FDA术语词汇全
FDA专业词汇整理

melting points 熔点 lab scale 实验室规模 API 主药 XRPD X 光粉末衍射技术 Utilize 利用 phase transition 相变 morphology 形态 Approximately 大约的 Biopharmaceutics Classification System (BCS). 生物药剂学分类 系统 Hygroscopicity 引湿性 Density 密度 Bulk Density 松密度 Tapped Density 堆密度 Flow property 流动性 Amorphous 无定形的 Stress testing 影响因素实验 degradation pathway 降解途径 accelerated testing 加速实验 oxidation 氧化 photolysis 光解 reasonable 合理的 Degradation peaks 降解峰 peak purity 峰纯度 Thermostability 热稳定性 Photolytic Stability 光稳定性 Oxidative Stability 氧稳定性 Partition coefficient 分配系数 Caco-2 permeability: Caco-2 细胞
Expiry date 过期时间 Average weight 平均片重 Related Compound 有关物质 pharmaceutical development 制剂 开发 BCS Class 生物药剂学分类 aqueous solubility 水溶性 physiological pH range 生理 pH 范围 buffer solution 缓冲溶液 mean absolute bioavailability 平 均绝对生物利用度 terminal elimination half-life 最终消除的半衰期 Properties 特性,性质 safety and efficacy 安全性及有效 性 orally disintegrating tablet 口腔崩 解片 scored 有刻痕的 Route of administration 给药途径 Bioequivalence 生物等效性 shelf-life 货架期 Microbial Limits 微生物限度 pharmacopoeia criteria 药典标准 crystals 结晶粉末 Salt form 盐型 crystallization conditions 结晶条 件 solvents 溶剂
FDA药品审评相关术语

FDA药品审评相关术语新药申报的简化程序(ANDA)为已被批准药物的相似药品设立的简化申报办法与已知安全有效产品相比,ANDA药是有相同或非常近似的有效成分、剂型、效力、给药途径使用方法和标签的药品。
与NDA一样,ANDA包括药品化学成分和生产质控的一切必要资料;但不需要动物试验与临床试验的资料、然而在ANDA中必须有该相似药品与已被批准药物生物等效性的证据(见‘生物等效性’)。
加速批准:一种高度特殊的机制,目的是加速治疗严重威胁生命疾病或有优于已知疗法明显苗头的药品的审批。
它综台了有关各方因素,在维护公众健康和法规完整性的条件下加速了审批与批准、当审评可以可靠地使用‘替代指标’(参见“替代指标”),或当FDA认为一种有效药物在某种限定条件下可以安全使用时,就可以来阐这一机制。
替代指标往往比象生存率这样的终点指标来得快、在加速审批中,FDA根据申报者对药品临床利益的研究情况来批准药物。
启动函:FDA给NDA申报者的官方通信,即有关FDA决定的信函、批准函允许有关产品进入市场;可审批函指出在正式批准前有少量问题有待解决;而不予批准函则提出药物有重大缺欠而无法予以批准。
除非加以进一步改进。
顾问委员会:由外部专家组成的委员会,其功能是定期对FDA审评与管理药品的安全性与有效性问题提出意贝、FDA没有义务一定要接受该委员会的建议,但通常是采纳其建议的。
对新药申请的修正:对尚未批准的NDA或附件进行增补或改变的申报。
生物利用度:药物被吸收或被机体治疗部位利用的速率与程度。
生物等效性:比较非注册药与商标注册药时的科学基础、当两种产品在相同条件下以同一剂量给予时,两者的生物利用度没有明显差异,则认为具有生物等效性。
然而某些药物往往吸收速率不同。
如果某产品的吸收速率与另一产品不同,并在产品标签中加以说明,而巨这种区别不影响药品的安全性与有效性以及其他医学作用,则FDA可以认为两产品仍有生物等效性。
临床研究:这种人体上的研究用于区分药物作用和其他因素的影响例如病程发展的影响,安慰剂(外观与药物一样的无效物质)的影响等。
FDA常用英文词汇翻译

FDA常用英文词汇翻译FDA(FOOD AND DRUG ADMINISTRA TION):(美国)食品药品管理局IND(INVESTIGA TIONAL NEW DRUG):临床研究申请(指申报阶段,相对于NDA而言);研究中的新药(指新药开发阶段,相对于新药而言,即临床前研究结束)NDA(NEW DRUG APPLICATION):新药申请ANDA(ABBREVIATED NEW DRUG APPLICATION):简化新药申请EP诉(EXPORT APPLICATION):出口药申请(申请出口不被批准在美国销售的药品) TREATMENT IND:研究中的新药用于治疗ABBREVIATED(NEW)DRUG:简化申请的新药DMF(DRUG MASTER FILE):药物主文件(持有者为谨慎起见而准备的保密资料,可以包括一个或多个人用药物在制备、加工、包装和贮存过程中所涉及的设备、生产过程或物品。
只有在DMF持有者或授权代表以授权书的形式授权给FDA,FDA在审查IND、NDA、ANDA时才能参考其内容)HOLDER:DMF持有者CFR(CODE OF FEDERAL REGULATION):(美国)联邦法规PANEL:专家小组BA TCH PRODUCTION:批量生产;分批生产BA TCH PRODUCTION RECORDS:生产批号记录POST-OR PRE- MARKET SURVEILLANCE:销售前或销售后监督INFORMED CONSENT:知情同意(患者对治疗或受试者对医疗试验了解后表示同意接受治疗或试验)PRE<I>script</I>ION DRUG:处方药OTC DRUG(OVER—THE—COUNTER DRUG):非处方药U.S.PUBLIC HEALTH SERVICE:美国卫生福利部NIH(NATIONAL INSTITUTE OF HEALTH):(美国)全国卫生研究所CLINICAL TRIAL:临床试验ANIMAL TRIAL:动物试验ACCELERATED APPROV AL:加速批准STANDARD DRUG:标准药物INVESTIGATOR:研究人员;调研人员PREPARING AND SUBMITTING:起草和申报SUBMISSION:申报;递交BENIFIT(S):受益RISK(S):受害DRUG PRODUCT:药物产品DRUG SUBSTANCE:原料药ESTABLISHED NAME:确定的名称GENERIC NAME:非专利名称PROPRIETARY NAME:专有名称;INN(INTERNATIONAL NONPROPRIETARY NAME):国际非专有名称NARRA TIVE SUMMARY记叙体概要ADVERSE EFFECT:副作用ADVERSE REACTION:不良反应PROTOCOL:方案ARCHIV AL COPY:存档用副本REVIEW COPY:审查用副本OFFICIAL COMPENDIUM:法定药典(主要指USP、NF).USP(THE UNITED STATES PHARMACOPEIA):美国药典(现已和NF合并一起出版) NF(NATIONAL FORMULARY):(美国)国家药品集OFFICIAL=PHARMACOPEIAL= COMPENDIAL:药典的;法定的;官方的AGENCY:审理部门(指FDA) 医学全在线www.med126.c omSPONSOR:主办者(指负责并着手临床研究者)IDENTITY:真伪;鉴别;特性STRENGTH:规格;规格含量(每一剂量单位所含有效成分的量)LABELED AMOUNT:标示量REGULATORY SPECIFICATION:质量管理规格标准(NDA提供)REGULATORY METHODOLOGY:质量管理方法(FDA用于考核原料药或药物产品是否符合批准了的质量管理规格标准的整套步骤)REGULATORY METHODS V ALIDATION:管理用分析方法的验证(FDA对NDA提供的方法进行验证)Dietary supplement:食用补充品PRE<I>script</I>ION DRUG:处方药OTC DRUG(OVER—THE—COUNTER DRUG):非处方药U.S.PUBLIC HEALTH SERVICE:美国卫生福利部NIH(NATIONAL INSTITUTE OF HEALTH):(美国)全国卫生研究所CLINICAL TRIAL:临床试验ANIMAL TRIAL:动物试验ACCELERATED APPROV AL:加速批准STANDARD DRUG:标准药物INVESTIGATOR:研究人员;调研人员PREPARING AND SUBMITTING:起草和申报SUBMISSION:申报;递交BENIFIT(S):受益医.学全,在.线,提供w w w.m e d126.c omRISK(S):受害DRUG PRODUCT:药物产品DRUG SUBSTANCE:原料药ESTABLISHED NAME:确定的名称GENERIC NAME:非专利名称PROPRIETARY NAME:专有名称;INN(INTERNATIONAL NONPROPRIETARY NAME):国际非专有名称NARRA TIVE SUMMARY记叙体概要ADVERSE EFFECT:副作用ADVERSE REACTION:不良反应PROTOCOL:方案ARCHIV AL COPY:存档用副本REVIEW COPY:审查用副本OFFICIAL COMPENDIUM:法定药典(主要指USP、NF).USP(THE UNITED STATES PHARMACOPEIA):美国药典(现已和NF合并一起出版) NF(NATIONAL FORMULARY):(美国)国家药品集OFFICIAL=PHARMACOPEIAL= COMPENDIAL:药典的;法定的;官方的AGENCY:审理部门(指FDA)SPONSOR:主办者(指负责并着手临床研究者)IDENTITY:真伪;鉴别;特性STRENGTH:规格;规格含量(每一剂量单位所含有效成分的量)LABELED AMOUNT:标示量REGULATORY SPECIFICATION:质量管理规格标准(NDA提供)REGULATORY METHODOLOGY:质量管理方法(FDA用于考核原料药或药物产品是否符合批准了的质量管理规格标准的整套步骤)REGULATORY METHODS V ALIDATION:管理用分析方法的验证(FDA对NDA提供的方法进行验证)Dietary supplement:食用补充品。
FDA有关术语中英对照

FDA(FOOD AND DRUG ADMINISTRATION):(美国)食品药品管理局IND(INVESTIGATIONAL NEW DRUG):临床研究申请(指申报阶段,相对于NDA 而言);研究中的新药(指新药开发阶段,相对于新药而言,即临床前研究结束)NDA(NEW DRUG APPLICATION):新药申请ANDA(ABBREVIATED NEW DRUG APPLICATION):简化新药申请EP诉(EXPORT APPLICATION):出口药申请(申请出口不被批准在美国销售的药品)TREATMENT IND:研究中的新药用于治疗ABBREVIATED(NEW)DRUG:简化申请的新药DMF(DRUG MASTER FILE):药物主文件(持有者为谨慎起见而准备的保密资料,可以包括一个或多个人用药物在制备、加工、包装和贮存过程中所涉及的设备、生产过程或物品。
只有在DMF持有者或授权代表以授权书的形式授权给FDA,FDA在审查IND、NDA、ANDA时才能参考其内容)HOLDER:DMF持有者CFR(CODE OF FEDERAL REGULATION):(美国)联邦法规PANEL:专家小组BATCH PRODUCTION:批量生产;分批生产BATCH PRODUCTION RECORDS:生产批号记录POST-OR PRE- MARKET SURVEILLANCE:销售前或销售后监督INFORMED CONSENT:知情同意(患者对治疗或受试者对医疗试验了解后表示同意接受治疗或试验)PRESCRIPTION DRUG:处方药OTC DRUG(OVER—THE—COUNTER DRUG):非处方药FDA(FOOD AND DRUG ADMINISTRATION):(美国)食品药品管理局IND(INVESTIGATIONAL NEW DRUG):临床研究申请(指申报阶段,相对于NDA 而言);研究中的新药(指新药开发阶段,相对于新药而言,即临床前研究结束)NDA(NEW DRUG APPLICATION):新药申请ANDA(ABBREVIATED NEW DRUG APPLICATION):简化新药申请EP诉(EXPORT APPLICATION):出口药申请(申请出口不被批准在美国销售的药品)TREATMENT IND:研究中的新药用于治疗ABBREVIATED(NEW)DRUG:简化申请的新药DMF(DRUG MASTER FILE):药物主文件(持有者为谨慎起见而准备的保密资料,可以包括一个或多个人用药物在制备、加工、包装和贮存过程中所涉及的设备、生产过程或物品。
FDA常用词中英对照

