药用丁基胶塞质量标准[指南]
胶塞的质量标准

1.目的
建立丁基胶塞的质量标准。
2.适用范围
质量管理部、生产管理部。
3.职责
生产管理部人员、质检人员、库管人员
4.定义
无
5.引用标准
YBB00042002
6.材料及设备
6.1无
7.流程图
无
8.内容
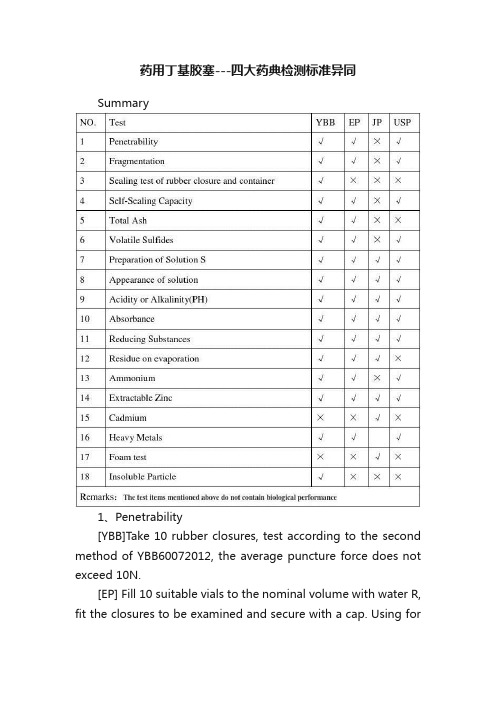
8.1. 穿刺落屑:按YBB00042002第二法对照法测定,落屑数≤5粒。
8.2 胶塞与容器密合性:取本品10个,置烧杯中,加水5分钟,取出,在70℃干
燥1小时,备用,另取10个与之配套的瓶加水至标示容量,用待测胶塞塞
紧,再加上与之配套的铝盖,压盖,放入高压蒸汽灭菌器中,121℃保持30分
钟,冷却至室温,放置24小时。
将瓶倒置,放入含10%亚甲兰溶液的容器
中,置于带抽气装置的容器中,抽真空至真空度为25kPa,维持30分钟,真
空装置恢复至常压,再放置30分钟,取出,用水冲洗瓶外壁,观察,亚甲兰
溶液不得渗入瓶内。
8.2.1 灰分遗留残渣< 50%
8.2.2 自密封性:取胶塞与容器密合性项下样品,采用穿剌力测定法YBB00042002
第二法中注射针,向胶塞不同部位穿剌,每个胶塞穿剌3次,每穿剌10次后
更换注射针,将样品倒置,放入含10%亚甲兰溶液的容器中,置于带抽气装置
的容器中,抽真空至真空度为25kPa,维持30分钟,真空装置恢复至常压,
再放置30分钟,取出,用水冲洗瓶外壁,观察,亚甲兰溶液不得渗入瓶内。
9.注意事项
无
10.附录及派生记录
无
11.相关文件
无
12.修订记录。
丁基橡胶药用瓶塞检测SOP

丁基橡胶药用瓶塞检测SOP1. 目的为规范注射液用卤化丁基橡胶药用瓶塞的检定,特制定本SOP。
2. 范围本SOP适用于直接与注射剂接触的卤化丁基橡胶塞(注射液用卤化丁基橡胶塞、预灌封注射器用氯化丁基橡胶塞、预灌封注射器用溴化丁基橡胶塞)的检定。
3. 定义无4. 职责4.1.QC负责本规程的起草、修订、培训及执行。
4.2.QA、QC组长、质量管理部经理负责本规程的审核。
4.3.质量总监负责批准本规程。
4.4.QA负责本规程执行的监督。
5. 引用标准5.1.注射液用卤化丁基橡胶塞国家食品药品监督管理局直接接触药品的包装材料和容器标准汇编5.2.预灌封注射器用氯化丁基橡胶活塞国家食品药品监督管理局直接接触药品的包装材料和容器标准汇编5.3.预灌封注射器用溴化丁基橡胶活塞国家食品药品监督管理局直接接触药品的包装材料和容器标准汇编5.4.《中华人民共和国药典》6. 材料6.1.仪器设备天平,恒温水浴箱,分光光度计,pH计,恒温干燥箱,变温电炉,干燥器,电导率仪,高温炉。
6.2.试剂溶液标准铅溶液:购入;氯化铵铵溶液:取氯化铵10.5g,加水溶解使成100ml,即得;标准锌溶液:称取硫酸锌(ZnSO4·7H2O)0.440g,置1000ml量瓶中,加水溶解并稀释至刻度,摇匀,即得(每1ml相当于100µg的Zn);0.02mol/L高锰酸钾滴定液: 按《高锰酸钾滴定液配制及标定SOP》操作;0.1mol/L硫代硫酸钠滴定液:按《硫代硫酸钠滴定液配制及标定SOP》操作;碳酸氢钠:购入;硝酸:购入;硝酸银试液:取硝酸银17.5g,加水适量使溶解成1000ml,摇匀,即得;0.1%氯化钾溶液:取氯化钾0.1g,加水使溶解成100 ml,即得;稀硫酸:取硫酸57ml,加水稀释至1000ml,即得;淀粉指示液:取可溶性淀粉0.5g,加水5ml搅匀后,缓缓倾入100ml沸水中,随加随搅拌,继续煮沸2min,放冷,倾取上层清液,即得,本液应临用新制;碱性碘化汞钾试液:二氯化汞饱和水溶液:取二氯化汞7g,溶于100ml水中,摇匀。
药用丁基胶塞---四大药典检测标准异同
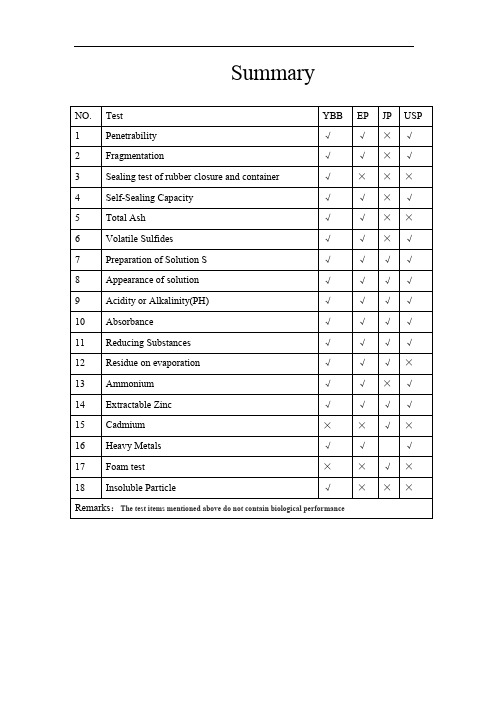
药用丁基胶塞---四大药典检测标准异同Summary1、Penetrability[YBB]Take 10 rubber closures, test according to the second method of YBB60072012, the average puncture force does not exceed 10N.[EP] Fill 10 suitable vials to the nominal volume with water R, fit the closures to be examined and secure with a cap. Using foreach closure a new, lubricated long-bevel(1) (bevel angle 12 ± 2°) hypodermic needle with an external diameter of 0.8 mm, pierce the closures with the needle perpendicular to the surface. The force required for piercing, determined with an accuracy of ± 0.25 N (25 gf), is not greater than 10 N (1 kgf) for each closure.[JP]None[USP]Fill 10 suitable vials to the nominal volume with water, fit the closures to be examined, and secure with a cap. Using a new hypodermic needle as described above for each closure, pierce the closure with the needle perpendicular to the surface.Requirement—The force for piercing is no greater than 10 N (1 kgf) f or each closure, determined with an accuracy of ± 0.25 N (25 gf).2、Fragmentation[YBB]Take 10 rubber closures, test according to the second method of YBB60082012, The total number of fragments does not exceed 5.[EP] For closures intended to be pierced by a hypodermic needle, carry out the following test. If the closures are to be used for aqueous preparations, place in12 clean vials a volume of water R corresponding to the nominal volume minus4 ml, close the vials with the closures to be examined, secure with a cap andallow to stand for 16 h. If the closures are to be used with dry preparations, close 12 clean vials with the closures to be examined. Using a lubricated long-bevel(1) (bevel angle 12 ±2°) hypodermic needle with an external diameter of 0.8 mm fitted to a clean syringe, inject into the vial 1 ml of water R and remove 1 ml of air ; carry out this operation 4 times for each closure,piercing each time at a different site. Use a new needle for each closure and check that the needle is not blunted during the test. Pass the liquid in the vials through a filter having approxi mately 0.5 μm pores. Count the fragments of rubber visible to the naked eye. The total number of fragments does not exceed5. This limit is based on the assumption that fragments witha diameter equal toor greater than 50 μm are visible to the naked eye; in cases of doubt or dispute, the fragments are examined with a microscope to verify their nature and size. [JP] None[USP] Closures for Liquid Preparations—Fill 12 clean vials with water to 4 mL less than the nominal capacity. Fit the closures to be examined, secure with acap, and allow to stand for 16 hours.Closures for Dry Preparations— Fit closures to be examined into 12 cleanvials, and secure each with a cap.Procedure— Using a hypodermic needle as described above fitted to a cleansyringe, inject into each vial 1 mL of water while removing 1 mL of air. Repeat this procedure 4 times for each closure, piercing each time at a different site.Use a new needle for each closure, checking that it is not blunted during the test.Filter the toatal volume of liquid in all the vials through a single filter with a nominal pore size no greater than 0.5 μm. Count the rubber fragments on the surface of the filter visible to the naked eye.Requirement— There are no more than 5 fragments visible. This limit is based on the assumption that fragments with adiameter >50 μm are visible to the naked eye. In case of doubt or dispute, the particles are examined microscopically to verify their nature and size.3、Sealing test of rubber closure and container[YBB] Take 10 rubber closures into the beaker, boil for 5 min , take out and dry 1h at 70℃ and ready to use. Fill 10 suitable vials to the nominal volume with water, fit the closures to be examined and secure with a cap. Heat in an autoclave so that a temperature of 121 ± 2 °C and maintain at this temperature for 30 min.Cool to room temperature and then place 24h, Immerse the vials upside down ina 10% solution of methylene blue R and reduce the external pressure by 25 kPafor 30 min. Restore atmospheric pressure and leave the vials immersed for 30 min. Rinse the outside of the vials. None of the vials contains any trace of colored solution.[EP]None[JP]None[USP] None4、Self-Sealing Capacity[YBB]Take the samples from sealing test of rubber closure and container, using for a hypodermic needle (YBB60082012 method 2), pierce each closure 3 times, change the hypodermic needle after it is used 10 times ,piercing each time at a different site.The vials upside down in a 10% solution of methylene blue R and reduce the external pressure by 25 kPa for 30 min. Restore atmospheric pressure and leave the vials immersed for 30 min. Rinse the outside of the vials. None of the vials contains any trace of colored solution.[EP]For closures intended to be used with multidosecontainers, carry out the following test. Fill 10 suitable vials to the nominal volume with water R, fit the closures to be examined and secure with a cap. Using for each closure a new hypodermic needle with an external diameter of 0.8 mm, pierce each closure 10 times, piercing each time at a different site. Immerse the vials upright in a 1 g/l solution of methylene blue R and reduce the external pressure by 27 kPa for 10 min. Restore atmospheric pressure and leave the vials immersed for 30 min.Rinse the outside of the vials. None of the vials contains any trace of coloured solution.