cochrane纳入的RCT文献质量评价中文版
cochrane纳入的RCT文献质量评价中文版教学内容

不全结局数据的数量,性质,处理方式导致失访偏倚
发表偏倚
Selective reporting.
说明如何审查选择性报道结局的可能性,以及审查结果
选择性报道结局导致发表偏倚
无缺失数据
缺失数据的产生不大可能与真实结局相关(对于生存数据,删失不大可能引入偏倚)
缺失数据的数目在各干预组相当,且各组缺失原因类似
对二分类变量,与观察事件的发生风险相比,缺失比例不足以影响预估的干预效应
对连续性结局数据,缺失数据的合理效应规模(均数差或标准均数差)不会大到影响观察的效应规模;
缺失的数据用合适的方法进行估算
无盲法或盲法不充分,但系统评价员判断结局很可能受到缺乏盲法的影响
尝试对关键的参与者和实施者行盲法,但盲法很可能被打破,结局很可能受到缺乏盲法的影响
风险未知
任何如下标准:
没有足够信息判断为低风险或高风险
研究未描述此情况
结局数据不完整
不全结局数据的数量,性质,处理方式导致失访偏倚
偏倚低风险标准
任何如下标准:
通过奇偶或出生日期产生序列
通过入院日期产生序列
通过类似住院号或门诊号产生序列
相对于上面提到的系统方法,其它非随机的方法少见的多,也更明显。通常包括对参与者进行判断或非随机的方法,例如:
临床医生判断如何分配
参与者判断如何分配
基于实验室检查或系列测试的结果分配
基于干预的可获取性进行分配
偏倚风险不清楚的判断标准
高风险判断标准
任何如下标准:
缺失数据的产生很大可能与真实结局相关,缺失数据的数目及缺失原因在各干预组相差较大
cochrane评分表

Risk of biasItem Authors'judgementDescriptionAdequate sequence generation? Yes Prinicipal author stated thatcomputer generatedallocation was usedAllocation concealment? Yes Prinicipal author stated thatallocation was concealedBlinding? Unclear No mention of study personnelor participants being blindto treatment groupIncomplete outcome data addressed? Yes All participants accountedfor, one 'drop out' recordedbut included by us in analysisFree of other bias? Unclear Possible uneven distributionof complete and incompleteparalysis at start of studybetween the two treatmentgroupsCochrane RCT质量评价标准:①随机方法是否正确。
②是否隐蔽分组。
③盲法的使用情况。
④失访或退出描述情况,有无采用意向性(ITT)分析。
以上质量标准中,如所有标准均为“充分”,则发生各种偏倚的可能性很小;如其中一条为不清楚,则有发生相应偏倚的中等度可能性;如其中一条为“不充分”或“未采用”,则有发生相应偏倚的高度可能性。
可参见:RCT的质量评价标准选择总结/bbs/topic/18137535?tpg=1&age=-1Quality assessmentThe quality of the trials was assessed and graded independently by two authors according to the criteria described in The Cochrane Handbook 4.2.6 (Higgins 2006). Gradings were compared and any inconsistencies between the authors in the interpretation of inclusion criteria and their significance to the selected study were discussed and resolved.The selected study was assessed for the following characteristics:1. The adequacy of the randomisation process (possible selection bias). Adequate randomisation includes any one of the following methods: computer generated or table of random numbers, drawing of lots, coin-toss, shuffling cards or throw of a dice. Inadequate methods of randomisation include the following: case record number, date of birth or alternate numbers.2. The adequacy of the allocation concealment (possible selection bias). Adequate methods of allocation concealment include either central randomisation (i.e. separate to other aspects of trial administration) or sequentially numbered sealed opaque envelopes. Inadequate concealment means an open allocation sequence in which either participants or trialists were able to foresee the upcoming assignment.3. The blinding of outcome assessors (i.e. whether the persons assessing the outcome of care were aware of which treatment the participant had received - possible performance bias).4. The extent and handling of losses to follow up (possible attrition bias). Adequate handling of losses to follow up involves a clear description and explanation being given of any significant difference between the losses of the intervention groups. An unacceptable loss in any one intervention group was considered to be loss greater than 20%.Study gradings A, B or C were employed for overall quality as follows.A: Minimisation of bias in all four categories above: i.e. adequate randomisation, few losses to follow up and intention-to-treat analysis, blinding of outcome assessors, high quality outcome assessment;B: Each of the criteria in A partially met;C: One or more of the criteria in A not met.Risk of bias in included studiesWe classified this study as grade C because of the uncertainty about blinding. The possibility of an uneven distribution of complete and incomplete palsies between the two groups is another potential source of bias and we conclude overall that this is a low quality study.Table 8.5.a: The Cochrane Collaboration’s tool for assessi ng risk of biasTable 8.5.c: Criteria for judging risk of bias in the ‘Risk of bias’ assessment toolFigure 8.6.a: Example of a ‘Risk of bias’ table for a single study (fictional)Table 8.7.a: Possible approach for summary assessments of the risk of bias for each important outcome (across domains) within and across studies。
cochrane纳入的RCT文献质量评价英文原版

Criteria for the judgement of ‘High risk’ of bias.
Participants or investigators enrolling participants could possibly foresee assignments and thus introduce selection bias, such as allocation based on:
RANDOM SEQUENCE GENERATION
Selection bias (biased allocation to interventions) due to inadequate generation of a randomised sequence.
Criteria for a judgement of ‘Low risk’ of bias.
Describe all measures used, if any, to blind outcome assessors from knowledge of which intervention a participant received. Provide any information relating to whether the intended blinding was effective.
Selection bias (biased allocation to interventions) due to inadequate concealment of allocations prior to assignment.
Performance bias.
cochrane纳入的RCT文献质量评价英文原版
Criteria for the judgement of ‘High risk’ of bias.
Participants or investigators enrolling participants could possibly foresee assignments and thus introduce selection bias, such as allocation based on:
Allocation by judgement of the clinician;
Allocation by preference of the participant;
Allocation based on the results of a laboratory test or a series of tests;
Describe the completeness of outcome data for each main outcome, including attrition and exclusions from the analysis. State whether attrition and exclusions were reported, the numbers in each intervention group (compared with total randomized participants), reasons for attrition/exclusions where reported, and any re-inclusions in analyses performed by the review authors.
(完整版)cochrane纳入的RCT文献质量评价中文版