FDA常用词中英对照FDA(food and drug adminisration):(美国)食品药品监督管理局NDA(new drug application):新药申请ANDA(abbreviated new drug application):简化新药申请EP(export application):出口药申请(申请出口不被批准在美国销售的药品)treatment IND:研究中的新药用于治疗abbreviated(new)drug:简化申请的新药DMF(drug master file):药物主文件(持有者为谨慎起见而准备的保密资料,可以包括一个或多个人用药物在制备,加工,包装和贮存过程中所涉及的设备,生产过程或物品.只有在DMF持有者或授权代表以授权书的形式授权给FDA,FDA在审查IND, NDA,ANDA时才能参考其内容) holder:DMF持有者CFR(code of federal regulation):(美国)联邦法规PANEL:专家小组batch production:批量生产;分批生产batch production records:生产批号记录post or pre-market surveillance:销售前或销售后监督informed consent:知情同意(患者对治疗或受试者对医疗试验了解后表示同意接受治疗或试验)prescription drug:处方药OTC drug(over—the—counter drug):非处方药U.S. public health service:美国卫生福利部NIH(national institute of health):(美国)全国卫生研究所animal trail:动物试验accelerated approval:加速批准standard drug:标准药物investigator :研究人员;调研人员preparing and submitting:起草和申报submission:申报;递交benefit(s):受益risk(s):受害drug product:药物产品drug substance:原料药established name:确定的名称generic name:非专利名称proprietary name:专有名称;INN(international nonproprietary name):国际非专有名称narrative summary: 记叙体概要adverse effect:副作用adverse reaction:不良反应protocol:方案archival copy:存档用副本review copy:审查用副本official compendium:法定药典(主要指USP, NF).USP(the united state pharmacopeia):美国药典(现已和NF合并一起出版)NF(national formulary):(美国)国家药品集official=pharmacopeial = compendial:药典的;法定的;官方的agency:审理部门(指FDA)sponsor:主办者(指负责并着手临床研究者)identity:真伪;鉴别;特性strength:规格;规格含量(每一剂量单位所含有效成分的量)labeled amount:标示量regulatory specification:质量管理规格标准(NDA提供)regulatory methodology:质量管理方法(FDA用于考核原料药或药物产品是否符合批准了的质量管理规格标准的整套步骤)regulatory methods validation:管理用分析方法的验证(FDA对NDA提供的方法进行验证) Dietary supplement:食用补充品ICH(International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use)人用药物注册技术要求国际协调会议ICH:Quality-质量Q1A(R2): Stability Testing of New Drug Substances and Products (Second Revision)新原料药和制剂的稳定性试验(第二版)Q1B: Photostability Testing of New Drug Substances and Products新原料药和制剂的光稳定性试验Q1C: Stability Testing for New Dosage Forms新制剂的稳定性试验Q1D: Bracketing and Matrixing Designs for Stability Testing of Drug Substances and Drug Products原料药和制剂稳定性试验的交叉和矩阵设计Q1E: Evaluation of Stability Data对稳定性数据的评估处理Q1F: Stability Data Package for Registration Applications in Climatic Zones III and IV在气候带III和IV,药物注册申请所提供的稳定性数据Q2A: Text on Validation of Analytical Procedures分析程序的验证Q2B: Validation of Analytical Procedures: Methodology分析程序的验证:方法学Q3A(R): Impurities in New Drug Substances (Revised Guideline)新原料药中的杂质(修订版)Q3B(R): Impurities in New Drug Products (Revised Guideline)新制剂中的杂质(修订版)Q3C: Impurities: Guideline for Residual Solvents杂质:残留溶剂指南Q3C(M): Impurities: Guideline for Residual Solvents (Maintenance)杂质:残留溶剂指南(修改内容)Q4: Pharmacopoeias药典Q4A: Pharmacopoeial Harmonisation 药典的协调Q4B: Regulatory Acceptance of Pharmacopoeial Interchangeability药典互替在法规上的可接受性Q5A: Viral Safety Evaluation of Biotechnology Products Derived from Cell Lines of Human or Animal Origin来源于人或者动物细胞系的生物技术产品的病毒安全性评估Q5B: Quality of Biotechnological Products: Analysis of the Expression Construct in Cells Used for Production of r-DNA Derived Protein Products生物技术产品的质量:源于重组DNA的蛋白质产品的生产中所用的细胞中的表达构建分析Q5C: Quality of Biotechnological Products: Stability Testing of Biotechnological/Biological Products生物技术产品的质量:生物技术/生物产品的稳定性试验Q5D: Derivation and Characterisation of Cell Substrates Used for Production of Biotechnological/Biological Products用于生产生物技术/生物产品的细胞底物的起源和特征描述Q5E: Comparability of Biotechnological/Biological Products Subject to Changes in Their Manufacturing Process基于不同生产工艺的生物技术产品/生物产品的可比较性Q6: Specifications for New Drug Substances and Products新原料药和制剂的质量规格Q6A: Specifications: Test Procedures and Acceptance Criteria for New Drug Substances and New Drug Products: Chemical Substances质量规格:新原料药和新制剂的检验程序和可接收标准:化学物质Q6B: Specifications: Test Procedures and Acceptance Criteria for Biotechnological/Biological Products质量规格:生物技术/生物产品的检验程序和可接收标准-- 作者:月萝兰魂-- 发布时间:2006-12-22 13:34:00--Q7: Good Manufacturing Practices for Pharmaceutical Ingredients活性药物成份的GMPQ7A: Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients活性药物成份的GMP指南Q8: Pharmaceutical Development药物研发Q9: Quality Risk Management质量风险管理ICH:Safety-安全S1A: Guideline on the Need for Carcinogenicity Studies of Pharmaceuticals药物致癌性研究需要的指南S1B: Testing for Carcinogenicity of Pharmaceuticals药物致癌性的检验S1C: Dose Selection for Carcinogenicity Studies of Pharmaceuticals药物致癌性研究之剂量选择S1C(R): Addendum: Addition of a Limit Dose and Related Notes附录:极限剂量和有关注释的的补充S2A: Guidance on Specific Aspects of Regulatory Genotoxicity Tests for Pharmaceuticals受法规管辖的药物基因毒性检验的特定方面的指南S2B: Genotoxicity: A Standard Battery for Genotoxicity Testing for Pharmaceuticals基因毒性:药物基因毒性检验的标准S3A: Note for Guidance on Toxicokinetics: The Assessment of Systemic Exposure in Toxicity Studies毒物代谢动力学指南的注释:毒性研究中的全身性暴露量的评估S3B: Pharmacokinetics: Guidance for Repeated Dose Tissue Distribution Studies药物代谢动力学:重复剂量的组织分布研究指南S4: Single Dose Toxicity Tests单剂量毒性检验S4A: Duration of Chronic Toxicity Testing in Animals (Rodent and Non-Rodent Toxicity Testing) 动物体内慢性毒性持续时间的检验(啮齿动物和非啮齿动物毒性检验)S5A: Detection of Toxicity to Reproduction for Medicinal Products药物对生殖发育的毒性的检验S5B(M): Maintenance of the ICH Guideline on Toxicity to Male Fertility: An Addendum to the Guideline on Detection of Toxicity to Reproduction for Medicinal Products对男性生殖能力的毒性的指南的变动:药物对生殖发育的毒性的检验指南增加了一个附录S6: Preclinical Safety Evaluation of Biotechnology-Derived Pharmaceuticals生物技术生产的药物的临床前安全评价S7A: Safety Pharmacology Studies for Human Pharmaceuticals人用药的安全药理学研究S7B: The Nonclinical Evaluation of the Potential for Delayed Ventricular Repolarization(QT Interval Prolongation) By Human Pharmaceuticals药物延迟心室复极化(QT间期)潜在作用的非临床评价S8: Immunotoxicology Studies for Human Pharmaceuticals人用药免疫毒理学研究M3(M): Maintenance of the ICH Guideline on Non-Clinical Safety Studies for the Conduct of Human Clinical Trials for Pharmaceuticals药物的对人临床试验的非临床安全研究指南的变动E-Efficacy(有效)E1: The Extent of Population Exposure to Assess Clinical Safety for Drugs Intended for Long-Term Treatment of Non-Life-Threatening Conditions对用于无生命危险情况下长期治疗的药物进行临床安全评估的族群暴露量范围E2A: Clinical Safety Data Management: Definitions and Standards for Expedited Reporting临床安全数据管理:速报制度的定义和标准E2B(R): Revision of the E2B(M) ICH Guideline on Clinical Safety Data Management Data Elements for Transmission of Individual Case Safety Reports个案安全报告送交的临床安全数据管理的数据要素指南(E2B(M))的修订版E2B (M): Maintenance of the Clinical Safety Data Management including: Data Elements for Transmission of Individual Case Safety Reports临床安全数据管理的变动包括:个案安全报告送交的数据要素E2B(M): Maintenance of the Clinical Safety Data Management including Questions and Answers 临床安全数据管理的变动,包括问答E2C: Clinical Safety Data Management: Periodic Safety Update Reports for Marketed Drugs临床安全数据管理:已上市药品的周期性安全数据更新报告Addendum to E2C: Periodic Safety Update Reports for Marketed DrugsE2C的附录:已上市药品的周期性安全数据更新报告E2D: Post-Approval Safety Data Management: Definitions and Standards for Expedited Reporting 批准后的安全数据管理:速报制度的定义和标准E2E: Pharmacovigilance Planning药物警戒计划E3: Structure and Content of Clinical Study Reports临床研究报告的结构和内容E4: Dose-Response Information to Support Drug Registration支持药品注册的剂量-效应资料E5: Ethnic Factors in the Acceptability of Foreign Clinical Data引入海外临床数据时要考虑的人种因素E6: Good Clinical Practice: Consolidated GuidelineGCP:良好的临床规范:统一的指南E7: Studies in Support of Special Populations: Geriatrics对特定族群的支持的研究:老人病学E8: General Considerations for Clinical Trials对临床试验的总的考虑E9: Statistical Principles for Clinical Trials临床试验的统计原则E10: Choice of Control Group and Related Issues in Clinical Trials临床试验中控制组和有关课题的选择E11: Clinical Investigation of Medicinal Products in the Pediatric Population小儿科药物的临床调查E12A: Principles for Clinical Evaluation of New Antihypertensive Drugs新抗高血压药物的临床评价原则E14: The Clinical Evaluation of QT/QTc Interval Prolongation and Proarrhythmic Potential for Non-Antiarrhythmic Drugs非抗心率失常药物的QT/QTc 间期和致心率失常潜在作用的临床评价Multidisciplinary Guidelines 多学科兼容的指南M1: Medical Terminology医学术语M2: Electronic Standards for Transmission of Regulatory Information (ESTRI)药政信息传递之电子标准M3: Timing of Pre-clinical Studies in Relation to Clinical Trials (See Safety Topics)有关临床试验的临床前研究的时间安排M4: The Common Technical Document (See CTD section for complete Status of the guidelines) 通用技术文件(见有关CTD章节)M5: Data Elements and Standards for Drug Dictionaries药物词典的数据要素和标准临床试验常用的英文缩略语TTP: time-to-progression 疾病进展时间SAE: severity Adverse Event 严重不良事件AE: Adverse Event 不良事件-- 作者:月萝兰魂-- 发布时间:2006-12-22 13:34:00--SOP: Standard Operating Procedure 标准操作规程CRF: Case Report form 病例报告表DLT: 剂量限制毒性MTD: 最大耐受剂量KPS: Karnofsky Performance Status行为状态评分CR: complete response完全缓解PR: partial response部分缓解SD: 病情稳定PD: progressive disease病情进展CTC: 常用药物毒性标准IEC: independent ethics committee 独立伦理委员会IRB : institutional review board 伦理委员会CRA: 临床研究助理CRO: Contract Research Organization 合同研究组织DFS: Disease Free Survival 无病生存期OS: (Overall Survival) 总生存时间IC: Informed consent 知情同意ADR: Adverse Drug Reaction 不良反应GAP:Good Agricultural Practice 中药材种植管理规范GCP:Good Clinical Practice 药物临床试验质量管理规范GLP:Good Laboratory Practice 药品实验室管理规范GMP:Good Manufacturing Practice 药品生产质量管理规范GSP:Good Supply Practice 药品经营质量管理规范GUP:Good Use Practice 药品使用质量管理规范PI :Principal investigator 主要研究者CI: Co-inveatigator 合作研究者SI :Sub-investigator 助理研究者COI :Coordinating investigtor 协调研究者DGMP: 医疗器械生产质量管理规范ICF: Informed consent form 知情同意书RCT : randomized controlled trial, 随机对照试验NRCCT: non-randomized concurrent controlled trial, 非随机同期对照试验EBM: evidence-based medicine 循证医学RCD: randomized cross-over disgn 随机交叉对照试验HCT: historial control trial, 历史对照研究RECIST: Response Evaluation Criteria In Solid Tumors. 实体瘤疗效反应的评价标准QC: Quality Control质量控制UADR: Unexpected Adverse Drug Reaction,非预期药物不良反应-- 作者:月萝兰魂-- 发布时间:2006-12-22 13:34:00--GMP英语PIC/S的全称为:Pharmaceutical Inspection Convention/Pharmaceutical Inspection Cooperation Scheme, PIC/S(制药检查草案), 药品检查协会(PIC/S) ,也有人称PIC/S为医药审查会议/合作计划(PIC/S)PIC的权威翻译:药品生产检查相互承认公约API(Active Pharmaceutical Ingrediet) 原料药又称:活性药物组分AirLock 气闸Authorized Person 授权人Batch/Lot 批次Batch Number/Lot-Number 批号;Batch Numbering System 批次编码系统;Batch Records 批记录;Bulk Product 待包装品;Calibration 校正;Clean area洁净区;Consignmecnt(Delivery)托销药品.ABPI Association of the British Pharmaceutical IndustryADR Adverse Drug ReactionAE Adverse EventAIM Active Ingredient ManufacturerANDA Abbreviated New Drug ApplicationANOV A Analysis of VarianceASM: Active Substance ManufacturerATC Anatomical Therapeutic ChemicalATX Animal Test Exemption CertificateBANBritish Approved NameBIRABritish Institute of Regulatory AffairsBNF British National FormularyBP British PharmacopoeiaC of A Certificate of AnalysisC of S Certificate of SuitabilityCENTRE FOR DRUG EV ALUATION (CDE)Centre for Pharmaceutical Administration (CPA)CMS Concerned Member StateCMS每个成员国COS Certificate of SuitabilityCPMP Committee for Proprietary Medicinal ProductsCRA Clinical Research AssociateCRF Case Report FormCRO Contract Research OrganisationCTA Clinical Trial ApplicationCTC Clinical Trial CertificateCTD Common Technical DocumentCTX Clinical Trials ExemptionDDD Defined Daily DoseDGC Daily Global ComparisonDIA Drug Information AssociationDMF Drug Master FileDrug Registration Branch (DR, Product Evaluation & Registration Division, CPAEDQM (European Directorate for the Quality of Medicines) 欧洲联盟药品质量指导委员会EEA 欧洲经济地区EGMA European Generics Medicine AssociationELA Established Licence ApplicationEMEA European Medicines Evaluation AgencyEMEA (European Agency for the Evaluation of Medicinal Products) 欧洲联盟药品评价机构EP European PharmacopoeiaEPAR European Public Assessment ReportsESRA European Society of Regulatory AffairsEuropean Pharmacopoeia Commission 欧洲药典委员会FDA Food and Drug Administrationfinal evaluation report (FER)free sale certificates (FSCs)Health Sciences Authority (HSA)HSA's Medicines Advisory Committee (MAC)IB Investigators BrochureICH International Conference for HarmonisationIDMC Independent Data-Monitoring CommitteeIEC Independent Ethics CommitteeIND Investigational New DrugINN International Non-proprietary NameInternational Conference on Harmonisation (ICH)IPC In Process ControlIRB Institutional Review BoardLICENCE HOLDERMA Marketing AuthorisationMAA Marketing Authorisation ApplicationMAA上市申请MAH Marketing Authorisation HolderMAH 销售许可持有者MCA Medicines Control AgencyMHW Ministry of Health and Welfare (Japan)MR Mutual RecognitionMRA 美国与欧盟的互认协议MRAs (Mutual Recognition Agreements) 互相认证同意MRFG Mutual Recognition Facilitation GroupMRPMutual Recognition ProcedureNASNew Active SubstanceNCENew Chemical EntityNDANew Drug Applicationnew chemical entities (NCEs)new drug applications (NDAs)NSAID Non Steroidal Anti Inflammatory DrugNTA Notice To ApplicantsOOS Out of SpecificationOTC Over The CounterPAGB Proprietary Association of Great BritainPh Eur European PharmacopoeiaPIL Patient Information LeafletPL Product LicencePOM Prescription Only MedicinePRODUCT OWNERPSU Periodic Safety UpdatesQA Quality AssuranceQC Quality ControlRAJ Regulatory Affairs JournalRMS Reference Member StateRMS相互认可另一成员国RSD Relative Standard DeviationRx Prescription OnlySAE Serious Adverse EventSMF Site Master FileSOP Standard Operating ProcedureSOP (STANDARD OPERA TION PROCEDURE) 标准运作程序SPC/SmPC Summary of Product Characteristicssummary of product characteristics(SPC)Therapeutic Goods Administration (TGA)USP US PharmacopoeiaVMF Veterinary Master FileVPC Veterinary Products CommitteeA.A.A Addition and Amendments 增补和修订AC Air Conditioner 空调器ADR Adverse Drug Reaction 药物不良反应AFDO Association of Food and Drug Officials 食品与药品官员协会(美国) ACC Accept 接受AQL Acceptable Quality Level 合格质量标准ADNA Abbreviated New Drug Application 简化的新药申请BOM Bill of Material 物料清单BPC Bulk pharmaceutical Chemiclls 原料药CBER Center for Biologics Evaluation Research 生物制品评价与研究中心CFU Colony Forming Unet 菌落形成单位DMF Drug Master File 药品管理档案CDER Cemter for Drug Evaluation amd Research 药物评价与研究中心CI Corporate Identity (Image) 企业识别(形象)CIP Cleaning in Place 在线清洗CSI Consumer Safety Insepctor 消费者安全调查员CLP Cleaning Line Procedure 在线清洗程序DAL Defect Action Level 缺陷作用水平DEA Drug Enforcement Adminestration 管制药品管理DS Documentation Systim 文件系统FDA Food and Drug Administration 食品与药品管理局(美国)GA TT General Agreemernt on Tariffs and Trade 关贸总协会GMP Good Manufacturing Practice Gvp 药品生质量管理规范GCP Good Clinical Practice 药品临床实验管理规范GLP Good Laboratory Practice 实验室管理规范GSP Good Supply Practice 药品商业质量规范GRP Gook RaTAIL Practice 药品零业质量管理规范GAP Good Agriculture Practice 药材生产管理规范GVP Gook Validation Prctice 验证管理规范GUP Gook Use Practice 药品重用规范HV AC Heating Ventilation Air Conditioning 空调净化系统ISO Intematonal Organization for Standardization 车际标准化组织MOU Memorandum of Understanding 谅解备忘录PF Porduction File 生产记录用表格OTC Over the Counter (Drug) 非处方药品PLA Product License Application 产品许可申请QA Quality Assurance 质量保证QC Quality Control 质量控制QMP Quality Management Procedure 质量管理程序SDA State Drug Administration 国家药品监督管理局SMP Standard Managmert Procedure 标准管理程序SOP Standard Operating Procedure 标准操作程序TQC Tatal Quality Control 全面质量管理USA Uneted States Pharmacopeia 美国药典-- 作者:月萝兰魂-- 发布时间:2006-12-22 13:35:00--ICH 安全性领域常用专业术语中英文对照表Dead offspring at birth 出生时死亡的子代Degradation 降解Delay of parturition 分娩延迟Deletion 缺失Descriptive statistics 描述性统计Distribution 分布Detection of bacterial mutagen 细菌诱变剂检测Detection of clastogen 染色体断裂剂检测Determination of metabolites 测定代谢产物Development of the offspring 子代发育Developmental toxicity 发育毒性Diminution of the background lawn 背景减少Direct genetic damage 直接遗传损伤DNA adduct DNA加合物DNA damage DNA损伤DNA repair DNA修复DNA strand breaks DNA链断裂Dose escalation 剂量递增Dose dependence 剂量依赖关系Dose level 剂量水平Dose-limiting toxicity 剂量限制性毒性Dose-raging studies 剂量范围研究Dose-relatived mutagenicity 剂量相关性诱变性Dose-related 剂量相关Dose-relatived cytotoxicity 剂量相关性细胞毒性Dose-relatived genotoxic activity 剂量相关性遗传毒性Dose-response curve 剂量-反应曲线Dosing route 给药途径Duration 周期Duration of pregnancy 妊娠周期Eaning 断奶Earlier physical malformation 早期躯体畸形Early embryonic development 早期胚胎发育Early embryonic development to implantation 着床早期的胚胎发育Electro ejaculation 电射精Elimination 清除Embryofetal deaths 胚胎和胎仔死亡Embryo-fetal development 胚胎-胎仔发育Embryo-fetal toxicity 胚胎-胎仔毒性Embryonic death 胚胎死亡Embryonic development 胚胎发育Embryonic period 胚胎期Embryos 胚胎Embryotoxicity 胚胎毒性Enantiomer 对映异构体End of pregnancy 怀孕终止Endocytic 内吞噬(胞饮)Endocytic activity 内吞噬活性Endogenous proteins 内源性蛋白Endogenous components 内源性物质Endogenous gene 内源性基因Endonuclease 核酸内切酶Emdpmiclease release from lysosomes 溶酶体释放核酸内切酶End-point 终点Epididymal sperm maturation 附睾精子成熟性Epitope 抗原决定部位Error prone repair 易错性修复Escalation 递增Escherichia coli strain 大肠杆菌菌株Escherichia coli 大肠杆菌Evaluation of test result 试验结果评价Exaggerated pharmacological response 超常增强的药理作用Excretion 排泄(清除) Exposure assessment 接触剂量评价Exposure period 接解期External metabolizing system 体外代谢系统F1-animals 子一代动物False positive result 假阳性结果Fecundity 多产Feed-back 反馈Fertilisation 受精Fertility 生育力Fertility studies 生育力研究Fetal abnormalities 胎仔异常Fetal and neonatal parameters 胎仔和仔鼠的生长发育参数Fetal development and growth 肿仔发育和生长Fetal period 胎仔期Fetotoxicity 胎仔毒性False negative result 假阴性结果First pass testing 一期试验Fluorescence in situ hybridization(FISH) 原位荧光分子杂交-- 作者:月萝兰魂-- 发布时间:2006-12-22 13:35:00--average deviation 平均差Bbar chart 直条图,条图bias 偏性binomial distribution 二项分布biometrics 生物统计学bivariate normal population 双变量正态总体Ccartogram 统计图case fatality rate(or case mortality) 病死率census 普查chi-sguare(X2) test 卡方检验central tendency 集中趋势class interval 组距classification 分组,分类cluster sampling 整群抽样coefficient of correlation 相关系数coefficient of regression 回归系数coefficient of variability(or coefficieut of variation) 变异系数collection of data 收集资料column 列(栏)combinative table 组合表combined standard deviation 合并标准差combined variance(or poolled variance) 合并方差complete survey 全面调查completely correlation 完全相关completely random design 完全随机设计confidence level 可信水平,置信水平confidence limit 可信限,置信限constituent ratio 构成比,结构相对数continuity 连续性control 对照control group 对照组coordinate 坐标correction for continuity 连续性校正correction for grouping 归组校正correction number 校正数correction value 校正值correlation 相关,联系correlation analysis 相关分析correlation coefficient 相关系数critical value 临界值cumulative frequency 累积频率Ddata 资料degree of dispersion 离散程度degree of freedom 自由度degree of variation 变异度dependent variable 应变量design of experiment 实验设计deviation from the mean 离均差diagnose accordance rate 诊断符合率difference with significance 差别不显著difference with significance 差别显著discrete variable 离散变量dispersion tendency 离中趋势distribution 分布,分配-- 作者:月萝兰魂-- 发布时间:2006-12-22 13:35:00--Eeffective rate 有效率eigenvalue 特征值enumeration data 计数资料equation of linear regression 线性回归方程error 误差error of replication 重复误差estimate value 估计值event 事件experiment design 实验设计experiment error 实验误差experimental group 实验组extreme value 极值Ffatality rate 病死率field survey 现场调查fourfold table 四格表freguency 频数freguency distribution 频数分布GGaussian curve 高斯曲线geometric mean 几何均数grouped data 分组资料Hhistogram 直方图homogeneity of variance 方差齐性homogeneity test of variances 方差齐性检验hypothesis test 假设检验hypothetical universe 假设总体Iincidence rate 发病率incomplete survey 非全面调检indepindent variable 自变量indivedual difference 个体差异infection rate 感染率inferior limit 下限initial data 原始数据inspection of data 检查资料intercept 截距interpolation method 内插法interval estimation 区间估计inverse correlation 负相关Kkurtosis coefficient 峰度系数Llatin sguare design 拉丁方设计least significant difference 最小显著差数least square method 最小平方法,最小乘法leptokurtic distribution 尖峭态分布leptokurtosis 峰态,峭度linear chart 线图linear correlation 直线相关linear regression 直线回归linear regression eguation 直线回归方程link relative 环比logarithmic normal distribution 对数正态分布logarithmic scale 对数尺度lognormal distribution 对数正态分布lower limit 下限Mmatched pair design 配对设计mathematical statistics 数理统计(学) maximum value 极大值mean 均值mean of population 总体均数mean square 均方mean variance 均方,方差measurement data 讲量资料median 中位数medical statistics 医学统计学mesokurtosis 正态峰method of least squares 最小平方法,最小乘法method of grouping 分组法method of percentiles 百分位数法mid-value of class 组中值minimum value 极小值mode 众数moment 动差,矩morbidity 患病率mortality 死亡率Nnatality 出生率natural logarithm 自然对数negative correlation 负相关negative skewness 负偏志no correlation 无相关non-linear correlation 非线性相关non-parametric statistics 非参数统计normal curve 正态曲线normal deviate 正态离差normal distribution 正态分布normal population 正态总体normal probability curve 正态概率曲线normal range 正常范围normal value 正常值normal kurtosis 正态峰normality test 正态性检验nosometry 患病率-- 作者:月萝兰魂-- 发布时间:2006-12-22 13:35:00--Oobserved unit 观察单位observed value 观察值one-sided test 单测检验one-tailed test 单尾检验order statistic 顺序统计量ordinal number 秩号ordinate 纵坐标Ppairing data 配对资料parameter 参数percent 百分率percentage 百分数,百分率percentage bar chart 百分条图percentile 百分位数pie diagram 园图placebo 安慰剂planning of survey 调查计划point estimation 点估计population 总体,人口population mean 总体均数population rate 总体率population variance 总体方差positive correlation 正相关positive skewness 正偏态prevalence rate 患病率probability 概率,机率probability error 偶然误差proportion 比,比率prospective study 前瞻研究prospective survey 前瞻调查public health statistics 卫生统计学Qquality eontrol 质量控制quartile 四分位数Rrandom 随机random digits 随机数字random numbers table 随机数目表random sample 随机样本random sampling 随机抽样random variable 随机变量randomization 随机化randomized blocks 随机区组,随机单位组randomized blocks analysis of variance 随机单位组方差分析randomized blocks design 随机单位组设计randomness 随机性range 极差,全距range of normal values 正常值范围rank 秩,秩次,等级rank correlation 等级相关rank correlation coefficent 等级相关系数rank-sum test 秩和检验ranked data 等级资料rate 率ratio 比recovery rate 治愈率registration 登记regression 回归regression analysis 回归分析regression coefficient 回归系数regression eguation 回归方程relative number 相对数relative ratio 比较相对数relative ratio with fixed base 定基比remainder error 剩余误差replication 重复retrospective survey 回顾调查Ridit analysis 参照单位分析Ridit value 参照单位值Ssample 样本sample average 样本均数sample size 样本含量sampling 抽样sampling error 抽样误差sampling statistics 样本统计量sampling survay 抽样调查scaller diagram 散点图schedule of survey 调查表semi-logarithmic chart 半对数线图semi-measursement data 半计量资料semi-guartile range 四分位数间距sensitivity 灵敏度sex ratio 性比例sign test 符号检验significance 显著性,意义significance level 显著性水平significance test 显著性检验significant difference 差别显著simple random sampling 单纯随机抽样simple table 简单表size of sample 样本含量skewness 偏态slope 斜率sorting data 整理资料sorting table 整理表sources of variation 变异来源square deviation 方差standard deviation(SD) 标准差standard error (SE) 标准误standard error of estimate 标准估计误差standard error of the mean 均数的标准误standardization 标准化standardized rate 标化率standardized normal distribution 标准正态分布statistic 统计量statistics 统计学statistical induction 统计图statistical inference 统计归纳statistical map 统计推断statistical method 统计地图statistical survey 统计方法statistical table 统计调查statistical test 统计表statistical treatment 统计检验stratified sampling 统计处理stochastic variable 分层抽样sum of cross products of 随机变量deviation from mean 离均差积和sum of ranks 秩和sum of sguares of deviation from mean 离均差平方和superior limit 上限survival rate 生存率symmetry 对称(性)systematic error 系统误差systematic sampling 机械抽样-- 作者:月萝兰魂-- 发布时间:2006-12-22 13:35:00--Tt-distribution t分布t-test t检验tabulation method 划记法test of normality 正态性检验test of one-sided 单侧检验test of one-tailed 单尾检验test of significance 显著性检验test of two-sided 双侧检验test of two-tailed 双尾检验theoretical frequency 理论频数theoretical number 理论数treatment 处理treatment factor 处理因素treatment of date 数据处理two-factor analysis of variance 双因素方差分析two-sided test 双侧检验two-tailed test 双尾检验type I error 第一类误差type II error 第二类误差typical survey 典型调查Uu test u检验universe 总体,全域ungrouped data 未分组资料upper limit 上限Vvariable 变量variance 方差,均方variance analysis 方差分析variance ratio 方差比variate 变量variation coefficient 变异系数velocity of development 发展速度velocity of increase 增长速度Wweight 权数weighted mean 加权均数Zzero correlation 零相关-- 作者:月萝兰魂-- 发布时间:2006-12-22 13:36:00--世界500强制药企业名称中英对照排名公司名称中文名称总部收入百万美元77 Pfizer 辉瑞美国45950.092 Johnson & Johnson 强生美国41862.0114 GlaxoSmithKline 葛兰素史克英国35050.9193 Novartis 诺华瑞士24864.0205 Roche Group 罗氏瑞士23212.9222 Merck 默克美国22485.9239 Bristol-Myers Squibb 百时美施贵宝美国20894.0 248 Aventis 安万特法国20162.4254 Abbott Laboratories 雅培美国19680.6269 AstraZeneca 阿斯利康英国18849.0330 Wyeth 惠氏美国15850.6433 Eli Lilly 礼来大药厂美国12582.5100 BASF 巴斯夫德国37757.0125 Dow Chemical 道化学美国32632.0129 Bayer 拜耳德国32331.1365 Akzo Nobel 阿克苏诺贝尔荷兰14770.7。
FDA有关术语