[JP]None[USP]Procedure—Fill 10 suitable vials with water to the nominal volume. Fit the closures that are to be examined, and cap. Using a new hypodermic needle as described above for each closure, pierce each closure 10 times, piercing each time at a different site. Immerse the 10 vials in a solution of 0.1% (1 g per L) methylene blue, and reduce the external pressure by 27 kPa for 10 minutes.Restore to atmospheric pressure, and leave the vials immersed for 30 minutes.Rinse the outside of the vials.Requirement—None of the vials contain any trace of blue solution.5、Total Ash[YBB] Take the rubber closures, test according to YBB600212012, should be comply with the standard.[EP]The total ash (2.4.16) is within ± 10 per cent of the result obtained with the type sample.[JP]None[USP] None6、Volatile Sulfides[YBB]Take the rubber closures, according to the YBB60052012, should be comply with the standard.[EP] Place closures, cut if necessary, with a total surface area of 20 ± 2 cm2 in a 100 ml conical flask and add 50 ml of a 20 g/l solution of citric acid R. Place a piece of lead acetate paper R over the mouth of the flask and maintain the paper in position by placing over it an inverted weighing bottle. Heat in an autoclave at 121 ± 2℃ for 30 min. Any black stain on the paper is not more intense than that of a standard prepared at the same time in the same manner using 0.154 mg of sodium sulphide R and 50 ml of a 20 g/l solution of citric acid R.[JP]None[USP] Procedure—Place closures, cut if necessary, with a total surface area of 20 ± 2 cm2 in a 100-mL flask, and add 50 mL of a 20 g per L citric acid solution. In the same manner and at the same time, prepare a control solution in a separate 100-mL flask by dissolving 0.154 mg of sodium sulfide in 50 mL of a 20 g per L citric acid solution. Place a piece of lead acetate paper over the mouth of each flask, and hold the paper in position by placing over it an inverted weighing bottle. Heat the flasks in an autoclave at 121 ± 2℃for 30minutes.Requirement—Any black stain on the paper produced by Solution S is not more intense than that produced by the control solution.7、Preparation of Solution S[YBB] Put a number of uncut closures corresponding to a surface area of about 200 cm2 in a suitable glass container, cover with purified water( sample: water=1:2), boil for 5 min and rinse 5 times with the same volume of purified water. Place the washedclosures in a conical flask, add the same volume of water and weigh.Cover the mouth of the flask with a borosilicate-glass beaker. Heat in an autoclave so that a temperature of 121 ± 2 °C is reached within 20 min to 30 min and maintain at this temperature for 30 min. Cool to room temperature.Make up to the original mass with purified water. Shake and immediately separate the solution from the rubber by decantation. Shake solution S before each test Blank. Prepare a blank in the same manner using 400 mL of water for purified water.[EP] Solution S.Introduce a number of uncut closures corresponding to a surface area of about 100 cm2in a suitable glass container, cover with water for injections R, boil for 5 min and rinse 5 times with cold water for injections R.Place the washed closures in a wide-necked flask (glass type I, 3.2.1), add 200 ml of water for injections R and weigh. Cover the mouth of the flask with a borosilicate-glass beaker. Heat in an autoclave so that a temper ature of 121 ±2 °C is reached within 20 min to 30 min and maintain at this temperature for 30min. Cool to room temperature over about 30 min. Make up to the original mass with water for injections R. Shake and immediately separate the solution from the rubber by decantation. Shake solution S before each testBlank. Prepare a blank in the same manner using 200 ml of water for injections R.[JP] Wash the rubber closures with water, and dry at room temperature. Place them in a glass container, add water exactly 10 times the mass of the test material, close with a suitablestopper, heat at 121℃ for 1 hour in an autoclave, take out the glass container, allow to cool to room temperature,then take out immediately the rubber closures, and use the remaining solution as the testsolution. Prepare the blank solution with water in the same manner. Perform the following tests with the test solution and the blank solution[USP] Place whole, uncut closures corresponding to a surface area of 100 ± 10 cm2 intoa suitable glass container. Cover the closures with 200 mL of Purified Water orWater for Injection. If it is not possible to achieve the prescribed closure surface area (100 ± 10 cm2) using uncut closures, select the number of closures that will most closely approximate 100 cm2, and adjust the volume of water used to the equivalent of 2 mL per each 1 cm2 of actual closure surface area used. Boil for 5 minutes, and rinse five times with cold Purified Water or Water for InjectionPlace the washed closures into a Type I glass wide-necked flask (see Containers—Glass 660 ), add the same quantity of Purified Water or Water for Injection initially added to the closures, and weigh. Cover the mouth of the flask with a Type I glass beaker. Heat in an autoclave so that a temperature of 121 ± 2℃ is reached within 20 to 30 minutes, and maintain this temperature for30 minutes. Cool to room temperature over a period of about 30 minutes. AddPurified Water or Water for Injection to bring it up to the original mass. Shake, and immediately decant and collect the solution. [NOTE—This solution must be shaken before beingused in each of the tests]Prepare a blank solution similarly, using 200 mL of Purified Water or Water for Injection omitting the closures8、Appearance of solution[YBB]According to the part 2 of Chinese pharmacopoeia, 2010 edition of appendixⅨB and appendix IX A, standard solution is not more opalescent than reference suspension II. Standard solution is not more intensely coloured than No.5 reference solution[EP] Solution S is not more opalescent than reference suspension II for type I closures and is not more opalescent than reference suspension III for type II closures(2.2.1). Solution S is not more intensely coloured than reference solution GY5(2.2.2, Method II).[JP] Place 5 mL of the test solution in a glass-stoppered test tube of about 15 mm in inner diameter and about 200 mm in length, and shake vigorously for 3 minutes.The foam arisen disappears almost completely within 3 minutes.[USP] Determination of Turbidity (Opalescence)Procedure A: Visual Comparison— Use identical test tubes made of colorless, transparent, neutral glass with a flat base and an internal diameter of 15 to 25 mm. Fill one tube to a depth of 40 mm with Solution S, one tube to the same depth with water, and four others to the same depth with Reference Suspensions A, B, C,and D. Compare the solutions in diffuse daylight 5 minutes after preparation of the Reference Suspensions, viewing vertically against a black background. The light conditions shall be such that Reference Suspension A can be readily distinguished fromwater and that Reference Suspension B can be readily distinguished fromReference Suspension A.REQUIREMENT—Solution S is not more opalescent than Reference Suspension B for Type I closures, and not more opalescent than Reference Suspension C for Type II closures. Solution S is considered clear if its clarity is the same as that of water when examined as described above, or if its opalescence is not more pronounced than that of Reference Suspension A (refer to Table 3).Procedure B: Instrumental Comparison—Measure the turbidity of theReference Suspensions in a suitable calibrated