洗牌的卡片和信封
掷骰子
抽签
最小化
*最小化,可实现无随机元素,被认为相当于是随机的。
判断为高风险的标准
研究者描述序列的产生使用的是非随机的方法。通常是系统的非随机方法,例如:
通过奇偶或出生日期产生序列
通过入院日期产生序列
通过类似住院号或门诊号产生序列
相对于上面提到的系统方法,其它非随机的方法少见的多,也更明显。通常包括对参与者进行判断或非随机的方法,例如:
研究未描述此情况
选择性发表
选择性发表导致发表偏倚
偏倚低风险标准
任何如下标准:
实验的计划书可获取,系统评价感兴趣的所有首要或次要结局均按计划书预先说明的方式报道
实验计划书不可得,但很明显发表的报告包括所有的结局,包括预先说明的结局(这种性质的有说服力的文字可能少见)
高风险判断标准
任何如下标准:
不是所有的预先说明的首要结局均被报道
尝试对关键的参与者和实施者行盲法,但盲法很可能被打破,结局很可能受到缺乏盲法的影响
风险未知
任何如下标准:
没有足够信息判断为低风险或高风险
研究未描述此情况
结局数据不完整
不全结局数据的数量,性质,处理方式导致失访偏倚
偏倚低风险标准
任何如下标准:
无缺失数据
缺失数据的产生不大可能与真实结局相关(对于生存数据,删失不大可能引入偏倚)
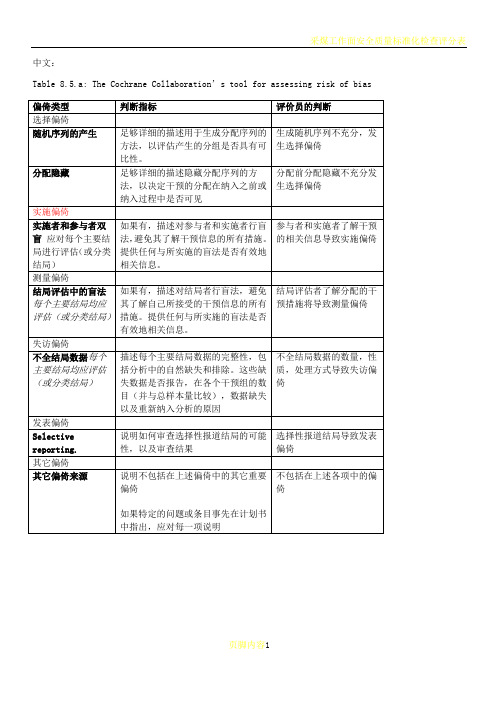
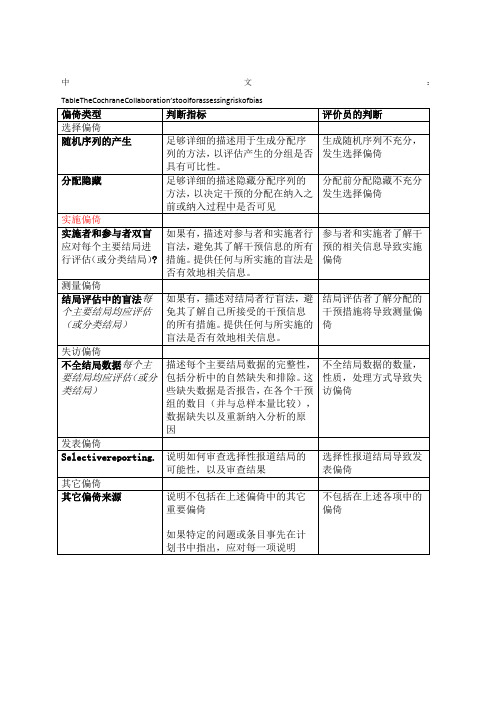
Table 8.5.a: The Cochrane Collaboration’s tool for assessing risk of bias
偏倚类型
判断指标
评价员的判断
选择偏倚
随机序列的产生
足够详细的描述用于生成分配序列的方法,以评估产生的分组是否具有可比性。
cochrane纳入的RCT文献质量评价(风险偏倚评估工具)中英文对照版

交替或循环
出生日期
病历号
其它明确的非隐藏过程
风险未知
没有足够信息判断为低风险或高风险。通常因分配隐藏的方法未描述或描述不充分。例如描述为使用信封分配,但为描述信封是否透明?密封?顺序编号?
对参与者和实施者的盲法
因参与者和实施者了解干预情况而导致实施偏倚
低风险判断标准
参与者以及纳入参与者的研究者因以下掩盖分配的方法或相当的方法,事先不了解分配情况
中心分配(包括电话,网络,药房控制随机)
相同外形的顺序编号的药物容器;
顺序编号、不透明、密封的信封
高风险判断标准
参与者以及纳入参与者的研究者可能事先知道分配,因而引入选择偏倚,譬如基于如下方法的分配:
使用摊开的随机分配表(如随机序列清单)
Selectionbias(biasedallocationtointerventions)duetoinadequateconcealmentofallocationspriortoassignment.
Performancebias.
BlindingofparticipantsandpersonnelAssessmentsshouldbemadeforeachmainoutcome(orclassofoutcomes).?
可能存在偏倚风险,但存在以下两种中的一种
没有足够信息评估是否存在其它重要的偏倚风险
没有足够的证据认为发现的问题会引入偏倚
summaryassessmentsoftheriskofbiasforeachimportantoutcome(acrossdomains)withinandacrossstudies
文献质量评估