FDA有关术语FDA(FOOD AND DRUG ADMINISTRATION):(美国)食品药品管理局IND(INVESTIGATIONAL NEW DRUG):临床研究申请(指申报阶段,相对于NDA而言);研究中的新药(指新药开发阶段,相对于新药而言,即临床前研究结束)NDA(NEW DRUG APPLICATION):新药申请ANDA(ABBREVIATED NEW DRUG APPLICATION):简化新药申请EP诉(EXPORT APPLICATION):出口药申请(申请出口不被批准在美国销售的药品)TREATMENT IND:研究中的新药用于治疗ABBREVIATED(NEW)DRUG:简化申请的新药DMF(DRUG MASTER FILE):药物主文件(持有者为谨慎起见而准备的保密资料,可以包括一个或多个人用药物在制备、加工、包装和贮存过程中所涉及的设备、生产过程或物品。
只有在DMF持有者或授权代表以授权书的形式授权给FDA,FDA在审查IND、NDA、ANDA时才能参考其内容)HOLDER:DMF持有者CFR(CODE OF FEDERAL REGULATION):(美国)联邦法规PANEL:专家小组BATCH PRODUCTION:批量生产;分批生产BATCH PRODUCTION RECORDS:生产批号记录POST-OR PRE- MARKET SURVEILLANCE:销售前或销售后监督INFORMED CONSENT:知情同意(患者对治疗或受试者对医疗试验了解后表示同意接受治疗或试验)PRESCRIPTION DRUG:处方药OTC DRUG(OVER—THE—COUNTER DRUG):非处方药以上信息来自中国医药资讯网U.S.PUBLIC HEALTH SERVICE:美国卫生福利部NIH(NATIONAL INSTITUTE OF HEALTH):(美国)全国卫生研究所CLINICAL TRIAL:临床试验ANIMAL TRIAL:动物试验ACCELERATED APPROVAL:加速批准STANDARD DRUG:标准药物INVESTIGATOR:研究人员;调研人员PREPARING AND SUBMITTING:起草和申报SUBMISSION:申报;递交BENIFIT(S):受益RISK(S):受害DRUG PRODUCT:药物产品DRUG SUBSTANCE:原料药ESTABLISHED NAME:确定的名称GENERIC NAME:非专利名称PROPRIETARY NAME:专有名称;INN(INTERNATIONAL NONPROPRIETARY NAME):国际非专有名称NARRATIVE SUMMARY记叙体概要ADVERSE EFFECT:副作用ADVERSE REACTION:不良反应PROTOCOL:方案ARCHIVAL COPY:存档用副本REVIEW COPY:审查用副本OFFICIAL COMPENDIUM:法定药典(主要指USP、NF)USP(THE UNITED STATES PHARMACOPEIA):美国药典(现已和NF合并一起出版)NF(NATIONAL FORMULARY):(美国)国家药品集OFFICIAL=PHARMACOPEIAL= COMPENDIAL:药典的;法定的;官方的AGENCY:审理部门(指FDA)SPONSOR:主办者(指负责并着手临床研究者)IDENTITY:真伪;鉴别;特性STRENGTH:规格;规格含量(每一剂量单位所含有效成分的量)LABELED AMOUNT:标示量REGULATORY SPECIFICATION:质量管理规格标准(NDA提供)REGULATORY METHODOLOGY:质量管理方法(FDA用于考核原料药或药物产品是否符合批准了的质量管理规格标准的整套步骤)REGULATORY METHODS VALIDATION:管理用分析方法的验证(FDA对NDA提供的方法进行验证)Dietary supplement:食用补充品种。
FDA临床试验常见词汇中译文对照.doc