turbidimeter (see Spectrophotometry and Light Scattering 851 ). The blank should be run and the results corrected for the blank. Reference Suspensions A, B, C, and D represent 3, 6, 18 and 30 Nephelometric Turbidity Units (NTU), respectively. Measure the turbidity of Solution S using the calibrated turbidimeter. REQUIREMENT—The turbidity of Solution S is not greater than that for Reference Suspension B (6 NTU FTU) for Type I closures, and is not greater than that forReference Suspension C (18 NTU FTU) for Type II closures (refer to Table 3).Determination of ColorColor Standard—Prepare a solution by diluting 3.0 mL of Matching Fluid O (see Color and Achromicity 631 ) with 97.0 mL of diluted hydrochloric acid. Procedure—Use identical tubes made of colorless, transparent, neutral glass with a flat base and an internal diameter of 15 to 25 mm. Fill one tube to a depth of 40 mm with Solution S, and the second with Color Standard. Compare the liquids in diffuse daylight, viewing vertically against a white background. Requirement—Solution S is not moreintensely colored than the Color Standard.9、Acidity or Alkalinity(PH)[YBB]Take each of 20 ml blank solution and solution S, respectively with KCl solution1 ml, According to the part2 of Chinese pharmacopoeia, 2010 edition ofappendixⅥH, the difference between the two may not be over 1.0.[EP] To 20ml of solution S add 0.1ml of bromothymol blue solution R1. Not more than 0.3ml of 0.01 M sodium hydroxide or 0.8 ml of 0.01 M hydrochloric acid is required to obtain either a blue or a yellow colour, respectively.[JP] T o 20 mL each of the test solution and the blank solution add 1.0 mL each of potassium chloride solution, prepared by dissolving 1.0 g of potassium chloride in water to make 1000 mL. The difference of pH between the two solutions is not more than 1.0[USP] Bromothymol Blue Solution—Dissolve 50 mg of bromothymol blue in a mixture of 4 mL of 0.02 M sodium hydroxide and 20 mL of alcohol. Dilute with water to 100 mL.Procedure—To 20 ml of Solution S add 0.1 ml of Bromothymol Blue Solution.If the solution is yellow, titrate with 0.01 N sodium hydroxide until a blue endpoint is reached. If the solution is blue, titrate with 0.01 N hydrochloric acid until a yellow endpoint is reached. If the solution is green, it is neutral and no titration is required.Blank Correction—Test 20 mL of Blank similarly. Correct the results obtained for Solution S by subtracting or adding the volume of titrant required for the Blank, as appropriate. (Reference Titrimetry 541 .)Requirement—Not more than 0.3 ml of 0.01 N sodium hydroxide produces a blue color, or not more than 0.8 ml of 0.01 N hydrochloric acid produces a yellow color, or no titration is required.10、Absorbance[YBB] Filter solution S on a membrane filter having approximately 0.45 μm pores.Measure the absorbance of the filtrate at wavelengths from 220 nm to 360 nm using the blank as compensation liquid. At these wavelengths, the absorbance does not exceed 0.1(according to the part 2 of Chinese pharmacopoeia, 2010 edition of appendixⅣA).[EP] Carry out the test within 5 h of preparation of solution S. Filter solution S on a membrane filter having approximately 0.45 μm pores rejecting the first few milliliters of filtrate. Measure the absorbance (2.2.25) of the filtrate at wavelengths from 220 nm to 360 nm using the blank (see solution S) as compensation liquid. At these wavelengths, the absorbance does not exceed 0.2 for type I closures or 4.0 for type II closures. If necessary, dilute the filtrate before measurement of the absorbance and correct the result for the dilution. [JP] Read the absorbance of the test solution between 220 nm and 350 nm against the blank solution as directed under Ultraviolet-visible Spectrophotometry <2.54>: it is not more than 0.20.[USP] Procedure — [NOTE—Perform this test within 5 hours of preparing Solution S.] Filter Solution S through a 0.45-μm pore size filter, discarding the first few mL of filtrate. Measure the absorbance of the filtrate at wavelengths between 220 and 360 nm in a 1-cm cell using the blank in a matched cell in the reference beam. If dilution of the filtrate is required beforemeasurement of the absorbance, correct the test results for the dilution.Requirement—The absorbances at these wavelengths do not exceed 0.2 for Type I closures or 4.0 for Type II closures.11、Reducing Substances[YBB] To 20.0 mL of solution S add 1 mL of dilute sulfuric acid R and 20.0 mL of0.002 M potassium permanganate. Boil for 3 min. Cool. Add 0.1 g of potassiumiodide R and titrate immediately with 0.01 M sodium thiosulfate until the colorturned light brown , using 5 drops of starch solution R as indicator. Carry out atitration using 20.0 mL of the blank. The difference between the titrationvolumes is not greater than7.0mL.[EP] Carry out the test within 4 h of preparation of solution S. To 20.0 ml of solution S add 1 ml of dilute sulphuric acid R and 20.0 ml of 0.002 M potassium permanganate. Boil for 3min. Cool. Add 1 g of potassium iodide R and titrate immediately with 0.01 M sodium thiosulphate, using 0.25 ml of starch solution R as indicator. Carry out a titration using 20.0 ml of the blank. The difference between the titration volumes is not greater than 3.0 ml for type I closures and7.0 ml for type II closures.[JP] Measure 100 mL of the test solution in a glass-stoppered,Erlenmyer flask, add10.0 mL of 0.002 mol/L potassium permanganate VS and 5 mL of dilutesulfuric acid, and boil for 3 minutes. After cooling, add 0.10g of potassium iodide,stopper, mix by shaking, then allow to stand for 10 minutes,and titrate<2.50> with 0.01 mol/L sodium thiosulfate VS(indicator: 5 drops of starch TS).Perform the blank test in the same manner, using 100 mL of the blank solution.The difference in mL of 0.002 mol/L potassium permanganate VS required between the tests is not more than 2.0 mL.[USP] Procedure— [NOTE—Perform this test within 4 hours of preparing Solution S.] T o 20.0 mL of Solution S add 1 mL of diluted sulfuric acid and 20.0 mL of0.002 M potassium permanganate. Boil for 3 minutes. Cool, add 1 g ofpotassium iodide, and titrate immediately with 0.01 M sodium thiosulfate, using0.25 mL of starch solution TS as the indicator. Perform a titration using 20.0mL of blank and note the difference in volume of 0.01 M sodium thiosulfaterequired.Requirement—The difference between the titration volumes is not greater than 3.0 mL for Type I closures and not greater than 7.0 mL for Type II closures.12、Residue on evaporation[YBB]Evaporate 100 mL of solution S and blank solution to dryness on a water-bath and dry at 100 °C to 105 °C. The residue weighs not more than 4.0 mg. [EP]Evaporate 50.0 ml of solution S to dryness on a water-bath and dry at 100 °C to 105 °C. The residue weighs not more than 2.0 mg for type I rubber and notmore than 4.0 mg for type II rubber.[JP]Measure 100 mL of the test solution, evaporate on a water bath to dryness, and dry the residue at 1059 C for 1 hour.The mass of the residue is not morethan 2.0 mg.