文献质量评估
各纳入RCT的方法学质量评价采用Cochrane5.1手册推荐的简单评估法[9],评价的关键指标:①随机方法是否正确;②是否做到分配隐藏,分配方法是否正确;③是否实施盲法;
④是否报告失访和退出情况;⑤基线是否可比。
对于分配隐藏,将试验评为A(完全隐藏)、B(不清楚是否隐藏)、C(隐藏不充分)和D(没有使用隐藏)4个等级。
在其他方面将试验评为A(是)、B(不清楚)、C(否)三级。
如各评价条目均为A级,则为低度偏倚,发生各种偏倚的可能性最小,质量评为A级;若有一条目或多个条目为B,则该试验有发生相应偏倚的中等度可能性,质量评为B级;如其中有一条目或多个条目为C,则该试验有发生相应偏倚的高度可能性,质量评为C级。
cochrane纳入的RCT文献质量评价英文原版

If particular questions/entries were pre-specified in the review’s protocol, responses should be provided for each question/entry.
Bias due to problems not covered elsewhere in the table.
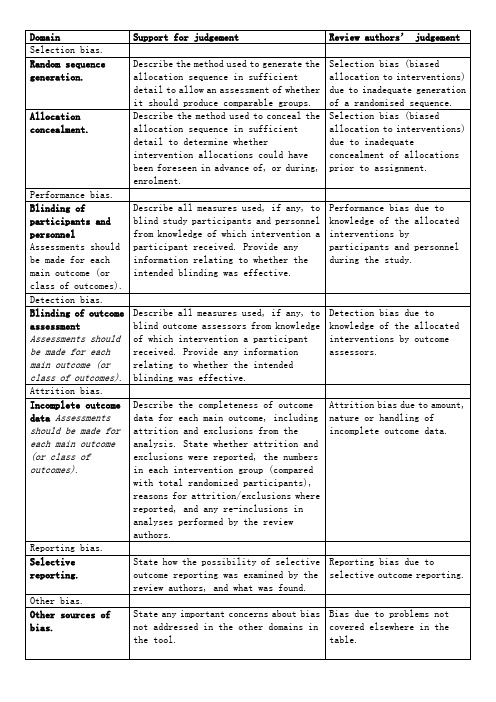
Domain
Support for judgement
Review authors’ judgement
Selection bias.
Random sequence generation.
Describe the method used to generate the allocation sequence in sufficient detail to allow an assessment of whether it should produce comparable groups.
Detection bias due to knowledge of the allocated interventions by outcome assessors.
Attrition bias.
cochrane纳入的RCT文献质量评价(风险偏倚评估工具)中英文对照版
中文:Table 8.5.a: The Cochrane Collaboration’s tool for assessing risk of biasTable 8.5.d: Criteria for judging risk of bias in the ‘Risk of bias’ assessment tool研究者描述随机序列产生过程譬如:参考随机数字表使用计算机随机数字生成器扔硬币洗牌的卡片和信封掷骰子抽签最小化*最小化,可实现无随机元素,被认为相当于是随机的。
研究者描述序列的产生使用的是非随机的方法。
通常是系统的非随机方法,例如:通过奇偶或出生日期产生序列通过入院日期产生序列通过类似住院号或门诊号产生序列相对于上面提到的系统方法,其它非随机的方法少见的多,也更明显。
通常包括对参与者进行判断或非随机的方法,例如:临床医生判断如何分配参与者判断如何分配基于实验室检查或系列测试的结果分配基于干预的可获取性进行分配中心分配(包括电话,网络,药房控制随机)相同外形的顺序编号的药物容器;顺序编号、不透明、密封的信封参与者以及纳入参与者的研究者可能事先知道分配,因而引入选择偏倚,譬如基于如下方法的分配:使用摊开的随机分配表(如随机序列清单)分发信封但没有合适的安全保障(如透明、非密封、非顺序编号)交替或循环出生日期病历号其它明确的非隐藏过程任何如下标准:无盲法或盲法不充分,但系统评价员判断结局不太可能受到缺乏盲法的影响参与者和主要实施者均实施可靠的盲法,且盲法不太可能被打破任何如下标准:无盲法或盲法不充分,但系统评价员判断结局很可能受到缺乏盲法的影响尝试对关键的参与者和实施者行盲法,但盲法很可能被打破,结局很可能受到缺乏盲法的影响任何如下标准:没有足够信息判断为低风险或高风险研究未描述此情况任何如下标准:无盲法或盲法不充分,但系统评价员判断结局不太可能受到缺乏盲法的影响参与者和主要实施者均实施可靠的盲法,且盲法不太可能被打破任何如下标准:无盲法或盲法不充分,但系统评价员判断结局很可能受到缺乏盲法的影响尝试对关键的参与者和实施者行盲法,但盲法很可能被打破,结局很可能受到缺乏盲法的影响任何如下标准:没有足够信息判断为低风险或高风险研究未描述此情况任何如下标准:无缺失数据缺失数据的产生不大可能与真实结局相关(对于生存数据,删失不大可能引入偏倚)缺失数据的数目在各干预组相当,且各组缺失原因类似对二分类变量,与观察事件的发生风险相比,缺失比例不足以影响预估的干预效应对连续性结局数据,缺失数据的合理效应规模(均数差或标准均数差)不会大到影响观察的效应规模;缺失的数据用合适的方法进行估算任何如下标准:缺失数据的产生很大可能与真实结局相关, 缺失数据的数目及缺失原因在各干预组相差较大对二分类变量,与观察事件的发生风险相比,缺失比例足以影响预估的干预效应对连续性结局数据,缺失数据的合理效应规模(均数差或标准均数差)足以影响观察的效应规模;意向治疗分析中存在实际干预措施与随机分配的干预相违背的情况对缺失数据进行简单的不合适的估算任何如下标准:没有报道缺失或排除的情况,无法判断高风险或低风险(如未说明随机的数量,未提供数据缺失的原因)研究未描述此情况任何如下标准:实验的计划书可获取,系统评价感兴趣的所有首要或次要结局均按计划书预先说明的方式报道实验计划书不可得,但很明显发表的报告包括所有的结局,包括预先说明的结局(这种性质的有说服力的文字可能少见)任何如下标准:不是所有的预先说明的首要结局均被报道一个或多个首要结局为采用预先说明的测量方法、分析方法或数据子集来报道系统评价感兴趣的一个或多个首要结局报道不全,以至于不能纳入meta分析研究未报道此研究应当包含的主要关键结局具有与特殊试验设计相关的潜在偏倚来源或被指欺诈或其它问题可能存在偏倚风险,但存在以下两种中的一种没有足够信息评估是否存在其它重要的偏倚风险没有足够的证据认为发现的问题会引入偏倚Table 8.7.a: Possible approach for summary assessments of the risk of bias for each important outcome (across domains) within and across studies英文:Table 8.5.a: The Cochrane Collaboration’s tool for assessing risk of biasTable 8.5.d: Criteria for judging risk of bias in the ‘Risk of bias’ assessment toolprocess such as:Referring to a random number table;Using a computer random number generator;Coin tossing;Shuffling cards or envelopes;Throwing dice;Drawing of lots;Minimization*.*Minimization may be implemented without a random element, and this isconsidered to be equivalent to being random.judgement The investigators describe a non-random component in the sequence generation process. Usually, the description would involve somesystematic, non-random approach, for example:Sequence generated by odd or even date of birth;Sequence generated by some rule based on date (or day) of admission;Sequence generated by some rule based on hospital or clinic recordnumber.Other non-random approaches happen much less frequently than thesystematic approaches mentioned above and tend to be obvious. Theyusually involve judgement or some method of non-random categorization ofparticipants, for example:Allocation by judgement of the clinician;Allocation by preference of the participant;Allocation based on the results of a laboratory test or a seriesof tests;Allocation by availability of the intervention.Criteria for a judgement Participants and investigators enrolling participants could not foreseeassignment because one of the following, or an equivalent method, was usedto conceal allocation:Central allocation (including telephone, web-based andpharmacy-controlled randomization);Sequentially numbered drug containers of identical appearance;Sequentially numbered, opaque, sealed envelopes.judgement Participants or investigators enrolling participants could possiblyforesee assignments and thus introduce selection bias, such as allocationbased on:Using an open random allocation schedule (e.g. a list of randomnumbers);Assignment envelopes were used without appropriate safeguards(e.g. if envelopes were unsealed or nonopaque or not sequentiallynumbered);Alternation or rotation;Date of birth;Case record number;Any other explicitly unconcealed procedure.Criteria for a judgement Any one of the following:No