FDA 临床试验常见词汇中译文对照Aaction letter 决定通知active comparator 活性药物对照组active control = AC 阳性对照,活性对照active ingredient 有效成分Active Substance Master File (ASMF) 欧洲药物主文件acute myocardial infarction 急性心肌梗死acute tibial fractures 急性胫骨骨折adalimumab (Humira) 阿达木单抗adaptive design 自适应设计adaptive randomization 自适应随机ADE = adverse drug event 药物不良事件Adenoviral Vectors 腺病毒载体adequate and well-controlled studies 充分严格的对照研究ADHD = Attention-deficit hyperactivity disorder注意力缺陷多动障碍; 注意力不足过动症; 多动症adhesion barrier product 防黏著产品adjuvant 助剂 ; 佐剂 auxiliary;adjuvant therapy 佐药疗法,辅助疗法ADL = activities of daily living 日常生活活动能力ADME = absorption, distribution, metabolism, and excretion(药物 )吸收、分配、代谢和排除ADR = adverse drug reaction 药物不良反应adrenal cortex 肾上腺皮质adrenal cortical hormone 肾上腺皮质激素adrenal gland 肾上腺adrenaline 肾上腺素adulterated devices 掺假器械adverse drug reaction = ADR 药物不良反应adverse effect 副作用adverse event = AE 不良事件adverse medical events 不良医学事件adverse reaction (adverse event) 药物不良反应advisory 提醒advocacy and support groups 倡导和支持团体AE = adverse event 不良事件AERS = Adverse Event Reporting System 不良事件报告系统BBIMO Bioresearch Monitoring Program 生物研究监测bioavailability (F) 生物利用度biochemical drugs 生化药品biocides 生物杀灭剂 ; 杀生物剂biocompatibility 生物相容性biodegradable 生物分解bio-engineered, transgenic food 转基因食物bioequivalence; bioequivalent 生物等效应biofilm 细菌薄膜 , 生物膜biologic 生物制品biological response modifiers BRM 生物应答调节剂biological therapeutic agents 生物治疗药剂biomarker 生物标志物biometrics 生物统计 ; 生物识别技术bion stimulator 生物体刺激器bionic knee 仿生膝关节biopharma: biopharmaceutical products 生物药物产品bipolar 双相燥郁症birth defect 出生缺陷 , 新生儿缺陷 , 先天缺陷BLA = biologic license application 生物制品许可申请blank control 空白对照blend uniformity analysis 混合均匀度分析blind 盲法blind codes 编制盲底blind review 盲态审核blinding method 盲法blinding/ masking 盲法,设盲blister packaging 泡罩包装 ; 水泡眼block 分段 ;层block size 每段的长度blocked randomization 区组随机Ccase history 病历case record form = CRF 病例报告表 / 病例记录表case report form 病例报告表cash curve 现金曲线cash trap 现金陷阱 ; 现金套牢categorical variable 分类变量CLIA Clinical Laboratory Improvement Amendments临床实验室改进修订案clinical (human) data 临床数据clinical endpoint 临床终点clinical equivalence 临床等效应clinical hold 临床试验暂停通知clinical investigator 临床研究者Clinical Pharmacists 临床药师Clinical Research Coordinator = CRC 临床研究协调者clinical study 临床研究Clinical Study Application = CSA 临床研究申请clinical study reportclinical trialclinical trial application = CTAclinical trial exemption = CTXclinical trial protocol = CTPClinical Trial Report = CTRclinically significant resultscohortcohort studiesco-investigator = CIcomparisonCompassionate Usecompetitive labelingComplementary And Alternative Therapy Complete responsecompliancecomposite variableCompression Testcomputer-assisted trial design= CATD Con Meds = concomitant medications confidence intervalconfidence levelConfidentiality Regarding Trial Participants controlcontrol groupcontrolled clinical trialsControlled TrialsCritical PathCRM = continual reassessmentmethod crossover designcross-over studycrossover therapyCRF = case report formdosage formdosage regimendose-ranging studydose-reaction relationdose-related adverse reactionsdouble blindingdouble dummydouble dummydouble dummy techniquedouble-blind studyDouble-Masked Study 临床试验的总结报告临床试验临床试验申请临床试验免责临床试验方案临床试验报告有临床意义队列队列研究合作研究者对照体恤使用优越标签补充性和非传统治疗完全有效遵守;对遵守法规情况的监管复合变量压缩试验计算机辅助试验设计联合用药可信区间置信水平为试验参与者保密对照对照组临床对照实验对照试验关键路径连续重新评估方法交叉设计交叉研究交叉治疗病例报告表剂型给药方案剂量范围研究剂量-反应关系剂量相关的不良反应双盲双模拟双模拟双盲双模拟技术双盲研究双盲研究DRGs = Diagnosis Related Group System drop outdrop testdrug eluting coronary stentsdrug productdrug substancedrug-drug interaction56drug-food interaction 疾病诊断相关分组脱落落震试验 ;跌落试验药物洗脱支架药物产品原料药药物 -药物相互作用药物 -食物的相互作用effectivenessefficacyefficacy (Of a drug or treatment) 疗效有效性测定药效;药品疗效EEMEA = European Medical Evaluation Agency; European Agency for the Evaluation of Medicinal Products; European Medicines Agency 药物评价机构emergency envelope 应急信件 Empiric Bayesian MultipleGamma-Poisson Shrinker; 欧洲医药品管理局empiricalendpointendpoint criteriafactorial designfactorial trialfailureFair Packaging and Labeling Act (1966) False Claims Actfalse therapeutic claimsfull analysis setfull factorial design 经验性贝氏法(伽玛泊松分布缩检法)经验性终点终点指标析因设计析因试验无效,失败公平包装和标签法防制不实请求法错误的疗效声明全分析集全因子试验法Iinclusion criteria入选标准inclusion/exclusion criteria入选/排除标准incremental exposure食品中递增摄入量incubation period/latency period潜伏期IND = Investigational New Drug临床研究新药INDA = investigational new drug application NDA前申报阶段indemnity insurance赔偿保险Independent Data Monitoring = IDM独立数据监察Independent Data Monitoring Committee = IDMC独立数据监察委员会independent ethics committee = IEC indicationsinvestigational new drug= IND investigational product 独立伦理委员会适应症临床研究新药试验药物investigatorinvestigator's brochure = IB 调研人员研究者手册Mmaskedmean absorption time = MAT mean disintegration time = MDIT 设盲(药物在体内的(药物在体内的)平均吸收时间)平均崩解时间Mean Dissolution Time = MDT Mean Residence Time = MRT (药物在体内的(药物在体内的)平均释放时间)平均滞留时间medical governanceMedicaremedication guides (for patients) Medicines Control Agency = MCA MisbrandingMiscodingmissing valuemixed effect modelMLD = minimal lethal doseMoA = Mechanism of Actionmonitormonitoring planmonitoring reportMR = moderate responseMRA = Agreement on Mutual Recognition MTD = maximal tolerance dose multicenter trialmulti-drug resistancemultiple arm trialsmutual recognition procedure (EU) 医药治理老年医疗保险制度;联邦老年医保用药指南英国药品监督局错误标签 ; 冒牌编码错误缺失值混合效应模式最小致死剂量作用机制 ;作用机理监查员监查计划监查报告好转相互承认协定最大耐受剂量多中心试验多药物抗药性多治疗组的试验相互承认程序OOS = Overall survival 总生存率Pparallel group designparameter estimationparametric releaseparametric statisticspatient filepatient global; pt globalpatient historyper protocol ( PP) analysisPFS = progression-free survival PGE = patient global evaluation PHA = preliminary hazards analysis pharmaceutical equivalence pharmaceutics 平行组设计参数估计参数放行参数统计方法病人档案病人总体评价病历符合方案分析无疾病进展存活率病人总体评价预先危险分析药剂等效性药剂学pharmacodynamics=PD 药物效应动力学 ; 简称药效学pharmacoepidemiology 药物流行病学pharmacokinetics = PK 药代动力学 ; 简称药动学pharmacology 药理学Pharmacovigilance105 药物警戒pharmacy 配药学PharMetrics claims database 索赔数据库PhRMA = Pharmaceutical Research and Manufacturers of America美国药物研究与生产商协会PIC=Pharmaceutical Inspection Convention 药品检查协定PIC/S Pharmaceutical Inspection Cooperation Scheme 药物检查合作计划pipeline assets 开发中产品PK = pharmacokinetics 药物代谢动力学 ; 药动学,药代动力学placebo 安慰剂placebo control 安慰剂对照placebo controlled study 安慰剂对照研究placebo effect 安慰剂效应PMA = premarket approval 上市前许可 ; 销售前批准PMCs = post marketing commitments 承诺药品上市后的继续研究PMDRA = Post Marketing Drug Risk Assessment 上市后药品风险评估 (办公室 ) PMHx = Past Medical History 既往病史PMN = Premarket Notification 销售前通知PMS = Premenstrual syndrome 经前综合症POC (Proof-of-concept) Clinical Trials 概念证明POC = point-of-care testing 床旁分析polytomies 多分类pooled analysis = PA 荟萃分析postmarket surveillance 上市后监督post-marketing surveillance; postmarket safety surveillance 销售(上市)后监督power 把握度 ; 检验效能Pp = Process Performance 工序绩效Ppk = Process Performance Index 工序绩效指数precautions 慎用;注意事项precision 精密度preclinical (animal) data 临床前 (动物实验 )数据preclinical study 临床前研究predicate device = legally marketed device that is not subject to Premarket Approval (PMA)和已合法在市场上销售的且不需要做PMA“销售前批准”的Pre-market Approval (Application) = PMA 上市前许可(申请)premarket notification 上市前通知pre-marketing surveillance 销售(上市)前监督preparing and submitting 起草和申报prescription drug 处方药preservation 保藏prevalenceprevention trialsprimary (coronary) eventprimary endpointprimary mode of action = PMOA primary variableprincipal investigator = PIPrinciples of Qualificationprocess controlsprocess validationproduct codesproduct differentiationproduct license =PLproduct life cycle (PLC)prognosisprogression-free survival = PFS progressive Disease PDproof of principle studypropensity scoreprotocolprotocol amendmentprototype designprotozoaproven acceptable Range = PAR PTC = Product Technical ComplaintsQqualification system for licensed pharmacist qualified health claims Qualified Person = QPquality assurance = QAquality assurance unit = QAUquality control = QC quality management systemsquality of life trials or supportive care trials quality risk management = QRM quantitative risk assessmentRrandomizationrandomized trialrandomized, double blinded clinical trial range checkrating scaleRCT = randomized clinical trialsRCT = randomized controlled trial 患病率预防试验原位病变主要终点首要作用模式主要变量主要研究者确认(验证)原则工艺控制工艺验证产品的号码产品差异化,产品特色化产品许可证产品生命周期预后无进展生存病情进展原理循证研究倾向性评分试验方案 ; 方案方案补正原型设计原生动物门确定可接受范围药品技术投诉执业药师资格准入制度有保留的健康宣称受权人质量保证质量保证部门质量控制质量管理体系生存质量试验质量风险管理量化风险评估随机化随机化试验随机双盲对照研究范围检查量表随机临床试验随机对照试验RDE: remote data entry 远距数据输入ready-to-eat foods 即食食品reagents 试剂recall 召回 ; 强制回收RECIST = Response Evaluation Criteria in Solid Tumors 实体瘤的疗效评价标准reconditioning 整改 ; 货物重整理;货物重包装recycled plastics 可循环利用塑料制品reference product 参比制剂reference samples 标准样品regulatory methodology 质量管理方法regulatory methods validation 管理用分析方法的验证( FDA 对 NDA 提供的方法进行验证)regulatory specification 质量管理规格标准( NDA 提供)rejection 排异remote monitoring system 远程监测系统 ; 远程监控REMS = Risk Evaluation and Mitigation Strategies 风险评估和减缓战略risk 受害risk assessment (risk analysis + risk evaluation) 风险评估,论证risk classification 风险分类 ;Risk Communications Advisory Committee 风险交流咨询委员会risk evaluation (part of risk assessment) 风险评价risk/ benefit analysis 风险 -获益分析risk-benefit ratio 效益 / 风险比route of administration 给药途径royalties 专利使用费RPN = Risk Priority Number 风险优先指数RR = Response rate 缓解率RSD = (intra-day and inter-day) relative standard deviations (日内和日间 ) 相对标准差Ssafety advisory 安全建议safety evaluation 安全性评价safety evaluators 安全性评估人员safety set 安全性评价的数据集screening trials 筛选性试验SD = standard deviation 标准 (偏 )差SE = substantial equivalence 实质上的等同Seal Strength Test 密封强度试验sequence 试验次序SFDA 129= State Food And Drug Administration 国家食品药品监督管理局SG & A= Sales, General and Administration 销售、管理和一般费用shaft 传动轴SHEA = Society for Healthcare Epidemiology of America 美国医院流行病学学会sheaths 护套shelf life 保存期限 ; 保质期SIC codes = Standard Industrial Classification codes 标准产业分类代码side effects 副作用significance level 显著性水平Significant Risk (SR) 显著的危险性simple randomization 简单随机simulation model 仿真模型single blinding 单盲single-blind study 单盲研究single-masked study 单盲研究site assessment = SA 现场评估site audit 试验机构稽查SMDA = Safe Medical Devices Act of 1990 1990 年安全医疗器械法SMF = Site Master File 生产场所主文件sNDA = supplemental NDA 疗效补充新药上市申请sponsor-investigator = SI 申办研究者spontaneous reports; voluntary reports 药品不良反应自愿报告SPS = Agreement on the Application Of Sanitary and Phytosanitary Measures卫生与植物卫生措施实施协议;简称 SPS协议SSI = surgical site infection 手术部位感染SSOPs = Sanitation Standard Operating Procedures 卫生标准操作程序standard curve 标准曲线standard deviation 标准偏差standard drug 标准药物standard operating procedure = SOP 标准操作规程standard treatment 标准治疗Standards Of Care131 医护标准State Food and Drug Administration = SFDA 国家食品药品监督管理局statistic 统计量statistical analysis plan = SAP 统计分析计划statistical model 统计模型statistical significance 统计学意义statistical tables 统计分析表Statisticians in the Pharmaceutical Industry = PSI 制药业统计学家协会steady-state Area Under the Curve = AUCss稳态药时曲线下面积/ 稳态血药浓度-时间曲线下面积stratified 分层study audit 研究稽查study endpoint 研究终点Study Personnel List = SPL 研究人员名单study site 研究中心study type 研究类型subchronic toxicity studies 亚慢性毒性研究subgroup 亚组sub-investigator 助理研究者subject 受试者subject diary = SD 受试者日记subject enrollment 受试者入选subject enrollment log = SEL 受试者入选表Subject Identification Code List = SIC 受试者识别代码表subject recruitment 受试者招募subject screening log = SSL 受试者筛选表submission 申报;递交subspecialties, internal medicine 亚专科 ,内科substantial equivalence to legally marketed (predicate) device 和已合法在市场上销售的且不需要做 PMA“销售前批准”的相似产品有实质上的等同Ttrain-the-trainer program 培训者培训计划treatment group 试验组treatment IND 治疗性试验性新药申请treatment trials 治疗性试验trial error 试验误差trial initial meeting 试验启动会议trial master file 试验总档案trial objective 试验目的trial site 试验场所TRICARE 军队医疗系统triple blinding 三盲two one-side test 双单侧检验UAE = unexpected adverse event 预料外不良事件unblinding 破盲 ;揭盲under reporting bias 少报偏差unexplained syncope 不明原因晕厥unresectable 不能手术切除variability 变异variable 变量WHO International Collaborating Center for Drug Monitoring(世界卫生组织 )国际药物监测合作中心WHO International Conference of Drug Regulatory Authorities = WHO-ICDRAWHO 国际药品管理当局会议WHO Programme for International Drug Monitoring = PIDMWHO 国际药物监测合作计划。
FDA常用词中英对照

