619\20150619 2015-06-19 09-39-29\100-1000MS+3MIN- 1.5_(0.02%FA).M
Last changed : 6/19/2015 9:39:29 AM by Li Shan(LCMS-02)
Analysis Method : D:\AGLIENT 1260\METHOD\50-1000MS+3MIN_1.5(0.02%FA)(50-100A).M Last changed : 6/19/2015 12:18:02 PM by Li Shan(LCMS-02) (modified after loading)
Method Info : Postive,MS:50-1000,Column ID:A-RP-132,40℃
Catalog No : HY-16725 Batch#16649 A-RP-132
Additional Info : Peak(s) manually integrated
min
0.5
1
1.5
2
2.530
200000
400000600000800000 MSD1 TIC, MS File (D:\AGLIENT 1260\DATA\20150619\20150619 2015-06-19 09-39-29\BIZ2015-619-DJL8.D) ES-API, Pos, Sca
2.149
MS Signal: MSD1 TIC, MS File, ES-API, Pos, Scan, Frag: 50 Spectra averaged over upper half of peaks. Noise Cutoff: 1000 counts.
Reportable Ion Abundance: > 10%.
Retention Mol. Weight Time (MS) MS Area or Ion
2.149 5091700 412.10 I 411.10 I 206.10 I
m/z
100
200
300
400
500
600
700
800
20406080100*MSD1 SPC, time=2.127:2.181 of D:\AGLIENT 1260\DATA\20150619\20150619 2015-06-19 09-39-29\BIZ2015-619-DJL8.D ES-API,Max: 515536
206.1
412.1
411.1
*** End of Report ***。