blinding or incomplete blinding, but the review authors judgethat the outcome is not likely to be influenced by lack of blinding;Blinding of participants and key study personnel ensured, andunlikely that the blinding could have been broken.judgementAny one of the following:No blinding or incomplete blinding, and the outcome is likely tobe influenced by lack of blinding;Blinding of key study participants and personnel attempted, butlikely that the blinding could have been broken, and the outcomeis likely to be influenced by lack of blinding.judgement ‘Unclear risk’ ofAny one of the following:Insufficient information to permit judgement of ‘Low risk’ or ‘High risk’;The study did not address this outcome.Criteria for a judgement Any one of the following:No blinding of outcome assessment, but the review authors judge thatthe outcome measurement is not likely to be influenced by lack ofblinding;Blinding of outcome assessment ensured, and unlikely that theblinding could have been broken.judgementAny one of the following:No blinding of outcome assessment, and the outcome measurement islikely to be influenced by lack of blinding;Blinding of outcome assessment, but likely that the blinding couldhave been broken, and the outcome measurement is likely to beinfluenced by lack of blinding.judgement ‘Unclear risk’ ofAny one of the following:Insufficient information to permit judgement of ‘Low risk’ or‘High risk’;The study did not address this outcome.Criteria for a judgement Any one of the following:No missing outcome data;Reasons for missing outcome data unlikely to be related to trueoutcome (for survival data, censoring unlikely to be introducingbias);Missing outcome data balanced in numbers across interventiongroups, with similar reasons for missing data across groups;For dichotomous outcome data, the proportion of missing outcomescompared with observed event risk not enough to have a clinicallyrelevant impact on the intervention effect estimate;For continuous outcome data, plausible effect size (difference inmeans or standardized difference in means) among missing outcomesnot enough to have a clinically relevant impact on observed effectsize;Missing data have been imputed using appropriate methods.judgement Any one of the following:Reason for missing outcome data likely to be related to trueoutcome, with either imbalance in numbers or reasons for missingdata across intervention groups;For dichotomous outcome data, the proportion of missing outcomescompared with observed event risk enough to induce clinicallyrelevant bias in intervention effect estimate;For continuous outcome data, plausible effect size (difference inmeans or standardized difference in means) among missing outcomesenough to induce clinically relevant bias in observed effect size;‘As-treated’ analysis done with substantial departure of theintervention received from that assigned at randomization;Potentially inappropriate application of simple imputation.judgement ‘Unclear risk’ ofAny one of the following:Insufficient reporting of attrition/exclusions to permit judgement of ‘Low risk’ or ‘High risk’ (e.g. number ran domized not stated,no reasons for missing data provided);The study did not address this outcome.Criteria for a judgement Any of the following:The study p rotocol is available and all of the study’spre-specified (primary and secondary) outcomes that are of interestin the review have been reported in the pre-specified way;The study protocol is not available but it is clear that thepublished reports include all expected outcomes, including thosethat were pre-specified (convincing text of this nature may beuncommon).judgementAny one of the following:Not all of the study’s pre -specified primary outcomes have beenreported;One or more primary outcomes is reported using measurements,analysis methods or subsets of the data (e.g. subscales) that werenot pre-specified;One or more reported primary outcomes were not pre-specified(unless clear justification for their reporting is provided, suchas an unexpected adverse effect);One or more outcomes of interest in the review are reportedincompletely so that they cannot be entered in a meta-analysis;The study report fails to include results for a key outcome thatwould be expected to have been reported for such a study.judgementThere is at least one important risk of bias. For example, the study: Had a potential source of bias related to the specific study designused; orHas been claimed to have been fraudulent; orHad some other problem.judgement ‘Unclear risk’ ofThere may be a risk of bias, but there is either:Insufficient information to assess whether an important risk of bias exists; orInsufficient rationale or evidence that an identified problem willintroduce bias.Table 8.7.a: Possible approach for summary assessments of the risk of bias for each important outcome (across domains) within and across studies。
cochrane纳入的RCT文献质量评价英文原版