[USP]None13、Ammonium[YBB] Precision measuring 10 ml of solution S, adding alkaline potassium iodide solution 2 ml, place 15 minutes, should not be color; if it colored , compare with reference solution (with 2.0 ml ammonium chloride solution (take ammoniumchloride 31.5 mg, add right amount chlorine free water and dissolve diluted to1000 ml), 8 ml blank reference solution, 2ml alkaline potassium mercuric iodide solution for mixing), should not be more intensely colored (0.0002%)[EP] maximum 2 ppm.Dilute 5 ml of solution S to 14 ml with water R. The solution complies with limit test A.[JP]None[USP] Alkaline Potassium Tetraiodomercurate Solution—Prepare a 100 mL solution containing 11 g of potassium iodide and 15 g of mercuric iodide in water.Immediately before use, mix 1 volume of this solution with an equal volume ofa 250 g per L solution of sodium hydroxide.Test Solution— Dilute 5 mL of Solution S to 14 mL with water. Make alkaline if necessary by adding 1 N sodium hydroxide, and dilute with water to 15 mL.Add 0.3 mL of Alkaline Potassium TetraiodomercurateSolution, and close the container.Ammonium Standard Solution—Prepare a solution of ammonium chloride in water (1 ppm NH4). Mix 10 mL of the 1 ppm ammonium chloride solution with5 mL water and 0.3 mL of Alkaline Potassium Tetraiodomercurate Solution.Close the container.Requirement—After 5 minutes, any yellow color in the Test Solution is no darker than the Ammonium Standard Solution (no more than 2 ppm of NH4 in Solution S).14、Extractable Zinc[YBB] Filter solution S on a membrane filter having approximately 0.45μm pores, Precision measuring filtrate 10 ml, add 1 ml 2 mol/L of hydrochloric acid and 3 drops of potassium ferrocyanide test solution(weight 4.2 g potassium ferrocyanide trihydrate, dissolve and diluted with water to 100 ml, shake evenly, this product should be new prepared) for mixing, should not be color; if it colored, compare with reference solution( with 3 ml standard zinc solution (weight 44.0g Zinc sulfate seven hydrated compounds, with new boiled and cooled purified water dissolved and diluted to 1000 ml, this product should be new prepared), shall not be deeper(0.0002%),7ml blank reference solution, 1ml 2mol/L hydrochloric acid and 3 drops of potassium ferrocyanide solution for mixing), should not be more intensely colored.(0.0003%)[EP] maximumof 5 μg of extractable Zn per millilitre of solution S.Atomic absorption spectrophotometry (2.2.23, Method I). Test solution. Dilute10.0 ml of solution S to 100 ml with 0.1 M hydrochloric acid.Reference solutions. Prepare the reference solutions using zinc standard solution(10 ppm Zn) R diluted with 0.1 M hydrochloric acid.Source: zinc hollow-cathode lamp.Wavelength: 213.9 nm.Flame: air-acetylene.[JP]T o 10.0 mL of the test solution add diluted dilute nitric acid (1 in 3) to make 20 mL, and use this solution as the sample solution. Further, to 1.0 mL of Standard Zinc Solutionfor atomic absorption spectrophotometry add diluted nitricacid (1 in 3) to make exactly 20 mL, and use this solution asthe standard solution. Perform the tests according to the Atomic Absorption Spectrophotometry <2.23>,using these solutions, under the following conditions.The absorbanceof the sample solution is not more than that of the standardsolution.Gas: Combustible gasóAcetylene.。
免洗免灭药用胶塞质量标准

免洗免灭药用胶塞质量标准一、材质合规1.免洗免灭药用胶塞应使用符合国家相关标准的材质制造,如医用级硅橡胶、聚乙烯等。
2.材质应无毒、无异味、无刺激性,不含有对人体有害的物质。
二、物理性能1.免洗免灭药用胶塞应具有一定的弹性和回弹性,以确保良好的密封性能。
2.免洗免灭药用胶塞应具有足够的抗压性能,以承受药品包装的压力。
3.免洗免灭药用胶塞应具有优良的耐寒性能,以确保在低温环境下正常工作。
三、化学性能1.免洗免灭药用胶塞应具有优良的化学稳定性,能够耐受药品的腐蚀和溶剂的影响。
2.免洗免灭药用胶塞应不与药品发生反应,不影响药品的质量和稳定性。
3.免洗免灭药用胶塞应具有良好的耐清洗性能,能够在清洗过程中保持洁净。
四、密封性能1.免洗免灭药用胶塞应具有良好的密封性能,能够有效地防止药品泄漏和污染。
2.免洗免灭药用胶塞应与药瓶配合紧密,确保药品在运输和储存过程中的稳定性。
3.免洗免灭药用胶塞应具有防漏性能,能够防止药品在运输和储存过程中的泄漏。
五、生物相容性1.免洗免灭药用胶塞应具有良好的生物相容性,不会对人体产生不良反应。
2.免洗免灭药用胶塞应符合国家相关标准,如GB/T 16886等。
六、无菌性能1.免洗免灭药用胶塞应经过严格的灭菌处理,确保无菌性能。
2.免洗免灭药用胶塞应在无菌环境下生产、包装和储存,确保整个生产过程中的无菌性。
七、耐候性能1.免洗免灭药用胶塞应具有优良的耐候性能,能够在不同气候条件下保持稳定。
2.免洗免灭药用胶塞应能够耐受紫外线、氧化等因素的影响,不发生老化反应。
3.免洗免灭药用胶塞应能够抵抗药品包装中的化学物质和气体等的影响,不发生变质或变形。
八、抗老化性能1.免洗免灭药用胶塞应具有优良的抗老化性能,能够在长时间的使用过程中保持稳定。
2.免洗免灭药用胶塞应能够抵抗自然环境和人为因素(如摩擦、撞击等)的影响,不发生磨损或损坏。
3.免洗免灭药用胶塞应能够抵抗药品包装中的化学物质和气体等的影响,不发生老化反应。
丁基橡胶塞 质量标准

丁基橡胶塞质量标准一、外观质量1.丁基橡胶塞表面应光滑、平整,无明显的气泡、裂纹和杂质。
2.丁基橡胶塞的色泽应均匀,符合设计要求。
3.丁基橡胶塞的形状和尺寸应符合设计图纸要求。
二、尺寸精度1.丁基橡胶塞的尺寸应符合设计图纸要求,误差不超过±0.1mm。
2.丁基橡胶塞的孔径和孔深应符合设计图纸要求,误差不超过±0.2mm。
三、物理性能1.丁基橡胶塞应具有良好的弹性和回弹性,以满足使用要求。
2.丁基橡胶塞应具有较好的耐温性能,能在规定的使用温度下保持其物理性能不变。
3.丁基橡胶塞应具有较好的耐老化性能,经过规定的老化试验后,仍能保持良好的使用性能。
四、化学性能1.丁基橡胶塞应具有较好的化学稳定性,能耐受常见的化学物质的侵蚀。
2.丁基橡胶塞应不含有对人体有害的物质,符合相关卫生标准。
五、密封性能1.丁基橡胶塞应具有较好的密封性能,能够有效地防止液体和气体的泄漏。
2.在规定的使用压力下,丁基橡胶塞不应出现明显的变形或损坏。
六、耐老化性能1.丁基橡胶塞应能在规定的温度和湿度条件下,经过一定时间的老化试验后,仍能保持良好的使用性能。
2.丁基橡胶塞的老化试验包括但不限于热老化、紫外线老化、臭氧老化等。
七、无毒无害1.丁基橡胶塞应采用无毒、无害的原材料制作,不含有对人体有害的物质。
2.丁基橡胶塞应符合相关的卫生标准和环保要求。
3.八、符合设计要求4.丁基橡胶塞应满足设计图纸中的各项技术要求,包括但不限于材质、结构、尺寸、性能等。
5.在生产和检验过程中,应严格遵守相关的工艺流程和检验标准,确保产品质量符合设计要求。
药用丁基胶塞---四大药典检测标准异同
Summary1、Penetrability[YBB]Take 10 rubber closures, test according to the second method of YBB60072012, the average puncture force does not exceed 10N.[EP] Fill 10 suitable vials to the nominal volume with water R, fit the closures to be examined and secure with a cap. Using for each closure a new, lubricated long-bevel(1) (bevel angle 12 ± 2°) hypodermic needle with an external diameter of 0.8 mm, pierce the closures with the needle perpendicular to the surface. The force required for piercing, determined with an accuracy of ± 0.25 N (25 gf), is not greater than 10 N (1 kgf) for each closure.[JP]None[USP]Fill 10 suitable vials to the nominal volume with water, fit the closures to be examined, and secure with a cap. Using a new hypodermic needle as described above for each closure, pierce the closure with the needle perpendicular to the surface.Requirement—The force for piercing is no greater than 10 N (1 kgf) for each closure, determined with an accuracy of ± 0.25 N (25 gf).2、Fragmentation[YBB]Take 10 rubber closures, test according to the second method of YBB60082012, The total number of fragments does not exceed 5.[EP] For closures intended to be pierced by a hypodermic needle, carry out the following test. If the closures are to be used for aqueous preparations, place in12 clean vials a volume of water R corresponding to the nominal volume minus4 ml, close the vials with the closures to be examined, secure with a cap andallow to stand for 16 h. If the closures are to be used with dry preparations, close 12 clean vials with the closures to be examined. Using a lubricated long-bevel(1) (bevel angle 12 ±2°) hypodermic needle with an external diameter of 0.8 mm fitted to a clean syringe, inject into the vial 1 ml of water R and remove 1 ml of air ; carry out this operation 4 times for each closure, piercing each time at a different site. Use a new needle for each closure and check that the needle is not blunted during the test. Pass the liquid in the vials through a filter having approxi mately 0.5 μm pores. Count the fragments of rubber visible to the naked eye. The total number of fragments does not exceed5. This limit is based on the assumption that fragments with a diameter equal toor greater than 50 μm are visible to the naked eye; in cases of doubt or dispute, the fragments are examined with a microscope to verify their nature and size. [JP] None[USP] Closures for Liquid Preparations— Fill 12 clean vials with water to 4 mL less than the nominal capacity. Fit the closures to be examined, secure with acap, and allow to stand for 16 hours.Closures for Dry Preparations— Fit closures to be examined into 12 cleanvials, and secure each with a cap.Procedure— Using a hypodermic needle as