FDA常用词中英对照FDA(food and drug adminisration):(美国)食品药品监督管理局NDA(new drug application):新药申请ANDA(abbreviated new drug application):简化新药申请EP(export application):出口药申请(申请出口不被批准在美国销售的药品)treatment IND:研究中的新药用于治疗abbreviated(new)drug:简化申请的新药DMF(drug master file):药物主文件(持有者为谨慎起见而准备的保密资料,可以包括一个或多个人用药物在制备、加工、包装和贮存过程中所涉及的设备、生产过程或物品。
只有在DMF持有者或授权代表以授权书的形式授权给FDA,FDA在审查IND、 NDA、ANDA时才能参考其内容)holder:DMF持有者CFR(code of federal regulation):(美国)联邦法规PANEL:专家小组batch production:批量生产;分批生产batch production records:生产批号记录post or pre-market surveillance:销售前或销售后监督informed consent:知情同意(患者对治疗或受试者对医疗试验了解后表示同意接受治疗或试验)prescription drug:处方药OTC drug(over—the—counter drug):非处方药U.S. public health service:美国卫生福利部NIH(national institute of health):(美国)全国卫生研究所animal trail:动物试验accelerated approval:加速批准standard drug:标准药物investigator :研究人员;调研人员preparing and submitting:起草和申报submission:申报;递交benefit(s):受益risk(s):受害drug product:药物产品drug substance:原料药established name:确定的名称generic name:非专利名称proprietary name:专有名称;INN(international nonproprietary name):国际非专有名称narrative summary: 记叙体概要adverse effect:副作用adverse reaction:不良反应protocol:方案archival copy:存档用副本review copy:审查用副本official compendium:法定药典(主要指USP、 NF).USP(the united state pharmacopeia):美国药典(现已和NF合并一起出版)NF(national formulary):(美国)国家药品集official=pharmacopeial = compendial:药典的;法定的;官方的agency:审理部门(指FDA)sponsor:主办者(指负责并着手临床研究者)identity:真伪;鉴别;特性strength:规格;规格含量(每一剂量单位所含有效成分的量)labeled amount:标示量regulatory specification:质量管理规格标准(NDA提供)regulatory methodology:质量管理方法(FDA用于考核原料药或药物产品是否符合批准了的质量管理规格标准的整套步骤)regulatory methods validation:管理用分析方法的验证(FDA对NDA提供的方法进行验证)Dietary supplement:食用补充品ICH(International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use)人用药物注册技术要求国际协调会议ICH:Quality-质量Q1A(R2): Stability Testing of New Drug Substances and Products (Second Revision) 新原料药和制剂的稳定性试验(第二版)Q1B: Photostability Testing of New Drug Substances and Products新原料药和制剂的光稳定性试验Q1C: Stability Testing for New Dosage Forms新制剂的稳定性试验Q1D: Bracketing and Matrixing Designs for Stability Testing of Drug Substances and Drug Products原料药和制剂稳定性试验的交叉和矩阵设计Q1E: Evaluation of Stability Data对稳定性数据的评估处理Q1F: Stability Data Package for Registration Applications in Climatic Zones III and IV在气候带III和IV,药物注册申请所提供的稳定性数据Q2A: Text on Validation of Analytical Procedures分析程序的验证Q2B: Validation of Analytical Procedures: Methodology分析程序的验证:方法学Q3A(R): Impurities in New Drug Substances (Revised Guideline)新原料药中的杂质(修订版)Q3B(R): Impurities in New Drug Products (Revised Guideline)新制剂中的杂质(修订版)Q3C: Impurities: Guideline for Residual Solvents杂质:残留溶剂指南Q3C(M): Impurities: Guideline for Residual Solvents (Maintenance)杂质:残留溶剂指南(修改内容)Q4: Pharmacopoeias药典Q4A: Pharmacopoeial Harmonisation 药典的协调Q4B: Regulatory Acceptance of Pharmacopoeial Interchangeability药典互替在法规上的可接受性Q5A: Viral Safety Evaluation of Biotechnology Products Derived from Cell Lines of Human or Animal Origin来源于人或者动物细胞系的生物技术产品的病毒安全性评估Q5B: Quality of Biotechnological Products: Analysis of the Expression Construct in Cells Used for Production of r-DNA Derived Protein Products生物技术产品的质量:源于重组DNA的蛋白质产品的生产中所用的细胞中的表达构建分析Q5C: Quality of Biotechnological Products: Stability Testing of Biotechnological/Biological Products生物技术产品的质量:生物技术/生物产品的稳定性试验Q5D: Derivation and Characterisation of Cell Substrates Used for Production of Biotechnological/Biological Products用于生产生物技术/生物产品的细胞底物的起源和特征描述Q5E: Comparability of Biotechnological/Biological Products Subject to Changes in Their Manufacturing Process基于不同生产工艺的生物技术产品/生物产品的可比较性Q6: Specifications for New Drug Substances and Products新原料药和制剂的质量规格Q6A: Specifications: Test Procedures and Acceptance Criteria for New Drug Substances and New Drug Products: Chemical Substances质量规格:新原料药和新制剂的检验程序和可接收标准:化学物质Q6B: Specifications: Test Procedures and Acceptance Criteria for Biotechnological/Biological Products质量规格:生物技术/生物产品的检验程序和可接收标准Q7: Good Manufacturing Practices for Pharmaceutical Ingredients活性药物成份的GMPQ7A: Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients活性药物成份的GMP指南Q8: Pharmaceutical Development药物研发Q9: Quality Risk Management质量风险管理ICH:Safety-安全S1A: Guideline on the Need for Carcinogenicity Studies of Pharmaceuticals药物致癌性研究需要的指南S1B: Testing for Carcinogenicity of Pharmaceuticals药物致癌性的检验S1C: Dose Selection for Carcinogenicity Studies of Pharmaceuticals药物致癌性研究之剂量选择S1C(R): Addendum: Addition of a Limit Dose and Related Notes附录:极限剂量和有关注释的的补充S2A: Guidance on Specific Aspects of Regulatory Genotoxicity Tests for Pharmaceuticals受法规管辖的药物基因毒性检验的特定方面的指南S2B: Genotoxicity: A Standard Battery for Genotoxicity Testing for Pharmaceuticals 基因毒性:药物基因毒性检验的标准S3A: Note for Guidance on Toxicokinetics: The Assessment of Systemic Exposure in Toxicity Studies毒物代谢动力学指南的注释:毒性研究中的全身性暴露量的评估S3B: Pharmacokinetics: Guidance for Repeated Dose Tissue Distribution Studies 药物代谢动力学:重复剂量的组织分布研究指南S4: Single Dose Toxicity Tests单剂量毒性检验S4A: Duration of Chronic Toxicity Testing in Animals (Rodent and Non-Rodent Toxicity Testing)动物体内慢性毒性持续时间的检验(啮齿动物和非啮齿动物毒性检验)S5A: Detection of Toxicity to Reproduction for Medicinal Products药物对生殖发育的毒性的检验S5B(M): Maintenance of the ICH Guideline on Toxicity to Male Fertility: An Addendum to the Guideline on Detection of Toxicity to Reproduction for Medicinal Products 对男性生殖能力的毒性的指南的变动:药物对生殖发育的毒性的检验指南增加了一个附录S6: Preclinical Safety Evaluation of Biotechnology-Derived Pharmaceuticals生物技术生产的药物的临床前安全评价S7A: Safety Pharmacology Studies for Human Pharmaceuticals人用药的安全药理学研究S7B: The Nonclinical Evaluation of the Potential for Delayed Ventricular Repolarization(QT Interval Prolongation) By Human Pharmaceuticals药物延迟心室复极化(QT间期)潜在作用的非临床评价S8: Immunotoxicology Studies for Human Pharmaceuticals人用药免疫毒理学研究M3(M): Maintenance of the ICH Guideline on Non-Clinical Safety Studies for the Conduct of Human Clinical Trials for Pharmaceuticals药物的对人临床试验的非临床安全研究指南的变动E-Efficacy(有效)E1: The Extent of Population Exposure to Assess Clinical Safety for Drugs Intended for Long-Term Treatment of Non-Life-Threatening Conditions对用于无生命危险情况下长期治疗的药物进行临床安全评估的族群暴露量范围E2A: Clinical Safety Data Management: Definitions and Standards for Expedited Reporting临床安全数据管理:速报制度的定义和标准E2B(R): Revision of the E2B(M) ICH Guideline on Clinical Safety Data Management Data Elements for Transmission of Individual Case Safety Reports个案安全报告送交的临床安全数据管理的数据要素指南(E2B(M))的修订版E2B (M): Maintenance of the Clinical Safety Data Management including: Data Elements for Transmission of Individual Case Safety Reports临床安全数据管理的变动包括:个案安全报告送交的数据要素E2B(M): Maintenance of the Clinical Safety Data Management including Questions and Answers临床安全数据管理的变动,包括问答E2C: Clinical Safety Data Management: Periodic Safety Update Reports for Marketed Drugs临床安全数据管理:已上市药品的周期性安全数据更新报告Addendum to E2C: Periodic Safety Update Reports for Marketed DrugsE2C的附录:已上市药品的周期性安全数据更新报告E2D: Post-Approval Safety Data Management: Definitions and Standards for Expedited Reporting批准后的安全数据管理:速报制度的定义和标准E2E: Pharmacovigilance Planning药物警戒计划E3: Structure and Content of Clinical Study Reports临床研究报告的结构和内容E4: Dose-Response Information to Support Drug Registration支持药品注册的剂量-效应资料E5: Ethnic Factors in the Acceptability of Foreign Clinical Data引入海外临床数据时要考虑的人种因素E6: Good Clinical Practice: Consolidated GuidelineGCP:良好的临床规范:统一的指南E7: Studies in Support of Special Populations: Geriatrics对特定族群的支持的研究:老人病学E8: General Considerations for Clinical Trials对临床试验的总的考虑E9: Statistical Principles for Clinical Trials临床试验的统计原则E10: Choice of Control Group and Related Issues in Clinical Trials临床试验中控制组和有关课题的选择E11: Clinical Investigation of Medicinal Products in the Pediatric Population 小儿科药物的临床调查E12A: Principles for Clinical Evaluation of New Antihypertensive Drugs新抗高血压药物的临床评价原则E14: The Clinical Evaluation of QT/QTc Interval Prolongation and Proarrhythmic Potential for Non-Antiarrhythmic Drugs非抗心率失常药物的QT/QTc 间期和致心率失常潜在作用的临床评价Multidisciplinary Guidelines 多学科兼容的指南M1: Medical Terminology医学术语M2: Electronic Standards for Transmission of Regulatory Information (ESTRI)药政信息传递之电子标准M3: Timing of Pre-clinical Studies in Relation to Clinical Trials (See Safety Topics) 有关临床试验的临床前研究的时间安排M4: The Common Technical Document (See CTD section for complete Status of the guidelines)通用技术文件(见有关CTD章节)M5: Data Elements and Standards for Drug Dictionaries药物词典的数据要素和标准临床试验常用的英文缩略语TTP: time-to-progression 疾病进展时间SAE: severity Adverse Event 严重不良事件AE: Adverse Event 不良事件SOP: Standard Operating Procedure 标准操作规程CRF: Case Report form 病例报告表DLT:剂量限制毒性MTD:最大耐受剂量KPS: Karnofsky Performance Status行为状态评分CR: complete response完全缓解PR: partial response部分缓解SD:病情稳定PD: progressive disease病情进展CTC:常用药物毒性标准IEC: independent ethics committee 独立伦理委员会IRB : institutional review board 伦理委员会CRA:临床研究助理CRO: Contract Research Organization 合同研究组织DFS: Disease Free Survival 无病生存期OS:(Overall Survival)总生存时间IC: Informed consent 知情同意ADR: Adverse Drug Reaction 不良反应GAP:Good Agricultural Practice 中药材种植管理规范GCP:Good Clinical Practice 药物临床试验质量管理规范GLP:Good Laboratory Practice 药品实验室管理规范GMP:Good Manufacturing Practice 药品生产质量管理规范GSP:Good Supply Practice 药品经营质量管理规范GUP:Good Use Practice 药品使用质量管理规范PI :Principal investigator 主要研究者CI: Co-inveatigator 合作研究者SI :Sub-investigator 助理研究者COI :Coordinating investigtor 协调研究者DGMP:医疗器械生产质量管理规范ICF: Informed consent form 知情同意书RCT : randomized controlled trial, 随机对照试验NRCCT: non-randomized concurrent controlled trial, 非随机同期对照试验EBM: evidence-based medicine 循证医学RCD: randomized cross-over design 随机交叉对照试验HCT: historical control trial, 历史对照研究RECIST: Response Evaluation Criteria In Solid Tumors. 实体瘤疗效反应的评价标准QC: Quality Control质量控制UADR: Unexpected Adverse Drug Reaction,非预期药物不良反应。
FDA常用词汇