judgement of ‘Low The investigators describe a random component in the sequence generation process such as:Referring to a random number table;Using a computer random number generator;Coin tossing;Shuffling cards or envelopes;Throwing dice;Drawing of lots;Minimization*.‘High The investigators describe a non-random component in the sequence generation process. Usually, the description would involve some systematic, non-random approach, for example: Sequence generated by odd or even date of birth;Sequence generated by some rule based on date (or day)of admission;Sequence generated by some rule based on hospital orclinic record number.Other non-random approaches happen much less frequently than the systematic approaches mentioned above and tend to be obvious.? They usually involve judgement or some method of non-random categorization of participants, for example: Allocation by judgement of the clinician;Allocation by preference of the participant;Allocation based on the results of a laboratory test ora series of tests;Allocation by availability of the intervention.Participants and investigators enrolling participants could not foresee assignment because one of the following, or an equivalent method, was used to conceal allocation: Central allocation (including telephone, web-based andpharmacy-controlled randomization);Sequentially numbered drug containers of identicalappearance;Sequentially numbered, opaque, sealed envelopes.‘High Participants or investigators enrolling participants could possibly foresee assignments and thus introduce selection bias, such as allocation based on:Using an open random allocation schedule (e.g. a listof random numbers);Assignment envelopes were used without appropriatesafeguards (e.g. if envelopes were unsealed or non-opaque or not sequentially numbered);Alternation or rotation;Date of birth;Case record number;Any other explicitly unconcealed procedure.Any one of the following:judgement of ‘LowNo blinding or incomplete blinding, but the reviewauthors judge that the outcome is not likely to beinfluenced by lack of blinding;Blinding of participants and key study personnelensured, and unlikely that the blinding could have beenbroken.Any one of the following:‘HighNo blinding or incomplete blinding, and the outcome islikely to be influenced by lack of blinding;Blinding of key study participants and personnelattempted, but likely that the blinding could have beenbroken, and the outcome is likely to be influenced bylack of blinding.Any one of the following:Insufficient information to permit judgement of ‘Lowrisk’ or ‘High risk’;The study did not address this outcome.Any one of the following:judgement of ‘LowNo blinding of outcome assessment, but the reviewauthors judge that the outcome measurement is not likelyto be influenced by lack of blinding;Blinding of outcome assessment ensured, and unlikelythat the blinding could have been broken.Any one of the following:‘HighNo blinding of outcome assessment, and the outcomemeasurement is likely to be influenced by lack ofblinding;Blinding of outcome assessment, but likely that theblinding could have been broken, and the outcomemeasurement is likely to be influenced by lack ofblinding.Any one of the following:Insufficient information to permit judgement of ‘Lowrisk’ or ‘High risk’;The study did not address this outcome.Any one of the following:judgement of ‘LowNo missing outcome data;Reasons for missing outcome data unlikely to be relatedto true outcome (for survival data, censoring unlikelyto be introducing bias);Missing outcome data balanced in numbers acrossintervention groups, with similar reasons for missingdata across groups;For dichotomous outcome data, the proportion of missingoutcomes compared with observed event risk not enoughto have a clinically relevant impact on the interventioneffect estimate;For continuous outcome data, plausible effect size(difference in means or standardized difference inmeans) among missing outcomes not enough to have aclinically relevant impact on observed effect size;Missing data have been imputed using appropriatemethods.Any one of the following:‘HighReason for missing outcome data likely to be related totrue outcome, with either imbalance in numbers orreasons for missing data across intervention groups;For dichotomous outcome data, the proportion of missingoutcomes compared with observed event risk enough toinduce clinically relevant bias in intervention effectestimate;For continuous outcome data, plausible effect size(difference in means or standardized difference inmeans) among missing outcomes enough to induceclinically relevant bias in observed effect size;‘As-treated’ analysis done with substantialdeparture of the intervention received from thatassigned at randomization;Potentially inappropriate application of simpleimputation.Any one of the following:Insufficient reporting of attrition/exclusions topermit judgement of ‘Low risk’ or ‘High risk’ (e.g.number randomized not stated, no reasons for missingdata provided);The study did not address this outcome.Any of the following:judgement of ‘LowThe study protocol is available and all of the study’spre-specified (primary and secondary) outcomes that areof interest in the review have been reported in thepre-specified way;The study protocol is not available but it is clear thatthe published reports include all expected outcomes,including those that were pre-specified (convincingtext of this nature may be uncommon).Any one of the following:‘HighNot all of the study’s pre-specified primary outcomeshave been reported;One or more primary outcomes is reported usingmeasurements, analysis methods or subsets of the data (e.g. subscales) that were not pre-specified;One or more reported primary outcomes were notpre-specified (unless clear justification for their reporting is provided, such as an unexpected adverse effect);One or more outcomes of interest in the review are reported incompletely so that they cannot be entered in a meta-analysis;The study report fails to include results for a key outcome that would be expected to have been reported for such a study.Had a potential source of bias related to the specificstudy design used; orHas been claimed to have been fraudulent; orHad some other problem.There may be a risk of bias, but there is either: Insufficient information to assess whether an importantrisk of bias exists; orInsufficient rationale or evidence that an identifiedproblem will introduce bias.。
rct文献质量评价表

rct文献质量评价表
rct(随机对照试验)文献质量评价表通常包括以下几
个方面的评价内容:
1.随机方法:评价随机方法是否正确,是否能够保证试验组和对照组的基线一致性。
2.对照设置:评价对照组的设置是否合理,是否能够反映实际临床情况,以及对照组是否与试验组具有可比性。
3.盲法实施:评价试验实施过程中是否采用盲法,是否能够减少主观偏倚的影响。
4.样本量:评价样本量是否足够大,以确保结果的稳定性和可靠性。
5.数据完整性:评价数据是否完整,是否有缺失或异常值,以及是否进行了合理的处理。
6.基线情况:评价试验组和对照组的基线情况是否相似,以确保试验结果的公正性和客观性。
7.疗效评价:评价疗效评价标准是否科学、客观、可重复,以及是否进行了合理的统计学分析。
8.安全性评价:评价安全性评价方法是否科学、客观、可重复,以及是否进行了合理的统计学分析。
9.试验流程:评价试验流程是否规范、合理,以及是否符合伦理要求。
10.结论可靠性:综合以上各点,对试验结论的可靠性进行评价。
在具体应用中,可以根据实际情况对rct文献质量评价表进行适当调整和增删。
(完整版)Cochrane协作网的RCT偏倚风险评价工具