described above fitted to a cleansyringe, inject into each vial 1 mL of water while removing 1 mL of air. Repeat this procedure 4 times for each closure, piercing each time at a different site.Use a new needle for each closure, checking that it is not blunted during the test.Filter the toatal volume of liquid in all the vials through a single filter with a nominal pore size no greater than 0.5 µm. Count the rubber fragments on the surface of the filter visible to the naked eye.Requirement— There are no more than 5 fragments visible. This limit is based on the assumption that fragments with a diameter >50 µm are visible to the naked eye. In case of doubt or dispute, the particles are examined microscopically to verify their nature and size.3、Sealing test of rubber closure and container[YBB] Take 10 rubber closures into the beaker, boil for 5 min , take out and dry 1h at 70℃ and ready to use. Fill 10 suitable vials to the nominal volume with water, fit the closures to be examined and secure with a cap. Heat in an autoclave so that a temperature of 121 ± 2 °C and maintain at this temperature for 30 min.Cool to room temperature and then place 24h, Immerse the vials upside down ina 10% solution of methylene blue R and reduce the external pressure by 25 kPafor 30 min. Restore atmospheric pressure and leave the vials immersed for 30 min. Rinse the outside of the vials. None of the vials contains any trace of colored solution.[EP]None[JP]None[USP] None4、Self-Sealing Capacity[YBB]Take the samples from sealing test of rubber closure and container, using for a hypodermic needle (YBB60082012 method 2), pierce each closure 3 times, change the hypodermic needle after it is used 10 times ,piercing each time at a different site.The vials upside down in a 10% solution of methylene blue R and reduce the external pressure by 25 kPa for 30 min. Restore atmospheric pressure and leave the vials immersed for 30 min. Rinse the outside of the vials. None of the vials contains any trace of colored solution.[EP]For closures intended to be used with multidose containers, carry out the following test. Fill 10 suitable vials to the nominal volume with water R, fit the closures to be examined and secure with a cap. Using for each closure a new hypodermic needle with an external diameter of 0.8 mm, pierce each closure 10 times, piercing each time at a different site. Immerse the vials upright in a 1 g/l solution of methylene blue R and reduce the external pressure by 27 kPa for 10 min. Restore atmospheric pressure and leave the vials immersed for 30 min.Rinse the outside of the vials. None of the vials contains any trace of coloured solution.[JP]None[USP]Procedure—Fill 10 suitable vials with water to the nominal volume. Fit the closures that are to be examined, and cap. Using a new hypodermic needle as described above for each closure, pierce each closure 10 times, piercing each time at a different site. Immerse the 10 vials in a solution of 0.1% (1 g per L) methylene blue, and reduce the external pressure by 27 kPa for 10 minutes.Restore to atmospheric pressure, and leave the vials immersed for 30 minutes.Rinse the outside of the vials.Requirement—None of the vials contain any trace of blue solution.5、Total Ash[YBB] Take the rubber closures, test according to YBB600212012, should be comply with the standard.[EP]The total ash (2.4.16) is within ± 10 per cent of the result obtained with the type sample.[JP]None[USP] None6、Volatile Sulfides[YBB]Take the rubber closures, according to the YBB60052012, should be comply with the standard.[EP] Place closures, cut if necessary, with a total surface area of 20 ± 2 cm2 in a 100 ml conical flask and add 50 ml of a 20 g/l solution of citric acid R. Place a piece of lead acetate paper R over the mouth of the flask and maintain the paper in position by placing over it an inverted weighing bottle. Heat in an autoclave at 121 ± 2℃ for 30 min. Any black stain on the paper is not more intense than that of a standard prepared at the same time in the same manner using 0.154 mg of sodium sulphide R and 50 ml of a 20 g/l solution of citric acid R.[JP]None[USP] Procedure— Place closures, cut if necessary, with a total surface area of 20 ± 2 cm2 in a 100-mL flask, and add 50 mL of a 20 g per L citric acid solution. In the same manner and at the same time, prepare a control solution in a separate 100-mL flask by dissolving 0.154 mg of sodium sulfide in 50 mL of a 20 g per L citric acid solution. Place a piece of lead acetate paper over the mouth of each flask, and hold the paper in position by placing over it an inverted weighing bottle. Heat the flasks in an autoclave at 121 ± 2℃for 30minutes.Requirement—Any black stain on the paper produced by Solution S is not more intense than that produced by the control solution.7、Preparation of Solution S[YBB] Put a number of uncut closures corresponding to a surface area of about 200 cm2 in a suitable glass container, cover with purified water( sample: water=1:2), boil for 5 min and rinse 5 times with the same volume of purified water. Place the washed closures in a conical flask, add the same volume of water and weigh.Cover the mouth of the flask with a borosilicate-glass beaker. Heat in an autoclave so that a temperature of 121 ± 2 °C is reached within 20 min to 30 min and maintain at this temperature for 30 min. Cool to room temperature.Make up to the original mass with purified water. Shake and immediately separate the solution from the rubber by decantation. Shake solution S before each test Blank. Prepare a blank in the same manner using 400 mL of water for purified water.[EP] Solution S.Introduce a number of uncut closures corresponding to a surface area of about 100 cm2in a suitable glass container, cover with water for injections R, boil for 5 min and rinse 5 times with cold water for injections R.Place the washed closures in a wide-necked flask (glass type I, 3.2.1), add 200 ml of water for injections R and weigh. Cover the mouth of the flask with a borosilicate-glass beaker. Heat in an autoclave so that a temperature of 121 ±2 °C is reached within 20 min to 30 min and maintain at this temperature for 30min. Cool to room temperature over about 30 min. Make up to the original mass with water for injections R. Shake and immediately separate the solution from the rubber by decantation. Shake solution S before each testBlank. Prepare a blank in the same manner using 200 ml of water for injections R.