FDA常用词汇AAccelerated: 加速条件Accuracy: 准确性AIP(Application Integrity Policy): 申请完全政策制裁ANDA(Abbreviation New Drug Application):仿制药或仿制新药申请API(Active Pharmaceutical Ingredient):原料药或活性药。
原简称BPC(Bulk Pharmaceutical Chemical),现常用API。
在药典和一些论文中也常用Drug Substance 或Substance 来代表原料药。
Appearance: 外观Assay:含量Assessment: 药厂自我评估(厂家组织进行的对药厂本身设施有关文件的模拟FDA检查)Audit:审查(预审查,多用于美方原料药用户,在FDA和PAI之前到药厂进行现场预检查)Auditor:审核员Audit trail: 审计追踪BBasket:篮子式Batch production records: 批生产纪录Batch records:批号(量)纪录(即batch production and control records 批量生产和检验纪录)BPC:(Bulk Pharmaceutical chemical)原料药Bracketing stability design:稳定性试验的括号分组设计Blend uniformity: 均匀度CCalibration: 校正或校准(对设备,仪器和衡器等的准确度进行校正)Certification of Areas for GMP compliance: (检验企业实施现行药品生产管路规范部门的标准操作规程)CFR 21 Part 11(Code of federal Registry Part11):联邦法典标题21第11部分CGMP(Current Good Manufacturing Practice):现行药品生产质量管理规范Change control form:也简称CCF 变更控制表Change control:变更控制Cleaning validation:清洁验证CMC(Chemistry and manufacture control)化学和生产的控制Compliance:符合性Compatibility:共存性或兼容性Content uniformity test: 产品含量均匀性测定Container closure system: 容器封闭系统COA(Certification of analysis ):分析合格证书,检验报告或检验报告单DDelayed release:延期放行Design qualification: 设计确认Dissolution test:溶出度测试Deviation records:偏差纪录DMF(drug master file):药物主文件或原料药档案Drug product:成品药Drug substance:原料药EEIR(Establishment inspection report):确定检查报告Electronic signature:电子签名Equipment qualification:对设备,设施,仪器等性能的鉴定Excipients:赋形剂或辅料Excit meeting:现场检查结束会Extended release: 缓慢释放EMEA(The European Medicines Evaluation Agency):欧洲医药评审委员会FFinished pharmaceuticals (drug product, finishes product, finished dosage form):制剂药(成品药)其定义位已原料药为起始物料,加一定的赋形剂,制成具有一定剂型可直接用于治疗的药剂。
FDA英语术语大全

FDA英语术语大全FDA是什么意思,FDA得英文全称是什么?FDA(FOOD AND DRUG ADMINISTRATION):(美国)食品药品管理局IND(INVESTIGATIONAL NEW DRUG):临床研究申请(指申报阶段,相对于NDA而言);研究中的新药(指新药开发阶段,相对于新药而言,即临床前研究结束)NDA(NEW DRUG APPLICATION):新药申请ANDA(ABBREVIATED NEW DRUG APPLICATION):简化新药申请EP诉(EXPORT APPLICATION):出口药申请(申请出口不被批准在美国销售的药品)TREATMENT IND:研究中的新药用于治疗ABBREVIATED(NEW)DRUG:简化申请的新药DMF(DRUG MASTER FILE):药物主文件(持有者为谨慎起见而准备的保密资料,可以包括一个或多个人用药物在制备、加工、包装和贮存过程中所涉及的设备、生产过程或物品。
只有在DMF持有者或授权代表以授权书的形式授权给FDA,FDA在审查IND、NDA、ANDA时才能参考其内容)HOLDER:DMF持有者CFR(CODE OF FEDERAL REGULATION):(美国)联邦法规PANEL:专家小组BATCH PRODUCTION:批量生产;分批生产BATCH PRODUCTION RECORDS:生产批号记录POST-OR PRE- MARKET SURVEILLANCE:销售前或销售后监督INFORMED CONSENT:知情同意(患者对治疗或受试者对医疗试验了解后表示同意接受治疗或试验)PRESCRIPTION DRUG:处方药OTC DRUG(OVER—THE—COUNTER DRUG):非处方药U.S.PUBLIC HEALTH SERVICE:美国卫生福利部NIH(NATIONAL INSTITUTE OF HEALTH):(美国)全国卫生研究所CLINICAL TRIAL:临床试验ANIMAL TRIAL:动物试验ACCELERATED APPROVAL:加速批准STANDARD DRUG:标准药物INVESTIGATOR:研究人员;调研人员PREPARING AND SUBMITTING:起草和申报SUBMISSION:申报;递交BENIFIT(S):受益RISK(S):受害DRUG PRODUCT:药物产品DRUG SUBSTANCE:原料药ESTABLISHED NAME:确定的名称GENERIC NAME:非专利名称PROPRIETARY NAME:专有名称;INN(INTERNATIONAL NONPROPRIETARY NAME):国际非专有名称NARRATIVE SUMMARY记叙体概要ADVERSE EFFECT:副作用ADVERSE REACTION:不良反应PROTOCOL:方案ARCHIVAL COPY:存档用副本REVIEW COPY:审查用副本OFFICIAL COMPENDIUM:法定药典(主要指USP、NF).USP(THE UNITED STATES PHARMACOPEIA):美国药典(现已和NF合并一起出版)NF(NATIONAL FORMULARY):(美国)国家药品集OFFICIAL=PHARMACOPEIAL= COMPENDIAL:药典的;法定的;官方的AGENCY:审理部门(指FDA)SPONSOR:主办者(指负责并着手临床研究者)IDENTITY:真伪;鉴别;特性STRENGTH:规格;规格含量(每一剂量单位所含有效成分的量)LABELED AMOUNT:标示量REGULATORY SPECIFICATION:质量管理规格标准(NDA提供)REGULATORY METHODOLOGY:质量管理方法(FDA用于考核原料药或药物产品是否符合批准了的质量管理规格标准的整套步骤)REGULATORY METHODS VALIDATION:管理用分析方法的验证(FDA对NDA提供的方法进行验证)Dietary supplement:食用补充品。
美国FDA技术术语译名参考

英文全名
Ampule Bottle Clinical units Electrolyte Hydrobromide Inhalation Millicurie Milliliter Square centimeter microgram
中文译名
安瓿 瓶 临床单位 电解质 氢溴酸 吸入 毫居里 毫升 平方厘米 微克
中文译名
大约 颈动脉弯曲 反射作用 相当于 克(张培贤编译)
4
英文名称
英文名称 Spray, metered Suppository Suspension Suspension/drops Suspension, extended release Swab Syrup Tablet Tablet, chewable Tablet, coated particles Tablet, delayed release Tablet, dispersable Tablet, effervescent Tablet, extended release Tampon Tape Tincture Troche/lozenge
缩写
AMPICIL CI DIPROP ER HCL IU MEQ N/A U UMOLAR
英文全名
Ampicillin Curie Dipropionate Extended release Hydrochloride International units Milliequivale nt Not applicable Units Micromolar
美国 FDA 技术术语译名参考
新药研究开发信息
美国 FDA 批准的新药剂型英汉译名对照表
英文名称 Aerosol Aerosol, metered Bar, chemable Capsule Capsule, coated pellets Capsule, delayed rel pellets Capsule, extended release Capsule, microemulsion Concentrate Cream Cream, augmented Dressing Elixir emulsion Enema Fiber, extended release Film, extended release Gas 中文名称 气雾剂 定量气雾剂 龃嚼棒 胶囊 包衣微丸胶囊 延迟释放小丸胶囊 缓释胶囊 微乳胶囊 浓缩剂 霜剂,乳膏 强化乳剂 敷料 酏剂 乳剂 灌肠剂 缓释纤维 缓释膜 气体 英文名称 Gel Granule Granule, delayed release Granule, effervescent Granule, for reconstitution Granule, for reconstitution er Gum, cheming Implant Injectable Injectable, lipid complex Injectable, liposomal Insert Insert, extended release Intrauterine device Jelly Liquid Lotion Lotion, augmented 中文名称 凝胶剂 颗粒剂 延迟释放颗粒剂 泡腾颗粒剂 重整颗粒剂 重整颗粒剂 龃嚼胶 植入剂 注射剂 复合脂质体注射剂 脂质体注射剂 插入剂 缓释插入剂 宫内装置 凝胶,胶冻 液体 洗剂 强化洗剂
FDA和EDQM术语

FDA和EDQM术语:ACCELERATED APPROV AL:加速批准INVESTIGATOR:研究人员;调研人员PREPARING AND SUBMITTING:起草和申报RISK(S):受害DRUG SUBSTANCE:原料药GENERIC NAME:非专利名称PROPRIETARY NAME:专有名称;INN(INTERNATIONAL NONPROPRIETARY NAME):国际非专有名称ARCHIV AL COPY:存档用副本REVIEW COPY:审查用副本OFFICIAL COMPENDIUM:法定药典(主要指USP、NF).OFFICIAL=PHARMACOPEIAL= COMPENDIAL:药典的;法定的;官方的AGENCY:审理部门(指FDA)STRENGTH:规格;规格含量(每一剂量单位所含有效成分的量)REGULATORY SPECIFICATION:质量管理规格标准(NDA提供)REGULATORY METHODOLOGY:质量管理方法REGULATORY METHODS V ALIDATION:管理用分析方法的验证ICH(International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use)人用药物注册技术要求国际协调会议ICH文件分为质量、安全性、有效性和综合学科4类。
质量技术要求文件以Q开头,再以a,b,c,d代表小项:Q1:药品的稳定性Q2:方法学Q3:杂质Q4:药典Q5:生物技术产品质量Q6:标准规格Q7:GMP Q7a:(原料药的优良制造规范指南)药物活性成分的GMP.GMP英语PIC/S的全称为:Pharmaceutical Inspection Convention/Pharmaceutical Inspection Cooperation Scheme, PIC/S(制药检查草案), 药品检查协会(PIC/S) ,也有人称PIC/S为医药审查会议/合作计划(PIC/S)PIC的权威翻译:药品生产检查相互承认公约PANEL:专家小组Air Lock 气闸Authorized Person 授权人Batch Numbering System 批次编码系统;Consignmecnt(Delivery)托销药品。
药品注册英语术语

Glossary(术语):Regulatory Affairs(RA):药政事务drug authority :药政当局investigation and research before project approval :立项前的调研Market Authorization(MA):上市许可post-approval commitment study :上市后的承诺研究post-approval variation application :补充申请life cycle :生命周期Chemistry,Manufacturing,and Controls(CMC):药品的化学、生产和控制cross-functional teams :公司内部各部门look at the big picture :从大局考虑think strategically :进行战略性思考risks and benefits:风险和获益Food and Drug Administration(FDA):美国食品药品监督管理局European Medicines Agency(EMA):欧洲药品管理局International Multi-center Clinical Tria l(IMCT):国际多中心临床试验Bioequivalence study(BE study):生物等效性试验generic drug:仿制药Center for Drug Evaluation(CDE):SFDA下属的药品审评中心Quality by Design(QbD):质量源于设计CMC Pilot Program :FDA 在业内开展的关于 QbD 的试点研究early launch :早日上市design space :设计空间Business Development(BD):业务发展部门Imported Drug License(IDL):进口药品注册证Manufacturing License(ML):生产许可证Clinical Trial Permission(CTP):临床试验批件Active Pharmaceutical Ingredient(API):原料药Orange Book :橙皮书business value :商业价值the Pharmacopoeia o f the Pe o p le's Re pu b lic o f China(ChP):中国药典the United States Pharmacopoeia(USP):美国药典the European Pharmacopoeia(Ph.Eu r.或EP):欧洲药典List o f Essential Drugs(EDL):基本药物目录Reimbursement Drug List(RDL):医保目录)typing e r or :打印错误slip of the pen :笔误Drug Master File(DMF):药物主文件Certificate o f Analysis(Co A):检验报告Marketing(MKT):市场部market share :市场占有率sales volume :销量investigator b ro chure(IB):研究者手册protocol :临床试验方案priority:优先度package insert(PI):说明书labeling :包装标签Patie nt Information Leaflet(PIL):患者使用的说明书Summary of Product Characteristics(SmPC,SPC):产品特性摘要foil:铝箔carton:装药品的小盒shipping label :运输包装标签Medical :医学部provincial drug administration(PDA):省级药监局,包括省、自治区和直辖市药品监督管理部门Institute for Food and Drug Control:药检所National Institute for the Control of Pharmaceutical and Biological Produc ts(NICPBP):中国药品生物制品检定所,简称“中检所”supplementary dossier :补充资料approval letter :注册批件out o f specification(OOS):超出标准、不合格adverse effect(AE):不良事件trial waiver :减免临床试验Clinical :临床部门Commercial :商业部门new chemical entity(NCE):新化学实体ke y opinion leader(KOL):关键意见领袖off-label use:标签外使用patient pool :患者库deadline :最后期限global trial :全球性的临床试验,即国际多中心临床试验regional trial :区域性的临床试验。
FDA临床试验常见词汇中译文对照