风险不清楚
信息不充分不足以判断“低风险”或“高风险”
研究未提及这一结果
结果数据不完整
低风险
未缺失结果数据
缺失的结果数据原因与真实结果不太可能相关(如生存数据,删失数据不太可能引入偏倚)
缺失结果数据在各干预组的数量均衡,组间缺失数据具有相似的原因
风险不清楚
报告减员随机人数未说明,未提供缺失数据的原因)
研究未提及这一结果
选择性结果报告
低风险
有研究方案,且系统评价关心的方案中预先指定的(主要和次要)结果指标均有报告
没有研究方案,但所有期望的结局指标,包括在发表文章中预先指定的指标均有报告
高风险
根据干预措施的可及性分组等
风险不清楚
关于随机序列产生过程的信息不充分不足以判断“低风险”或“高风险”
分配隐藏
低风险
中心化分配(包括电话、基于网络和药房控制的随机化)
同一外观、连续编号的药物容器
按顺序编号、不透光、密封的信封
高风险
使用开放的随机分配表(如随机数字清单)
使用无适当保护措施的分配信封(如信封未密封、透光信封、信封未按顺序编号)
对于二分类结果数据,与观察的事件风险相比,缺失结果数据的比例足以对干预效应估计产生临床相关影响
对于连续性结果数据,缺失结果数据中似真的效应大小(均数差或标准均数差)足以对观测效应大小产生临床相关影响
采用“视为治疗(as-treated)”分析,但改变随机入组时干预措施的人数较多
可能不恰当地使用简单估算方法
尝试对受试者和主要研究人员施盲,但很可能破盲,并且结果很可能受缺乏盲法的影响
风险不清楚
信息不充分不足以判断“低风险”或“高风险”
cochrane文献评价手册

cochrane文献评价手册【原创实用版】目录1.Cochrane 文献评价手册的概述2.手册的目的和适用范围3.手册的主要内容4.手册的评价标准和方法5.手册的优点和不足6.对我国相关领域的影响和启示正文1.Cochrane 文献评价手册的概述Cochrane 文献评价手册是由 Cochrane 协作组织编写的一本关于如何评价和分析医学文献的指南。
Cochrane 协作组织是一个国际性的、非营利的医学研究组织,致力于通过系统评价和荟萃分析的方法,为临床决策提供高质量的证据。
2.手册的目的和适用范围本手册的主要目的是为系统评价和荟萃分析提供一种标准和方法,以便为临床决策提供可靠的证据。
它适用于所有从事医学研究、临床实践和卫生政策制定的人员。
3.手册的主要内容手册主要包括以下几个方面:(1)文献筛选:包括文献的检索、筛选和纳入;(2)文献的质量评估:包括随机对照试验、队列研究、病例对照研究等不同类型文献的质量评估;(3)数据提取和分析:包括数据的提取、整理和分析;(4)结果的报告和解释:包括结果的报告方式、如何解释结果等。
4.手册的评价标准和方法手册提供了一套详细的评价标准和方法,包括风险偏倚、质量评估、数据提取和分析等。
这些标准和方法被广泛接受和应用,是进行系统评价和荟萃分析的基础。
5.手册的优点和不足手册的优点在于提供了一套系统、全面、实用的评价方法和标准,对于提高研究的可靠性和质量具有重要的指导作用。
然而,手册也存在一些不足,例如部分内容较为复杂,对研究者的统计学和医学知识要求较高。
6.对我国相关领域的影响和启示Cochrane 文献评价手册对我国的医学研究、临床实践和卫生政策制定产生了深远的影响。
它提供了一种科学的、可靠的评价方法,有助于提高我国医学研究的质量和水平。
cochrane纳入的RCT文献质量评价(风险偏倚评估工具)中英文对照版讲解学习

缺失数据的产生不大可能与真实结局相关(பைடு நூலகம்于生存数据,删失不大可能引入偏倚)
缺失数据的数目在各干预组相当,且各组缺失原因类似
对二分类变量,与观察事件的发生风险相比,缺失比例不足以影响预估的干预效应
对连续性结局数据,缺失数据的合理效应规模(均数差或标准均数差)不会大到影响观察的效应规模;
缺失的数据用合适的方法进行估算
因参与者和实施者了解干预情况而导致实施偏倚
偏倚低风险标准
任何如下标准:
无盲法或盲法不充分,但系统评价员判断结局不太可能受到缺乏盲法的影响
参与者和主要实施者均实施可靠的盲法,且盲法不太可能被打破
偏倚高风险标准
任何如下标准:
无盲法或盲法不充分,但系统评价员判断结局很可能受到缺乏盲法的影响
尝试对关键的参与者和实施者行盲法,但盲法很可能被打破,结局很可能受到缺乏盲法的影响
实验计划书不可得,但很明显发表的报告包括所有的结局,包括预先说明的结局(这种性质的有说服力的文字可能少见)
高风险判断标准
任何如下标准:
不是所有的预先说明的首要结局均被报道
随机序列的产生
随机序列产生不充分导致选择偏倚
判断为低风险的标准
研究者描述随机序列产生过程譬如:
参考随机数字表
使用计算机随机数字生成器
扔硬币
洗牌的卡片和信封
掷骰子
抽签
最小化
*最小化,可实现无随机元素,被认为相当于是随机的。
判断为高风险的标准
研究者描述序列的产生使用的是非随机的方法。通常是系统的非随机方法,例如:
其它偏倚
其它偏倚来源
说明不包括在上述偏倚中的其它重要偏倚
如果特定的问题或条目事先在计划书中指出,应对每一项说明
循证护理文献质量评价工具