[JP] Wash the rubber closures with water, and dry at room temperature. Place them in a glass container, add water exactly 10 times the mass of the test material, close with a suitable stopper, heat at 121℃ for 1 hour in an autoclave, take out the glass container, allow to cool to room temperature,then take out immediately the rubber closures, and use the remaining solution as the testsolution. Prepare the blank solution with water in the same manner. Perform the following tests with the test solution and the blank solution[USP] Place whole, uncut closures corresponding to a surface area of 100 ± 10 cm2 intoa suitable glass container. Cover the closures with 200 mL of Purified Water orWater for Injection. If it is not possible to achieve the prescribed closure surface area (100 ± 10 cm2) using uncut closures, select the number of closures that will most closely approximate 100 cm2, and adjust the volume of water used to the equivalent of 2 mL per each 1 cm2 of actual closure surface area used. Boil for 5 minutes, and rinse five times with cold Purified Water or Water for InjectionPlace the washed closures into a Type I glass wide-necked flask (see Containers—Glass 660 ), add the same quantity of Purified Water or Water for Injection initially added to the closures, and weigh. Cover the mouth of the flask with a Type I glass beaker. Heat in an autoclave so that a temperature of 121 ± 2℃ is reached within 20 to 30 minutes, and maintain this temperature for30 minutes. Cool to room temperature over a period of about 30 minutes. AddPurified Water or Water for Injection to bring it up to the original mass. Shake, and immediately decant and collect the solution. [NOTE—This solution must be shaken before being used in each of the tests]Prepare a blank solution similarly, using 200 mL of Purified Water or Water for Injection omitting the closures8、Appearance of solution[YBB]According to the part 2 of Chinese pharmacopoeia, 2010 edition of appendixⅨB and appendix IX A, standard solution is not more opalescent than reference suspension II. Standard solution is not more intensely coloured than No.5 reference solution[EP] Solution S is not more opalescent than reference suspension II for type I closures and is not more opalescent than reference suspension III for type II closures(2.2.1). Solution S is not more intensely coloured than reference solution GY5(2.2.2, Method II).[JP] Place 5 mL of the test solution in a glass-stoppered test tube of about 15 mm in inner diameter and about 200 mm in length, and shake vigorously for 3 minutes.The foam arisen disappears almost completely within 3 minutes.[USP] Determination of Turbidity (Opalescence)Procedure A: Visual Comparison— Use identical test tubes made of colorless, transparent, neutral glass with a flat base and an internal diameter of 15 to 25 mm. Fill one tube to a depth of 40 mm with Solution S, one tube to the same depth with water, and four others to the same depth with Reference Suspensions A, B, C,and D. Compare the solutions in diffuse daylight 5 minutes after preparation of the Reference Suspensions, viewing vertically against a black background. The light conditions shall be such that Reference Suspension A can be readily distinguished from water and that Reference Suspension B can be readily distinguished fromReference Suspension A.REQUIREMENT—Solution S is not more opalescent than Reference Suspension B for Type I closures, and not more opalescent than Reference Suspension C for Type II closures. Solution S is considered clear if its clarity is the same as that of water when examined as described above, or if its opalescence is not more pronounced than that of Reference Suspension A (refer to Table 3).Procedure B: Instrumental Comparison—Measure the turbidity of theReference Suspensions in a suitable calibrated turbidimeter (see Spectrophotometry and Light Scattering 851 ). The blank should be run and the results corrected for the blank. Reference Suspensions A, B, C, and D represent 3, 6, 18 and 30 Nephelometric Turbidity Units (NTU), respectively. Measure the turbidity of Solution S using the calibrated turbidimeter. REQUIREMENT—The turbidity of Solution S is not greater than that for Reference Suspension B (6 NTU FTU) for Type I closures, and is not greater than that forReference Suspension C (18 NTU FTU) for Type II closures (refer to Table 3).Determination of ColorColor Standard— Prepare a solution by diluting 3.0 mL of Matching Fluid O (see Color and Achromicity 631 ) with 97.0 mL of diluted hydrochloric acid. Procedure—Use identical tubes made of colorless, transparent, neutral glass with a flat base and an internal diameter of 15 to 25 mm. Fill one tube to a depth of 40 mm with Solution S, and the second with Color Standard. Compare the liquids in diffuse daylight, viewing vertically against a white background. Requirement—Solution S is not more intensely colored than the Color Standard.9、Acidity or Alkalinity(PH)[YBB]Take each of 20 ml blank solution and solution S, respectively with KCl solution1 ml, According to the part2 of Chinese pharmacopoeia, 2010 edition ofappendixⅥH, the difference between the two may not be over 1.0.[EP] To 20ml of solution S add 0.1ml of bromothymol blue solution R1. Not more than 0.3ml of 0.01 M sodium hydroxide or 0.8 ml of 0.01 M hydrochloric acid is required to obtain either a blue or a yellow colour, respectively.[JP] To 20 mL each of the test solution and the blank solution add 1.0 mL each of potassium chloride solution, prepared by dissolving 1.0 g of potassium chloride in water to make 1000 mL. The difference of pH between the two solutions is not more than 1.0[USP] Bromothymol Blue Solution—Dissolve 50 mg of bromothymol blue in a mixture of 4 mL of 0.02 M sodium hydroxide and 20 mL of alcohol. Dilute with water to 100 mL.Procedure— To 20 ml of Solution S add 0.1 ml of Bromothymol Blue Solution.If the solution is yellow, titrate with 0.01 N sodium hydroxide until a blue endpoint is reached. If the solution is blue, titrate with 0.01 N hydrochloric acid until a yellow endpoint is reached. If the solution is green, it is neutral and no titration is required.Blank Correction—Test 20 mL of Blank similarly. Correct the results obtained for Solution S by subtracting or adding the volume of titrant required for the Blank, as appropriate. (Reference Titrimetry 541 .)Requirement— Not more than 0.3 ml of 0.01 N sodium hydroxide produces a blue color, or not more than 0.8 ml of 0.01 N hydrochloric acid produces a yellow color, or no titration is required.10、Absorbance[YBB] Filter solution S on a membrane filter having approximately 0.45 μm pores.Measure the absorbance of the filtrate at wavelengths from 220 nm to 360 nm using the blank as compensation liquid. At these wavelengths, the absorbance does not exceed 0.1(according to the part 2 of Chinese pharmacopoeia, 2010 edition of appendixⅣA).[EP] Carry out the test within 5 h of preparation of solution S. Filter solution S on a membrane filter having approximately 0.45 μm pores rejecting the first few milliliters of filtrate. Measure the absorbance (2.2.25) of the filtrate at wavelengths from 220 nm to 360 nm using the blank (see solution S) as compensation liquid. At these wavelengths, the absorbance does not exceed 0.2 for type I closures or 4.0 for type II closures. If necessary, dilute the filtrate before measurement of the absorbance and correct the result for the dilution. [JP] Read the absorbance of the test solution between 220 nm and 350 nm against the blank solution as directed under Ultraviolet-visible Spectrophotometry <2.54>: it is not more than 0.20.[USP] Procedure — [NOTE—Perform this test within 5 hours of preparing Solution S.] Filter Solution S through a 0.45-µm pore size filter, discarding the first few mL of filtrate. Measure the absorbance of the filtrate at wavelengths between 220 and 360 nm in a 1-cm cell using the blank in a matched cell in the reference beam. If dilution of the filtrate is required before measurement of the absorbance, correct the test results for the dilution.Requirement—The absorbances at these wavelengths do not exceed 0.2 for Type I closures or 4.0 for Type II closures.11、Reducing Substances[YBB] To 20.0 mL of solution S add 1 mL of dilute sulfuric acid R and 20.0 mL of0.002 M potassium permanganate. Boil for 3 min. Cool. Add 0.1 g of potassiumiodide R and titrate immediately with 0.01 M sodium thiosulfate until the colorturned light brown , using 5 drops of starch solution R as indicator. Carry out atitration using 20.0 mL of the blank. The difference between the titrationvolumes is not greater than7.0mL.