FDA临床试验常见词汇中译文对照Aaction letter 决定通知active comparator 活性药物对照组active control = AC 阳性对照,活性对照active ingredient 有效成分Active Substance Master File (ASMF) 欧洲药物主文件acute myocardial infarction 急性心肌梗死acute tibial fractures 急性胫骨骨折adalimumab (Humira) 阿达木单抗adaptive design 自适应设计adaptive randomization 自适应随机ADE = adverse drug event 药物不良事件Adenoviral Vectors 腺病毒载体adequate and well-controlled studies 充分严格的对照研究ADHD = Attention-deficit hyperactivity disorder注意力缺陷多动障碍; 注意力不足过动症; 多动症adhesion barrier product 防黏著产品adjuvant 助剂; 佐剂auxiliary;adjuvant therapy 佐药疗法,辅助疗法ADL = activities of daily living 日常生活活动能力ADME = absorption, distribution, metabolism, and excretion(药物)吸收、分配、代谢和排除ADR = adverse drug reaction 药物不良反应adrenal cortex 肾上腺皮质adrenal cortical hormone 肾上腺皮质激素adrenal gland 肾上腺adrenaline 肾上腺素adulterated devices 掺假器械adverse drug reaction = ADR药物不良反应adverse effect 副作用adverse event = AE 不良事件adverse medical events 不良医学事件adverse reaction (adverse event) 药物不良反应advisory 提醒advocacy and support groups 倡导和支持团体AE = adverse event 不良事件AERS = Adverse Event Reporting System 不良事件报告系统BBIMO Bioresearch Monitoring Program 生物研究监测bioavailability (F) 生物利用度biochemical drugs 生化药品biocides 生物杀灭剂; 杀生物剂biocompatibility 生物相容性biodegradable 生物分解bio-engineered, transgenic food 转基因食物bioequivalence; bioequivalent 生物等效应biofilm 细菌薄膜, 生物膜biologic 生物制品biological response modifiers BRM 生物应答调节剂biological therapeutic agents 生物治疗药剂biomarker 生物标志物biometrics 生物统计; 生物识别技术bion stimulator 生物体刺激器bionic knee 仿生膝关节biopharma: biopharmaceutical products 生物药物产品bipolar 双相燥郁症birth defect 出生缺陷, 新生儿缺陷, 先天缺陷BLA = biologic license application 生物制品许可申请blank control 空白对照blend uniformity analysis 混合均匀度分析blind 盲法blind codes 编制盲底blind review 盲态审核blinding method 盲法blinding/ masking 盲法,设盲blister packaging 泡罩包装; 水泡眼block 分段;层block size 每段的长度blocked randomization 区组随机Ccase history 病历case record form = CRF病例报告表/病例记录表case report form 病例报告表cash curve 现金曲线cash trap 现金陷阱; 现金套牢categorical variable 分类变量CLIA Clinical Laboratory Improvement Amendments临床实验室改进修订案clinical (human) data 临床数据clinical endpoint临床终点clinical equivalence 临床等效应clinical hold 临床试验暂停通知clinical investigator 临床研究者Clinical Pharmacists 临床药师Clinical Research Coordinator = CRC临床研究协调者clinical study 临床研究Clinical Study Application = CSA临床研究申请clinical study report 临床试验的总结报告clinical trial 临床试验clinical trial application = CTA 临床试验申请clinical trial exemption = CTX 临床试验免责clinical trial protocol = CTP 临床试验方案Clinical Trial Report = CTR临床试验报告clinically significant results 有临床意义cohort 队列cohort studies 队列研究co-investigator = CI合作研究者comparison 对照Compassionate Use 体恤使用competitive labeling 优越标签Complementary And Alternative Therapy 补充性和非传统治疗Complete response 完全有效compliance 遵守;对遵守法规情况的监管composite variable 复合变量Compression Test 压缩试验computer-assisted trial design= CATD计算机辅助试验设计Con Meds = concomitant medications 联合用药confidence interval 可信区间confidence level 置信水平Confidentiality Regarding Trial Participants 为试验参与者保密control对照control group 对照组controlled clinical trials 临床对照实验Controlled Trials 对照试验Critical Path 关键路径CRM = continual reassessment method 连续重新评估方法crossover design 交叉设计cross-over study 交叉研究crossover therapy 交叉治疗CRF = case report form 病例报告表dosage form 剂型dosage regimen 给药方案dose-ranging study 剂量范围研究dose-reaction relation 剂量-反应关系dose-related adverse reactions 剂量相关的不良反应double blinding 双盲double dummy 双模拟double dummy 双模拟double dummy technique 双盲双模拟技术double-blind study 双盲研究Double-Masked Study 双盲研究DRGs = Diagnosis Related Group System 疾病诊断相关分组drop out 脱落drop test 落震试验;跌落试验drug eluting coronary stents 药物洗脱支架drug product 药物产品drug substance 原料药drug-drug interaction56 药物-药物相互作用drug-food interaction 药物-食物的相互作用EEPS = Electronic Entry Processing System 电子录入处理系统effectiveness 疗效efficacy 有效性测定efficacy (Of a drug or treatment) 药效;药品疗效EEMEA = European Medical Evaluation Agency; European Agency for the Evaluation of Medicinal Products; European Medicines Agency 药物评价机构; 欧洲医药品管理局emergency envelope 应急信件Empiric Bayesian Multiple Gamma-Poisson Shrinker经验性贝氏法(伽玛泊松分布缩检法)empirical 经验性endpoint 终点endpoint criteria 终点指标factorial design 析因设计factorial trial 析因试验failure 无效,失败Fair Packaging and Labeling Act (1966) 公平包装和标签法False Claims Act 防制不实请求法false therapeutic claims 错误的疗效声明full analysis set 全分析集full factorial design 全因子试验法Iinclusion criteria 入选标准inclusion/exclusion criteria 入选/排除标准incremental exposure 食品中递增摄入量incubation period/latency period 潜伏期IND = Investigational New Drug 临床研究新药INDA = investigational new drug application NDA前申报阶段indemnity insurance 赔偿保险Independent Data Monitoring = IDM独立数据监察Independent Data Monitoring Committee = IDMC独立数据监察委员会independent ethics committee = IEC 独立伦理委员会indications 适应症investigational new drug = IND 临床研究新药investigational product 试验药物investigator 调研人员investigator's brochure = IB 研究者手册Mmasked 设盲mean absorption time = MAT(药物在体内的)平均吸收时间mean disintegration time = MDIT(药物在体内的)平均崩解时间Mean Dissolution Time = MDT (药物在体内的)平均释放时间Mean Residence Time = MRT(药物在体内的)平均滞留时间medical governance 医药治理Medicare 老年医疗保险制度;联邦老年医保medication guides (for patients) 用药指南Medicines Control Agency = MCA英国药品监督局Misbranding 错误标签; 冒牌Miscoding 编码错误missing value 缺失值mixed effect model 混合效应模式MLD = minimal lethal dose 最小致死剂量MoA = Mechanism of Action 作用机制;作用机理monitor 监查员monitoring plan监查计划monitoring report 监查报告MR = moderate response 好转MRA = Agreement on Mutual Recognition 相互承认协定MTD = maximal tolerance dose 最大耐受剂量multicenter trial 多中心试验multi-drug resistance 多药物抗药性multiple arm trials 多治疗组的试验mutual recognition procedure (EU) 相互承认程序OOS = Overall survival 总生存率Pparallel group design 平行组设计parameter estimation 参数估计parametric release 参数放行parametric statistics 参数统计方法patient file 病人档案patient global; pt global 病人总体评价patient history 病历per protocol ( PP) analysis 符合方案分析PFS = progression-free survival 无疾病进展存活率PGE = patient global evaluation 病人总体评价PHA = preliminary hazards analysis 预先危险分析pharmaceutical equivalence 药剂等效性pharmaceutics药剂学pharmacodynamics=PD 药物效应动力学; 简称药效学pharmacoepidemiology 药物流行病学pharmacokinetics = PK 药代动力学; 简称药动学pharmacology 药理学Pharmacovigilance105 药物警戒pharmacy 配药学PharMetrics claims database 索赔数据库PhRMA = Pharmaceutical Research and Manufacturers of America美国药物研究与生产商协会PIC=Pharmaceutical Inspection Convention 药品检查协定PIC/S Pharmaceutical Inspection Cooperation Scheme 药物检查合作计划pipeline assets 开发中产品PK = pharmacokinetics 药物代谢动力学; 药动学,药代动力学placebo 安慰剂placebo control 安慰剂对照placebo controlled study 安慰剂对照研究placebo effect 安慰剂效应PMA = premarket approval 上市前许可; 销售前批准PMCs = post marketing commitments 承诺药品上市后的继续研究PMDRA = Post Marketing Drug Risk Assessment 上市后药品风险评估(办公室) PMHx = Past Medical History 既往病史PMN = Premarket Notification 销售前通知PMS = Premenstrual syndrome 经前综合症POC (Proof-of-concept) Clinical Trials 概念证明POC = point-of-care testing 床旁分析polytomies 多分类pooled analysis = PA 荟萃分析postmarket surveillance 上市后监督post-marketing surveillance; postmarket safety surveillance 销售(上市)后监督power 把握度; 检验效能Pp = Process Performance 工序绩效Ppk = Process Performance Index 工序绩效指数precautions 慎用;注意事项precision 精密度preclinical (animal) data 临床前(动物实验)数据preclinical study 临床前研究predicate device = legally marketed device that is not subject to Premarket Approval (PMA)和已合法在市场上销售的且不需要做PMA“销售前批准”的Pre-market Approval (Application) = PMA上市前许可(申请)premarket notification 上市前通知pre-marketing surveillance 销售(上市)前监督preparing and submitting 起草和申报prescription drug 处方药preservation 保藏prevalence 患病率prevention trials预防试验primary (coronary) event 原位病变primary endpoint 主要终点primary mode of action = PMOA 首要作用模式primary variable 主要变量principal investigator = PI主要研究者Principles of Qualification 确认(验证)原则process controls 工艺控制process validation 工艺验证product codes 产品的号码product differentiation 产品差异化,产品特色化product license = PL 产品许可证product life cycle (PLC) 产品生命周期prognosis 预后progression-free survival = PFS 无进展生存progressive Disease PD 病情进展proof of principle study 原理循证研究propensity score 倾向性评分protocol 试验方案; 方案protocol amendment 方案补正prototype design 原型设计protozoa 原生动物门proven acceptable Range = PAR 确定可接受范围PTC = Product Technical Complaints 药品技术投诉Qqualification system for licensed pharmacist 执业药师资格准入制度qualified health claims 有保留的健康宣称Qualified Person = QP 受权人quality assurance = QA质量保证quality assurance unit = QAU质量保证部门quality control = QC 质量控制quality management systems 质量管理体系quality of life trials or supportive care trials 生存质量试验quality risk management = QRM 质量风险管理quantitative risk assessment 量化风险评估Rrandomization 随机化randomized trial 随机化试验randomized, double blinded clinical trial 随机双盲对照研究range check 范围检查rating scale 量表RCT = randomized clinical trials 随机临床试验RCT = randomized controlled trial 随机对照试验RDE: remote data entry 远距数据输入ready-to-eat foods 即食食品reagents 试剂recall 召回; 强制回收RECIST = Response Evaluation Criteria in Solid Tumors 实体瘤的疗效评价标准reconditioning 整改; 货物重整理;货物重包装recycled plastics 可循环利用塑料制品reference product 参比制剂reference samples 标准样品regulatory methodology 质量管理方法regulatory methods validation 管理用分析方法的验证(FDA对NDA提供的方法进行验证)regulatory specification 质量管理规格标准(NDA提供)rejection 排异remote monitoring system 远程监测系统; 远程监控REMS = Risk Evaluation and Mitigation Strategies 风险评估和减缓战略risk 受害risk assessment (risk analysis + risk evaluation) 风险评估,论证risk classification 风险分类;Risk Communications Advisory Committee 风险交流咨询委员会risk evaluation (part of risk assessment) 风险评价risk/ benefit analysis 风险-获益分析risk-benefit ratio 效益/风险比route of administration 给药途径royalties 专利使用费RPN = Risk Priority Number 风险优先指数RR = Response rate 缓解率RSD = (intra-day and inter-day) relative standard deviations (日内和日间) 相对标准差Ssafety advisory 安全建议safety evaluation 安全性评价safety evaluators 安全性评估人员safety set 安全性评价的数据集screening trials 筛选性试验SD = standard deviation 标准(偏)差SE = substantial equivalence 实质上的等同Seal Strength Test 密封强度试验sequence 试验次序SFDA 129= State Food And Drug Administration 国家食品药品监督管理局SG & A= Sales, General and Administration 销售、管理和一般费用shaft 传动轴SHEA = Society for Healthcare Epidemiology of America 美国医院流行病学学会sheaths 护套shelf life 保存期限; 保质期SIC codes = Standard Industrial Classification codes 标准产业分类代码side effects 副作用significance level 显著性水平Significant Risk (SR) 显著的危险性simple randomization 简单随机simulation model 仿真模型single blinding单盲single-blind study 单盲研究single-masked study 单盲研究site assessment = SA现场评估site audit 试验机构稽查SMDA = Safe Medical Devices Act of 1990 1990年安全医疗器械法SMF = Site Master File 生产场所主文件sNDA = supplemental NDA 疗效补充新药上市申请sponsor-investigator = SI 申办研究者spontaneous reports; voluntary reports 药品不良反应自愿报告SPS = Agreement on the Application Of Sanitary and Phytosanitary Measures卫生与植物卫生措施实施协议;简称SPS协议SSI = surgical site infection 手术部位感染SSOPs = Sanitation Standard Operating Procedures 卫生标准操作程序standard curve 标准曲线standard deviation 标准偏差standard drug 标准药物standard operating procedure = SOP 标准操作规程standard treatment 标准治疗Standards Of Care131 医护标准State Food and Drug Administration = SFDA国家食品药品监督管理局statistic 统计量statistical analysis plan = SAP 统计分析计划statistical model 统计模型statistical significance 统计学意义statistical tables 统计分析表Statisticians in the Pharmaceutical Industry = PSI制药业统计学家协会steady-state Area Under the Curve = AUCss稳态药时曲线下面积/稳态血药浓度-时间曲线下面积stratified 分层study audit 研究稽查study endpoint 研究终点Study Personnel List = SPL研究人员名单study site研究中心study type 研究类型subchronic toxicity studies 亚慢性毒性研究subgroup 亚组sub-investigator 助理研究者subject 受试者subject diary = SD 受试者日记subject enrollment 受试者入选subject enrollment log = SEL受试者入选表Subject Identification Code List = SIC受试者识别代码表subject recruitment 受试者招募subject screening log = SSL受试者筛选表submission 申报;递交subspecialties, internal medicine 亚专科,内科substantial equivalence to legally marketed (predicate) device 和已合法在市场上销售的且不需要做PMA“销售前批准”的相似产品有实质上的等同Ttrain-the-trainer program 培训者培训计划treatment group 试验组treatment IND 治疗性试验性新药申请treatment trials 治疗性试验trial error 试验误差trial initial meeting 试验启动会议trial master file 试验总档案trial objective 试验目的trial site 试验场所TRICARE 军队医疗系统triple blinding 三盲two one-side test 双单侧检验UAE = unexpected adverse event 预料外不良事件unblinding 破盲;揭盲under reporting bias 少报偏差unexplained syncope 不明原因晕厥unresectable 不能手术切除variability 变异variable 变量WHO International Collaborating Center for Drug Monitoring(世界卫生组织)国际药物监测合作中心WHO International Conference of Drug Regulatory Authorities= WHO-ICDRAWHO国际药品管理当局会议WHO Programme for International Drug Monitoring = PIDMWHO 国际药物监测合作计划。
FDA英语术语大全
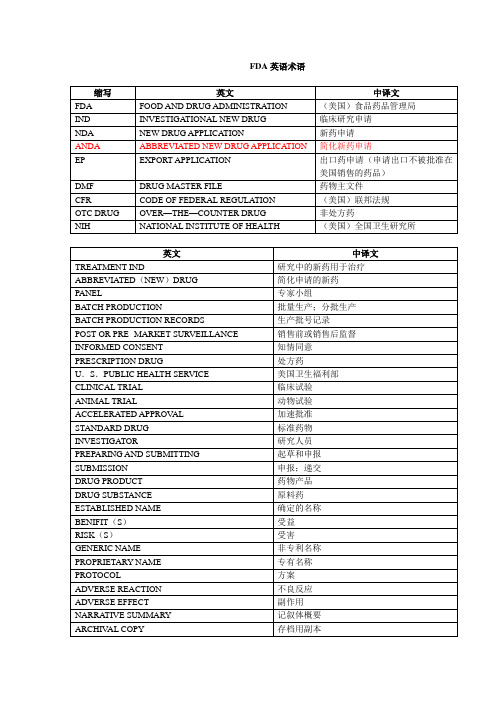
INVESTIGATOR
研究人员
PREPARING AND SUBMITTING
起草和申报
SUBMISSION
申报;递交
DRUG PRODUCT
药物产品
DRUG SUBSTANCE
原料药
ESTABLISHED NAME
确定的名称
BENIFIT(S)
受益
RISK(S)
受害
GENERIC NAME
销售前或销售后监督
INFORMED CONSENT
知情同意
PRESCRIPTION DRUG
处方药
U.S.PUBLIC HEALTH SERVICE
美国卫生福利部
CLINICAL TRIAL
临床试验
ANIMAL TRIAL
动物试验
ACCELERATED APPROVAL
加速批准
STANDARD DRUG
(美国)全国卫生研究所
英文
中译文
TREATMENTIND
研究中的新药用于治疗
ABBREVIATED(NEW)DRUG
简化申请的新药
PANEL
专家小组
BATCH PRODUCTION
批量生产;分批生产
BATCH PRODUCTION RECORDS
生产批号记录
POST-OR PRE- MARKET SURVEILLANCE
FDA英语术语
缩写
英文
中译文
FDA
FOOD AND DRUG ADMINISTRATION
(美国)食品药品管理局
IND
INVESTIGATIONAL NEW DRUG
临床研究申请
NDA
FDA临床试验常见词汇中译文对照