循证护理文献质量评价工具
循证护理文献质量评价工具是用于对医学或护理学科领域中的文献进
行质量评价的工具,其目的是确定文献的可信度和可靠性,以便提供客观、证据化的医学或护理实践建议。
以下是一些常见的循证护理文献质量评价工具:
1. Cochrane协作网络的风险偏倚工具(Risk of Bias Tool):用
于评价随机对照试验(RCTs)的质量,包括研究设计、随机化、盲法、缺
失数据等方面的风险偏倚。
2. Newcastle-Ottawa量表(NOS):用于评价病例对照研究和队列
研究的质量,包括选择对照组、病例和对照组间的匹配、病例和对照组间
的比较、评价因素是否对结果造成影响等方面。
3. Jadad量表:用于评价RCTs的质量,包括随机化的方法、盲法、
隐瞒分组等方面。
4.GRADE系统:用于评价循证医学研究证据的质量和可靠性,包括研
究设计、风险偏倚、一致性、精度、重要性等方面。
以上工具都可以通过评分的方式,对文献的各个方面进行客观评价和
分析,从而确定其在临床实践中的应用价值和可靠性,进一步提高医学和
护理的水平和效果。
cochrane纳入的RCT文献质量评价风险偏倚评估工具中英文对照
结局评估者了解分配的干预措施将导致测量偏倚
失访偏倚
不全结局数据每个主要结局均应评估(或分类结局)
描述每个主要结局数据的完整性,包括分析中的自然缺失和排除。这些缺失数据是否报告,在各个干预组的数目(并与总样本量比较),数据缺失以及重新纳入分析的原因
Selectionbias.
Randomsequencegeneration.
Describethemethodusedtogeneratetheallocationsequenceinsufficientdetailtoallowanassessmentofwhetheritshouldproducecomparablegroups.
基于实验室检查或系列测试的结果分配
基于干预的可获取性进行分配
偏倚风险不清楚的判断标准
没有足够的信息判断随机序列的产生存在高风险或低风险
分配隐藏
分配前不充足的分配隐藏导致选择偏倚
低风险判断标准
参与者以及纳入参与者的研究者因以下掩盖分配的方法或相当的方法,事先不了解分配情况
中心分配(包括电话,网络,药房控制随机)
Selectionbias(biasedallocationtointerventions)duetoinadequategenerationofarandomisedsequence.
Allocationconcealment.
Describethemethodusedtoconcealtheallocationsequenceinsufficientdetailtodeterminewhetherinterventionallocationscouldhavebeenforeseeninadvanceof,orduring,enrolment.
循证医学RevMan(Review

循证医学RevMan(Review篇一:循证医学系统评价一、概念系统评价(Systematic review,SR):是一种全新的文献综合方法,针对某一具体临床问题(如疾病的病因、诊断、治疗、预后),系统、全面地收集全世界所有已发表或未发表的临床研究,采用临床流行病学的原则和方法严格评价文献,筛选出符合质量标准的文献,进行定性或定量合成,得出综合可靠的结论。
是被公认为客观地评价和综合针对某一特定问题的研究证据的最佳手段。
独特的优点:良好的重复性二、Cochrane系统评价定义:Cochrane协作网成员在Cochrane协作网统一工作手册指导下,在相应Cochrane评价组编辑部指导和帮助下所完成的系统评价。
循证医学实践中最高质量的证据。
Cochrane系统评价目前主要限于RCT。
回答各类临床问题的证据“金字塔”。
Cochrane协作网:提供最佳证据的国家协作组织,旨在通过制作、保存、传播和不断更新医疗卫生各领域治疗措施的系统评价提高医疗保健干预措施的效率,帮助人们制定遵循证据的医疗决策。
三、系统评价的分类根据研究的临床问题:病因、治疗、诊断、预后等方面的系统评价根据纳入的原始研究类型不同:临床对照试验和观察性研究的系统评价根据纳入原始研究的方式:前瞻性、回顾性和累积性系统评价根据分析时是否采用统计学方法(Meta分析):定性和定量的系统评价四、为什么要进行系统评价:1. 应对信息时代的挑战、海量信息需要整合2. 及时转化和应用研究成果、连接新旧知识的桥梁3. 提高统计效能、避免“只见树木不见森林”4. 克服传统文献综述的缺陷五、叙述性文献综述(Narrative Review,NR):由作者根据特定的目的和需要或兴趣,收集有关的文献资料,采用定性分析的方法,对论文中阐述的研究目的、方法、结果、结论和观点等进行分析和评价,用自己的判断和观点,整理综合而成文。
六、系统评价与叙述性文献综述的区别与联系确定一篇综述为叙述性文献综述还是系统评价,主要取决于是否采用科学的方法以减少偏倚和混杂因素的影响。
cochrane纳入的RCT文献质量评价英文原版

Support for judgement
Review authors’ judgement
Selection bias.
Random sequence generation.
Describe the method used to generate the allocation sequence in sufficient detail to allow an assessment of whether it should produce comparable groups.
If particular questions/entries were pre-specified in the review’s protocol, responses should be provided for each question/entry.
Bias due to problems not covered elsewhere in the table.
Attrition bias due to amount, nature or handling of incomplete outcome data.
Reporting bias.
Selective reporting.
State how the possibility of selective outcome reporting was examined by the review authors, and what was found.
The investigators describe a random component in the sequence generation process such as:
cochrane纳入的RCT文献质量评价(风险偏倚评估工具)中英文对照版..