[EP] Carry out the test within 4 h of preparation of solution S. To 20.0 ml of solution S add 1 ml of dilute sulphuric acid R and 20.0 ml of 0.002 M potassium permanganate. Boil for 3min. Cool. Add 1 g of potassium iodide R and titrate immediately with 0.01 M sodium thiosulphate, using 0.25 ml of starch solution R as indicator. Carry out a titration using 20.0 ml of the blank. The difference between the titration volumes is not greater than 3.0 ml for type I closures and7.0 ml for type II closures.[JP] Measure 100 mL of the test solution in a glass-stoppered,Erlenmyer flask, add10.0 mL of 0.002 mol/L potassium permanganate VS and 5 mL of dilutesulfuric acid, and boil for 3 minutes. After cooling, add 0.10 g of potassium iodide,stopper, mix by shaking, then allow to stand for 10 minutes,and titrate<2.50> with 0.01 mol/L sodium thiosulfate VS(indicator: 5 drops of starch TS).Perform the blank test in the same manner, using 100 mL of the blank solution.The difference in mL of 0.002 mol/L potassium permanganate VS required between the tests is not more than 2.0 mL.[USP] Procedure— [NOTE—Perform this test within 4 hours of preparing Solution S.] To 20.0 mL of Solution S add 1 mL of diluted sulfuric acid and 20.0 mL of0.002 M potassium permanganate. Boil for 3 minutes. Cool, add 1 g ofpotassium iodide, and titrate immediately with 0.01 M sodium thiosulfate, using0.25 mL of starch solution TS as the indicator. Perform a titration using 20.0mL of blank and note the difference in volume of 0.01 M sodium thiosulfaterequired.Requirement—The difference between the titration volumes is not greater than 3.0 mL for Type I closures and not greater than 7.0 mL for Type II closures.12、Residue on evaporation[YBB]Evaporate 100 mL of solution S and blank solution to dryness on a water-bath and dry at 100 °C to 105 °C. The residue weighs not more than 4.0 mg. [EP]Evaporate 50.0 ml of solution S to dryness on a water-bath and dry at 100 °C to 105 °C. The residue weighs not more than 2.0 mg for type I rubber and notmore than 4.0 mg for type II rubber.[JP]Measure 100 mL of the test solution, evaporate on a water bath to dryness, and dry the residue at 1059 C for 1 hour.The mass of the residue is not morethan 2.0 mg.[USP]None13、Ammonium[YBB] Precision measuring 10 ml of solution S, adding alkaline potassium iodide solution 2 ml, place 15 minutes, should not be color; if it colored , compare with reference solution (with 2.0 ml ammonium chloride solution (take ammoniumchloride 31.5 mg, add right amount chlorine free water and dissolve diluted to1000 ml), 8 ml blank reference solution, 2ml alkaline potassium mercuric iodide solution for mixing), should not be more intensely colored (0.0002%)[EP] maximum 2 ppm.Dilute 5 ml of solution S to 14 ml with water R. The solution complies with limit test A.[JP]None[USP] Alkaline Potassium Tetraiodomercurate Solution— Prepare a 100 mL solution containing 11 g of potassium iodide and 15 g of mercuric iodide in water.Immediately before use, mix 1 volume of this solution with an equal volume ofa 250 g per L solution of sodium hydroxide.Test Solution— Dilute 5 mL of Solution S to 14 mL with water. Make alkaline if necessary by adding 1 N sodium hydroxide, and dilute with water to 15 mL.Add 0.3 mL of Alkaline Potassium Tetraiodomercurate Solution, and close the container.Ammonium Standard Solution— Prepare a solution of ammonium chloride in water (1 ppm NH4). Mix 10 mL of the 1 ppm ammonium chloride solution with5 mL water and 0.3 mL of Alkaline Potassium Tetraiodomercurate Solution.Close the container.Requirement—After 5 minutes, any yellow color in the Test Solution is no darker than the Ammonium Standard Solution (no more than 2 ppm of NH4 in Solution S).14、Extractable Zinc[YBB] Filter solution S on a membrane filter having approximately 0.45μm pores, Precision measuring filtrate 10 ml, add 1 ml 2 mol/L of hydrochloric acid and 3 drops of potassium ferrocyanide test solution(weight 4.2 g potassium ferrocyanide trihydrate, dissolve and diluted with water to 100 ml, shake evenly, this product should be new prepared) for mixing, should not be color; if it colored, compare with reference solution( with 3 ml standard zinc solution (weight 44.0g Zinc sulfate seven hydrated compounds, with new boiled and cooled purified water dissolved and diluted to 1000 ml, this product should be new prepared), shall not be deeper(0.0002%),7ml blank reference solution, 1ml 2mol/L hydrochloric acid and 3 drops of potassium ferrocyanide solution for mixing), should not be more intensely colored.(0.0003%)[EP] maximumof 5 μg of extractable Zn per millilitre of solution S.Atomic absorption spectrophotometry (2.2.23, Method I). Test solution. Dilute10.0 ml of solution S to 100 ml with 0.1 M hydrochloric acid.Reference solutions. Prepare the reference solutions using zinc standard solution(10 ppm Zn) R diluted with 0.1 M hydrochloric acid.Source: zinc hollow-cathode lamp.Wavelength: 213.9 nm.Flame: air-acetylene.[JP]To 10.0 mL of the test solution add diluted dilute nitric acid (1 in 3) to make 20 mL, and use this solution as the sample solution. Further, to 1.0 mL of Standard Zinc Solutionfor atomic absorption spectrophotometry add diluted nitricacid (1 in 3) to make exactly 20 mL, and use this solution asthe standard solution. Perform the tests according to the Atomic Absorption Spectrophotometry <2.23>,using these solutions, under the following conditions.The absorbanceof the sample solution is not more than that of the standardsolution.Gas: Combustible gasóAcetylene.Supporting gasóAir.Lamp: Zinc hollow-cathode lamp.Wavelength: 213.9 nm.Standard Zinc Solution for atomic absorption spectrophotometry: Measure exactly 10 mL of the Standard Zinc Stock Solution, and add water to make exactly 1000 mL. Prepare before use. One mL of this solution contains 0.01 mg of zinc(Zn).