本资料由药智网收集整理action letter 决定通知active comparator 活性药物对照组active control = AC 阳性对照,活性对照active ingredient 有效成分Active Substance Master File (ASMF) 欧洲药物主文件acute myocardial infarction 急性心肌梗死acute tibial fractures 急性胫骨骨折adalimumab (Humira) 阿达木单抗adaptive design 自适应设计adaptive randomization 自适应随机ADE = adverse drug event 药物不良事件Adenoviral V ectors 腺病毒载体adequate and well-controlled studies 充分严格的对照研究ADHD = Attention-deficit hyperactivity disorder 注意力缺陷多动障碍; 注意力不足过动症; 多动症adhesion barrier product 防黏著产品adjuvant 助剂; 佐剂auxiliary;adjuvant therapy 佐药疗法,辅助疗法ADL = activities of daily living 日常生活活动能力ADME = absorption, distribution, metabolism, and excretion (药物)吸收、分配、代谢和排除ADR = adverse drug reaction 药物不良反应adrenal cortex 肾上腺皮质adrenal cortical hormone 肾上腺皮质激素adrenal gland 肾上腺adrenaline 肾上腺素adulterated devices 掺假器械adverse drug reaction = ADR药物不良反应adverse effect 副作用adverse event = AE 不良事件adverse medical events 不良医学事件adverse reaction (adverse event) 2 药物不良反应advisory 提醒advocacy and support groups3 倡导和支持团体AE = adverse event 4 不良事件AERS = Adverse Event Reporting System 不良事件报告系统药品注册。
FDA相关术语

FDA(FOOD AND DRUG ADMINISTRATION):(美国)食品药品管理局4*7NDA(NEW DRUG APPLICATION):新药申请LF=R8@rrmINDA(INVESTIGATIONAL NEW DRUG APPLICA TION):NDA前申报阶段~4}1yANDA(ABBREVIATED NEW DRUG APPLICA TION):简化新药申请>CEPA(EXPORT APPLICA TION):出口药申请(申请出口不被批准在美国销售的药品)Sz12 Q'gTREA TMENT IND:研究中的新药用于治疗(il'w`)NABBREVIATED(NEW)DRUG:简化申请的新药D4GYBDMF(DRUG MASTER FILE):药物主文件(持有者为谨慎起见而准备的保密资料,可以包括一个或多个人用药物在制备、加工、包装和贮存过程中所涉及的设备、生产过程或物品。
只有在DMF持有者或授权代表以授权书的形式授权给FDA,FDA在审查IND、NDA、ANDA时才能参考其内容)="b #HOLDER:DMF持有者@v-B5[VljdCFR(CODE OF FEDERAL REGULATION):(美国)联邦法规w*Rb*$-<pFPANEL:专家小组]h_,aDqBATCH PRODUCTION:批量生产;分批生产?orsD6BATCH PRODUCTION RECORDS:生产批号记录<v+onE}uDPOST-OR PRE- MARKET SURVEILLANCE:销售前或销售后监督1Lhfo/)_rCKINformED CONSENT:知情同意(患者对治疗或受试者对医疗试验了解后表示同意接受治疗或试验)-?:wz \PRESCRIPTION DRUG:处方药9Y~4[OTC DRUG(OVER—THE—COUNTER DRUG):非处方药46Z9*U.S.PUBLIC HEALTH SERVICE:美国卫生福利部Oxl^NIH(NATIONAL INSTITUTE OF HEALTH):(美国)全国卫生研究所eWNe3DCLINICAL TRIAL:临床试验fsH.Xj%^ANIMAL TRIAL:动物试验m7d+-)ACCELERA TED APPROV AL:加速批准r3($Dl9jmOSTANDARD DRUG:标准药物J/INVESTIGA TOR:调研人员7&dc\[9G0/PREPARING AND SUBMITTING:起草和申报TOwR^SUBMISSION:申报;递交+zOs4XIyBENIFIT(S):受益#VY<~b5O1NRISK(S):受害8KBv]xvlDRUG PRODUCT:药物产品SMKDRUG SUBSTANCE:原料药zpi,JQ}ESTABLISHED NAME:确定的名称1|&P*vGENERIC NAME:非专利名称7H&N*=W1oY8PROPRIETARY NAME:专有名称;p}7,INN(INTERNATIONAL NONPROPRIETARY NAME):国际非专有名称$UINARRATIVE SUMMARY记叙体概要JE`ADVERSE EFFECT:副作用Yp8LD>ulI9ADVERSE REACTION:不良反应bD~W_*IU{PROTOCOL:方案6zYZ6ARCHIVAL COPY:存档用副本YruREVIEW COPY:审查用副本h'ND'{pF,eOFFICIAL COMPENDIUM:法定药典(主要指USP、NF).%N2KNUSP(THE UNITED STATES PHARMACOPEIA):美国药典(现已和NF合并一起出版)U' H+ 0NF(NATIONAL formULARY):(美国)国家药品集j#yTM]%OFFICIAL=PHARMACOPEIAL= COMPENDIAL:药典的;法定的;官方的NQ=| :AGENCY:审理部门(指FDA))^*SPONSOR:主办者(指负责并着手临床研究者)izcIDENTITY:真伪;鉴别;特性)FYFFSTRENGTH:规格;规格含量(每一剂量单位所含有效成分的量)-vLABELED AMOUNT:标示量\'#~~REGULA TORY SPECIFICA TION:质量管理规格标准(NDA提供)4Jk{}REGULA TORY METHODOLOGY:质量管理方法(FDA用于考核原料药或药物产品是否符合批准了的质量管理规格标准的整套步骤)(Y^w!REGULA TORY METHODS V ALIDATION:管理用分析方法的验证(FDA对NDA提,5f}b供的方法进行验证)q+y@"w4PDietary supplement:食用补充品72[IODE (ORPHAN DRUG EXCLUSIVITY): 器官用药市场独占权y=k,XmSvL:RwNCE (NEW CHEMICAL ENTITY) : 新化合物Vu。
FDA术语表

FDA GlossaryCompiled by: Yaw-Tsong Lee Last updated: 13 September 2009[Top] [Laws & Regulations] [FDA Organization] [SFDA][Top] [Laws & Regulations] [FDA Organization] [SFDA]FDA Organization Charts[Top] [Laws & Regulations] [FDA Organization] [SFDA][Top] [Laws & Regulations] [FDA Organization] [SFDA]1 of the Bureau of Customs and Border Protection (CBP)2 An unintended reaction to a drug taken at doses normally used in man for prophylaxis, diagnosis, or therapy of disease, or for the modification of physiological function. In clinical trials, an ADR would include any injuries by overdosing, abuse/dependence, and unintended interactions with other medicinal products.3 An unwanted effect caused by the administration of drugs. Onset may be sudden or develop over time4 Organizations and groups that actively support participants and their families with valuable resources, including self-empowerment and survival tools.5 A negative experience encountered by an individual during the course of a clinical trial that is associated with the drug.6 An alanine aminotransferase (ALT) test measures the amount of this enzyme in the blood. ALT is measured to see if the liver is damaged or diseased.7 to check for liver disease or damage to the liver. Symptoms of liver disease can include jaundice, belly pain, nausea, and vomiting. An ALP test may also be used to check the liver when medicines that can damage the liver are taken or to check bone problems (sometimes found on X-rays), such as rickets, osteomalacia, bone tumors, Paget's disease, or too much of the hormone that controls bone growth (parathyroid hormone).8 Any of the treatment groups in a randomized trial.9 Low levels of AST are normally found in the blood. When body tissue or an organ such as the heart or liver is diseased or damaged, additional AST is released into the bloodstream. The amount of AST in the blood is directly related to the extent of the tissue damage.10 A renewable permit granted by the federal government to an institution or research center to conduct clinical trials.11 Benzodiazepines have also been used as a "date rape" drug because they can markedly impair and even abolish functions that normally allow a person to resist or even want to resist sexual aggression or assault12本类药物也称弱安定药,包括氯氮卓(利眠宁,chlordiazepoxide,商品名Librium)、地西泮(安定,diazepam,商品名valium)、硝西泮(硝基安定,nitrazepam)、氟西泮(氟安定,flurazepam)及奥沙西泮(去甲羟基安定,舒宁,oxazepam)。
- 1、下载文档前请自行甄别文档内容的完整性,平台不提供额外的编辑、内容补充、找答案等附加服务。
- 2、"仅部分预览"的文档,不可在线预览部分如存在完整性等问题,可反馈申请退款(可完整预览的文档不适用该条件!)。
- 3、如文档侵犯您的权益,请联系客服反馈,我们会尽快为您处理(人工客服工作时间:9:00-18:30)。
防撬包装[Top] [Laws & Regulations] [FDA Organization] [SFDA][Top] [Laws & Regulations] [FDA Organization] [SFDA]FDA Organization Charts[Top] [Laws & Regulations] [FDA Organization] [SFDA][Top] [Laws & Regulations] [FDA Organization] [SFDA]1 of the Bureau of Customs and Border Protection (CBP)2 a biologic response modifier, is a single-chain polypeptide containing 140 amino acids3 An unwanted effect caused by the administration of drugs. Onset may be sudden or develop over time4 Organizations and groups that actively support participants and their families with valuable resources, including self-empowerment and survival tools.5 A negative experience encountered by an individual during the course of a clinical trial that is associated with the drug.6The basic premise of AIP is: If FDA determines that a company’s applications are not reliable, the agency will not perform su bstantive review of any of the company’s applications until confidence in the data is restored.7 An alanine aminotransferase (ALT) test measures the amount of this enzyme in the blood. ALT is measured to see if the liver is damaged or diseased.8 to check for liver disease or damage to the liver. Symptoms of liver disease can include jaundice, belly pain, nausea, and vomiting. An ALP test may also be used to check the liver when medicines that can damage the liver are taken or to check bone problems (sometimes found on X-rays), such as rickets, osteomalacia, bone tumors, Paget's disease, or too much of the hormone that controls bone growth (parathyroid hormone).9 An allograft is a transplanted organ or tissue from a genetically non-identical member of the same species10 is a general linear model with a continuous outcome variable (quantitative) and two or more predictor variables where at least one is continuous (quantitative) and at least one is categorical (qualitative). ANCOVA is a merger of ANOVA and regression for continuous variables. ANCOVA tests whether certain factors have an effect on the outcome variable after removing the variance for which quantitative predictors (covariates) account. The inclusion of covariates can increase statistical power because it accounts for some of the variability11 Any of the treatment groups in a randomized trial.12 Low levels of AST are normally found in the blood. When body tissue or an organ such as the heart or liver is diseased or damaged, additional AST is released into the bloodstream. The amount of AST in the blood is directly related to the extent of the tissue damage.13 A renewable permit granted by the federal government to an institution or research center to conduct clinical trials.14 in an "as treated" (or "observed data") analysis only those patients still taking the assigned treatment are analyzed; those who drop out are "censored."15指由不直接涉及试验的人员所进行的一种系统性检查,以评价试验的实施、数据的记录和分析是否与试验方案、标准操作规程以及药物临床试验相关法规要求相符16一种批准用于治疗2型糖尿病的药物17 Benzodiazepines have also been used as a "date rape" drug because they can markedly impair and even abolish functions that normally allowa person to resist or even want to resist sexual aggression or assault18本类药物也称弱安定药,包括氯氮卓(利眠宁,chlordiazepoxide,商品名Librium)、地西泮(安定,diazepam,商品名valium)、硝西泮(硝基安定,nitrazepam)、氟西泮(氟安定,flurazepam)及奥沙西泮(去甲羟基安定,舒宁,oxazepam)。
临床主要用于镇静、催眠及对抗癫痫19指在设计临床试验方案、执行临床试验、分析评价临床试验结果时,有关影响因素所致的系统误差,致使疗效或安全性评价偏离真值。
20 expression of how much drug reaches the circulation (known to pharmacologists as the central compartment) after administration21 The property of being biologically compatible by not producing a toxic, injurious, or immunological response in living tissue22 A biofilm is a structured community of microorganisms encapsulated within a self-developed polymeric matrix and adherent to a living or inert surface23 A virus, therapeutic serum, toxin, antitoxin, vaccine, blood, blood component or derivative, allergenic product, or analogous product applicable to the prevention, treatment or cure of diseases or injuries of man. biological therapeutic agents that include blood and blood products, vaccines, allergenics, cell and tissue-based products, and gene therapy products24 Substances that stimulate the body's response to infection and disease. The body naturally produces small amounts of these substances. Scientists can produce some of them in the laboratory in large amounts for use in treating cancer, rheumatoid arthritis, and other diseases25 A biochemical feature or facet that can be used to measure the progress of disease or the effects of treatment26 Biosimilars or Follow-on biologics are terms used to describe officially-approved subsequent versions of innovator biopharmaceutical products made by a different sponsor following patent and exclusivity expiry on the innovator product. Biosimilars are also referred to as subsequent entry biologics (SEBs) in Canada27 A randomized trial is "Blind" if the participant is not told which arm of the trial he is on. A clinical trial is "Blind" if participants are unaware on whether they are in the experimental or control arm of the study; also called masked28在最后一份病例报告表输入数据库后,第一次揭盲之前对数据保持盲态的预分析审核,以便对统计分析计划作最后的决定。