cochrane纳⼊的RCT⽂献质量评价(风险偏倚评估⼯具)中英⽂对照版..中⽂:Table 8.5.a: The Cochrane s tool for assessing riskTable 8.5.d: Criteria for judging risk ofbias in the assessment tool Risk of bias分配隐藏分配前不充⾜的分配隐藏导致选择偏倚Table 8.7.a: Possible approach for summarya ssessments of the risk of bias for each important outcome (across domains) within and across studies英⽂:Table 8.5.a: The Cochrane Collaboration ' s tool for assessing risk of biasTable 8.5.d: Criteria for judging risk of bias in the ‘Risk of bias ' assessment toolriskorsOTHER BIASTable 8.7.a: Possible approach for summary assessments of the risk of bias foreach important outcome (across domains) within and across studiesthose that were pre-specified (convincing text of this naturemay be uncommon).Criteria for theudgement o f ‘Highrisk of bias. Any one of the following: ' Not all of the study'-spse pcrifeied primary outcomes havebeen reported; One or more primary outcomes is reported using measurements,analysis methods or subsets of the data (e.g. subscales) thatwere not pre-specified;One or more reported primary outcomes were not pre-specified(unless clear justification for their reporting is provided,such as an unexpected adverse effect);One or more outcomes of interest in the review are reportedincompletely so that they cannot be entered in a meta-analysis;The study report fails to include results for a key outcomethat would be expected to have been reported for such a study.Criteria for the udgement of ‘ Unclear isk ' of bias.Insufficient information to permit judgement of ‘Low risk ' or ‘Hi It is likely that the majority of studies will fall into this category. gh risk。
- 1、下载文档前请自行甄别文档内容的完整性,平台不提供额外的编辑、内容补充、找答案等附加服务。
- 2、"仅部分预览"的文档,不可在线预览部分如存在完整性等问题,可反馈申请退款(可完整预览的文档不适用该条件!)。
- 3、如文档侵犯您的权益,请联系客服反馈,我们会尽快为您处理(人工客服工作时间:9:00-18:30)。
Table 8.5.a: The Cochrane Collaboration’s tool for assessing risk of bias
Tabl e 8.5.d: Criteria for judging risk of bias in the ‘Risk of bias’ assessment tool
参考随机数字表
使用计算机随机数字生成器
扔硬币
洗牌的卡片和信封
掷骰子
抽签
最小化
*最小化,可实现无随机元素,被认为相当于是随机的。
研究者描述序列的产生使用的是非随机的方法。
通常是系统的非随机方法,例如:
通过奇偶或出生日期产生序列
通过入院日期产生序列
通过类似住院号或门诊号产生序列
相对于上面提到的系统方法,其它非随机的方法少见的多,也更明显。
通常包括对参与者进行判断或非随机的方法,例如:
临床医生判断如何分配
参与者判断如何分配
基于实验室检查或系列测试的结果分配
基于干预的可获取性进行分配
参与者以及纳入参与者的研究者因以下掩盖分配的方法或相当的方法,事先不了解分配情况
中心分配(包括电话,网络,药房控制随机)
相同外形的顺序编号的药物容器;
顺序编号、不透明、密封的信封
参与者以及纳入参与者的研究者可能事先知道分配,因而引入选择偏倚,譬如基于如下方法的分配:
使用摊开的随机分配表(如随机序列清单)
分发信封但没有合适的安全保障(如透明、非密
封、非顺序编号)
交替或循环
出生日期
病历号
其它明确的非隐藏过程
任何如下标准:
无盲法或盲法不充分,但系统评价员判断结局不
太可能受到缺乏盲法的影响
参与者和主要实施者均实施可靠的盲法,且盲法
不太可能被打破
任何如下标准:
无盲法或盲法不充分,但系统评价员判断结局很
可能受到缺乏盲法的影响
尝试对关键的参与者和实施者行盲法,但盲法很
可能被打破,结局很可能受到缺乏盲法的影响任何如下标准:
没有足够信息判断为低风险或高风险
研究未描述此情况
任何如下标准:
无盲法或盲法不充分,但系统评价员判断结局不
太可能受到缺乏盲法的影响
参与者和主要实施者均实施可靠的盲法,且盲法
不太可能被打破
无盲法或盲法不充分,但系统评价员判断结局很
可能受到缺乏盲法的影响
尝试对关键的参与者和实施者行盲法,但盲法很
可能被打破,结局很可能受到缺乏盲法的影响
任何如下标准:
没有足够信息判断为低风险或高风险
研究未描述此情况
任何如下标准:
无缺失数据
缺失数据的产生不大可能与真实结局相关(对于
生存数据,删失不大可能引入偏倚)
缺失数据的数目在各干预组相当,且各组缺失原
因类似
对二分类变量,与观察事件的发生风险相比,缺
失比例不足以影响预估的干预效应
对连续性结局数据,缺失数据的合理效应规模(均
数差或标准均数差)不会大到影响观察的效应规
模;
缺失的数据用合适的方法进行估算
任何如下标准:
缺失数据的产生很大可能与真实结局相关, 缺失
数据的数目及缺失原因在各干预组相差较大
对二分类变量,与观察事件的发生风险相比,缺
失比例足以影响预估的干预效应
对连续性结局数据,缺失数据的合理效应规模(均
数差或标准均数差)足以影响观察的效应规模;
意向治疗分析中存在实际干预措施与随机分配
的干预相违背的情况
对缺失数据进行简单的不合适的估算
任何如下标准:
没有报道缺失或排除的情况,无法判断高风险或
低风险(如未说明随机的数量,未提供数据缺失
的原因)
研究未描述此情况
任何如下标准:
实验的计划书可获取,系统评价感兴趣的所有首
要或次要结局均按计划书预先说明的方式报道
实验计划书不可得,但很明显发表的报告包括所
有的结局,包括预先说明的结局(这种性质的有
说服力的文字可能少见)
任何如下标准:
不是所有的预先说明的首要结局均被报道
一个或多个首要结局为采用预先说明的测量方
法、分析方法或数据子集来报道
系统评价感兴趣的一个或多个首要结局报道不
全,以至于不能纳入meta分析
研究未报道此研究应当包含的主要关键结局
至少有一种重要的偏倚风险,例如:
具有与特殊试验设计相关的潜在偏倚来源
或被指欺诈
或其它问题
可能存在偏倚风险,但存在以下两种中的一种
没有足够信息评估是否存在其它重要的偏倚风险
没有足够的证据认为发现的问题会引入偏倚
Table 8.7.a: Possible approach for summary assessments of the risk of bias for each important outcome (across domains) within and across studies。