[USP] Test Solution—Prepare a Test Solution by diluting 10.0 mL of Solution S to 100 mL with 0.1N hydrochloric acid. Prepare a test blank similarly, using the Blank forSolution S.Zinc Standard Solution— Prepare a solution (10 ppm Zn) by dissolving zinc sulfate in 0.1 N hydrochloric acid.Reference Solutions—Prepare not fewer than 3 Reference Solutions by diluting the Zinc Standard Solution with 0.1 N hydrochloric acid. The concentrations of zinc in these Reference Solutions are to span the expected limit of the Test Solution.Procedure—Use a suitable atomic absorption spectrophotometer (see Spectrophotometry and Light Scattering 851 ) equipped with a zinc hollow-cathode lamp and an air–acetylene flame. An alternative procedure such as an appropriately validated inductively coupled plasma analysis (ICP) may be used.Test each of the Reference Solutions at the zinc emission line of 213.9 nm at least 3 times. Record the steady readings. Rinse the apparatus with the test blank solution each time, to ensure that the reading returns to initial blank value.Prepare a calibration curve from the mean of the readings obtained for each Reference Solution. Record the absorbance of the Test Solution. Determine the ppm zinc concentration of the Test Solution using the calibration curve.Requirement— Solution S contains not more than 5 ppm of extractable zinc.15、Cadmium[YBB]None[EP]None[JP]Wash the rubber closures with water, dry at room temperature, cut into minute pieces, mix well, place 2.0 g of them in a crucible of platinum or quartz, moisten them with 2 mL of sulfuric acid, heat gradually to dryness, and ignite between 4509 Cand5009 C until the residue is incinerated. When in cineration was insufficient, moisten the residue with 1 mL of sulfuric acid, heat to dryness, and ignite again. Repeat the above-mentioned procedure if necessary. Cool the crucible,moisten the residue with water, add 2 to 4 mL of hydrochloric acid, heat on a water bath to dryness, add 1 to 5 mL of hydrochloric acid, and dissolve by heating. Then add 0.5 to 1 mL of a mixture of a solution of citric acid monohydrate (1 in 2) and hydrochloric acid (1:1) and 0.5 to 1 mL of a warmed solution of ammonium acetate (2 in 5). When any insoluble residue remains, filter through a glass filter. To the solution thus obtained add 10 mL ofa solution of diammonium hydrogen citrate (1 in 4), 2 drops of bromothymolblue TS and ammonium TS until the color of the solution changes from yellow to green. Then add 10 mL of ammonium sulfate solution (2 in 5) and water to make 100 mL. Next, add 20 mL of a solution of sodium N,N-diethyldithiocarbamate trihydrate (1 in 20), mix, allow to stand for a few minutes,add 20.0 mL of 4-methyl-2-pentanone, and mix by vigorous shaking.Allow to stand to separate the 4-methyl-2-pentanone layer from the solution, filter if necessary, and use as the sample solution. On the other hand, to 10.0 mL of Standard Cadmium Solution add 10 mL of a solution of diammonium hydrogen citrate (1 in 4) and 2 drops of bromothymol blue TS, proceed in the same manner as for the sample solution, and use this solution as the standard solution.Perform the tests according to the Atomic Absorption Spectrophotometry <2.23> under the following conditions, using the sample solution and the standard solution. The absorbance of the sample solution is notmore than that of thestandard solution.Gas: Combustible gas—Acetylene or hydrogen.Supporting gas—Air.Lamp: Cadmium hollow-cathode lamp.Wavelength: 228.8 nm.[USP] None。
药用丁基胶塞资料

药用丁基胶塞的使用安全性药用丁基胶塞的生产背景丁基橡胶瓶塞的内在洁净度、化学稳定性、气密性、生物性能都很好,但是因配方复杂及所加原材料浓度梯度的关系,与一些分子活性比较强的药物封装后,被药物吸收、吸附、浸出、渗透,产生了胶塞与药物的相容性问题,比较突出的是部分头孢菌素类、部分大输液类、以及较多中药注射液制剂等。
所以通过选用一种惰性柔软涂层,覆盖在胶塞表面,隔离药品与橡胶瓶塞的直接接触,这样可以明显改善与药物的相容性。
国家至2004年底前所有药用胶塞(包括输液、口服液等各剂型用胶塞)一律停止使用普通天然胶塞;所有药厂的药品橡胶塞都要使用丁基胶塞。
药品是一种特殊的商品, 其药效与质量直接关系到人身健康和安全, 药品包装的材料与结构形式, 尤其是直接接触药品的包装材料, 对保证药品稳定性起决定性作用。
不适宜的包装材料可引起活性药物成分的渗出、吸附, 甚至发生化学反应, 导致药品失效, 有时还会产生严重的毒副作用。
因此, 药包材的选择是否合适, 是评价药品质量的一项重要指标,丁基胶塞具有吸湿率低, 化学性好, 气密性好, 无生理毒副作用等显著特点, 特别适宜于用作药品密封。
因此天然胶塞已列入被淘汰之列, 而用丁基胶塞取代。
丁基橡胶是由异丁烯和少量异戊二烯(≤3 %)在超低温(- 95 ℃) 条件下聚合而成的共聚物[ 3] ,为白色或暗灰色透明性弹体,其结构式可用下列通式表示:丁基橡胶是气密性最好的橡胶, 其气体透过率约为天然橡胶的1 /20 , 丁基橡胶的耐热性、耐候性和耐臭氧氧化性都很突出。
最高使用温度可达200 ℃, 能长时间暴露于阳光和空气中而不易损坏。
丁基橡胶耐化学腐蚀性好, 耐酸、碱和极性溶剂。
此外, 丁基橡胶的电绝缘性和耐电晕性能比一般合成橡胶好。
耐水性能优异, 水渗透率极低。
减震性能好, 在- 30 ~50 ℃具有良好的减震性能。
在玻璃化温度(- 37 ℃) 时仍具有屈挠性。
丁基橡胶的缺点是硫化速度很慢, 需要高温或长时间硫化, 自黏性和互黏性差, 与其它橡胶的相容性差, 难以并用[ 4] 。
丁基胶塞含硅油质量标准

丁基胶塞含硅油质量标准丁基胶塞含硅油质量标准如下:1.硅油含量丁基胶塞中硅油含量不得超过 2.0%。
硅油是一种优秀的润滑剂和脱模剂,能有效提高丁基胶塞的加工性能和使用性能。
但是,如果硅油含量过高,会影响丁基胶塞的物理性能和密封性能。
因此,必须控制硅油含量。
2.蒸发残渣丁基胶塞在高温下蒸发的残渣含量应不大于 1.0%。
残渣过多会影响胶塞的纯净度,可能对药品的稳定性和药效产生不良影响。
因此,要严格控制残渣含量,保证胶塞的质量。
3.析出物丁基胶塞在使用过程中析出物的含量应不大于 1.0%。
析出物过多会影响胶塞的密封性能,甚至可能对药品的质量产生不良影响。
因此,要严格控制析出物含量,保证胶塞的稳定性和可靠性。
4.颜色丁基胶塞的颜色应为白色或浅灰色。
颜色均匀且无明显色差。
不同颜色的丁基胶塞可能代表不同的密度、硬度、透光率等特性。
因此,要严格控制颜色等级,保证胶塞的质量和性能。
5.硬度丁基胶塞的硬度应在60 Shore A至80 Shore A之间。
硬度是影响丁基胶塞物理性能和加工性能的重要因素。
不同硬度的胶塞适用于不同场合和需求。
因此,要严格控制硬度等级,保证胶塞的适用性和可靠性。
6.密度丁基胶塞的密度应在0.95g/cm³至1.05g/cm³之间。
密度是反映丁基胶塞物理性能的重要参数之一。
不同密度的胶塞具有不同的特性,如高密度胶塞的强度和硬度通常会更高。
要严格控制密度等级,以保证胶塞的质量和性能。
7.透光率丁基胶塞的透光率应不小于25%且不大于70%。
透光率越高,丁基胶塞的透明度越好,可以更好地观察瓶内药品的情况。
但是,如果透光率过高,可能会影响药品的稳定性。
因此,要选择合适的透光率等级,以保证胶塞的透明度和药品的稳定性。
8.折射率丁基胶塞的折射率应不小于1.49且不大于1.52。
折射率是反映物质光学性能的重要参数。
通过控制折射率等级,可以确保丁基胶塞具有较好的透光性和观察性,同时避免影响药品的光学性能。
药用丁基胶塞质量标准模板

药用丁基胶塞质量标准药用氯化丁基橡胶塞标准( 试行) YBB 0004 本标准适用于直接与注射剂接触的氯化丁基橡胶塞。
【外观】取本品数个, 目视检测, 表面色泽应均匀, 不得有污点、杂质、气泡、裂纹、缺胶、粗糙、胶丝、胶屑、海绵状、毛边; 不得有除边造成的残缺或锯齿现象; 不得有模具造成的明显痕迹。
【鉴别】( 1) 称取本品5~20g, 置于干燥的试管中, 将长约4毫米的钠片一片置于固定并倾斜的试管中, 使其恰好位于试样之上, 用火焰的尖端加热试管, 将钠融化在试样上, 继续加热2分钟, 使呈深红色, 冷却后加入乙醇, 将过剩的钠醇化, 加水约10ml溶解, 过滤, 滤液备用。
A: 取滤液1.5ml置于试管中, 加硝酸酸化, 煮沸1~2分钟, 加入硝酸银1滴, 应产生白色沉淀。
B: 取滤液0.2ml, 置于微量试管中, 加氯仿1滴, 加稀硫酸1滴, 加薪配置的氨水1滴( 或3%H2O2溶液2~3滴) , 经振荡混匀后, 静止5分钟, 氯仿层应不显色。
( 2) 红外光谱取本品约3g切成3mm×3mm小块置索氏抽提器中用丙酮或适宜的溶剂回流浸提8小时, 取残渣80℃烘干, 取0.1~0.2g置于裂解管的底部, 然后用试管夹水平的将裂解管移到酒精灯上加热, 当出现裂解产物冷凝在裂解管冷端时, 再继续加热至裂解基本完全但没碳化为止, 取少许裂解物滴在溴化钾片上, 在80℃烘干, 照分光光度法( 《中华人民共和国药典》二部ⅣC) 测定, 应与对照图谱基本一致。
【穿刺落屑】输液瓶用胶塞: 取10只被测胶塞和10只已知穿落屑数的胶塞分别装在与其相配的输液瓶上, 每只瓶中注入半瓶水。
加上铝盖,用手动封盖机封口,打开铝盖穿刺部位。
按先被测胶塞再已知穿刺落屑数胶塞的顺序交替穿刺胶塞。
穿刺时,胶塞保持直立,握持金属穿刺器( 见图1) 垂直向胶塞标记区域内穿刺, 晃动数秒后拨出穿刺器。
每次穿刺前用丙酮或甲基—异丁基酮擦拭穿刺器。
药用丁基胶塞的材质-概述说明以及解释

药用丁基胶塞的材质-概述说明以及解释1.引言1.1 概述药用丁基胶塞是一种常见的医疗器械材料,广泛应用于药品容器的密封和注射器的橡胶塞。
它由丁基橡胶制成,具有良好的柔软性、耐腐蚀性和密封性能。
药用丁基胶塞的主要作用是防止药物在储存和输送过程中的泄漏和污染,确保药品的质量和安全性。
在医药领域中,药用丁基胶塞被广泛用于药瓶、针筒和注射器等容器的密封。
丁基胶塞的材质选择非常重要,它直接影响着药品的稳定性、保存期限和使用效果。
因此,在选择药用丁基胶塞材质时需要考虑多方面因素。
首先,药用丁基胶塞的材质应具有优异的化学稳定性和生物相容性。
医药产品经常与各种药物接触,因此胶塞的材质在接触药物后不应引起有害物质的释放或产生任何不良反应。
此外,药用丁基胶塞应具有良好的耐腐蚀性,能够抵抗药品中的溶剂、溶剂和氧化剂等物质的侵蚀,以确保药品的纯度和效力。
其次,药用丁基胶塞的材质还应具有较好的机械性能。
胶塞的弹性和稳定性是确保其与容器紧密密封的重要特性,不仅要能够顺利穿透瓶口或针头,还要能够防止气体或液体的泄漏。
此外,在长时间使用或高压情况下,胶塞应能够保持一定的形状和稳定性,以确保药品的安全和有效性。
最后,为了满足不同的医药应用需求,药用丁基胶塞的材质还应具有可扩展性。
不同药品对胶塞材质的要求不同,因此需要根据具体的药品性质和应用环境,选择材质的硬度、耐高温性、阻氧性等特性。
这样可以更好地适应各种药品的保存和使用要求,提高药品的质量和稳定性。
综上所述,药用丁基胶塞的材质选择是确保药品质量和安全性的重要环节。
在选择材料时需要考虑化学稳定性、生物相容性、耐腐蚀性、机械性能和可扩展性等因素,以满足不同药品的需求。
随着科学技术的不断进步,我们可以期待药用丁基胶塞材质在未来的发展中更加先进和多样化,为医药行业带来更多的便利和创新。
1.2 文章结构文章结构部分的内容可以包括以下几个方面:(1)本文主要分为引言、正文和结论三个部分,以系统地介绍药用丁基胶塞的材质选择要点以及展望其未来的发展方向。
【免费下载】 药用丁基胶塞质量标准

药用丁基胶塞质量标准药用氯化丁基橡胶塞 标准(试行) YBB 00042002本标准适用于直接与注射剂接触的氯化丁基橡胶塞。
【外观】取本品数个,目视检测,表面色泽应均匀,不得有污点、杂质、气泡、裂纹、缺胶、粗糙、胶丝、胶屑、海绵状、毛边;不得有除边造成的残缺或锯齿现象;不得有模具造成的明显痕迹。
【鉴别】(1)称取本品5~20g ,置于干燥的试管中,将长约4毫米的钠片一片置于固定并倾斜的试管中,使其恰好位于试样之上,用火焰的尖端加热试管,将钠融化在试样上,继续加热2分钟,使呈深红色,冷却后加入乙醇,将过剩的钠醇化,加水约10ml 溶解,过滤,滤液备用。
A :取滤液1.5ml 置于试管中,加硝酸酸化,煮沸1~2分钟,加入硝酸银1滴,应产生白色沉淀。
B :取滤液0.2ml ,置于微量试管中,加氯仿1滴,加稀硫酸1滴,加薪配置的氨水1滴(或3%H2O2溶液2~3滴),经振荡混匀后,静止5分钟,氯仿层应不显色。
(2)红外光谱取本品约3g 切成3mm×3mm 小块置索氏抽提器中用丙酮或适宜的溶剂回流浸提8小时,取残渣80℃烘干,取0.1~0.2g 置于裂解管的底部,然后用试管夹水平的将裂解管移到酒精灯上加热,当出现裂解产物冷凝在裂解管冷端时,再继续加热至裂解基本完全但没碳化为止,取少许裂解物滴在溴化钾片上,在80℃烘干,照分光光度法(《中华人民共和国药典》2000年版二部ⅣC )测定,应与对照图谱基本一致。
【穿刺落屑】输液瓶用胶塞:取10只被测胶塞和10只已知穿落屑数的胶塞分别装在与其相配的输液瓶上,每只瓶中注入半瓶水。
加上铝盖,用手动封盖机封口,打开铝盖穿刺部位。
按先被测胶塞再已知穿刺落屑数胶塞的顺序交替穿刺胶塞。
穿刺时,胶塞保持直立,握持金属穿刺器(见图1)垂直向胶塞标记区域内穿刺,晃动数秒后拨出穿刺器。
每次穿刺前用丙酮或甲基—异丁基酮擦拭穿刺器。
穿刺器不得有损坏,并保持锋利(如穿器损坏,须换用新的)。
丁基橡胶药用瓶塞检验规程

编号:ZL-SOP-QC-001-00目的:建立丁基橡胶药用瓶塞检验操作规程范围:本规程适用于丁基橡胶药用瓶塞的检验责任人:质检科内容:1.器具:量筒、烧杯、刻度吸管、pH计、移液管、100ml容量瓶、TU-1800型紫外分光光度计、微孔滤膜、10ml移液管、电炉、天平、锥形瓶、电热恒温干燥箱、瓷蒸发皿、干燥器、纳氏比色管、高温电炉。
2.试剂:淀粉指示液、高锰酸钾液(0.002mol/L)、硫代硫酸钠滴定液(0.01mol/L)、醋酸盐缓冲液(pH3.5)、碱性碘化汞钾、氯化铵溶液、2mol/L 盐酸、亚铁氰化钾试液、标准锌溶液、标准铅溶液(1ml相当于10μg的Pb)、硫代乙酰胺试液、混合液、碘化钾、稀硫酸、0.1%氯化钾溶液。
3.检查:3.1外观:3.1.1操作步骤:取样品100个,目视检测。
3.1.2结果判定:表面色泽应均匀,不得有污点、杂质、气泡、裂纹、缺胶、粗糙、胶丝、海绵状毛边;不得有除边造成的残缺或锯齿现象;不得有模具造成的明显痕迹,判为合格。
3.2灰分:3.2.1操作步骤:3.2.1.1分别称取样品1.0g两份,放入已炽灼至恒重的两个坩埚中,精密称定;3.2.1.2放入高温电炉中,缓缓炽灼至完全炭化;3.2.1.3再放入800℃炽灼至完全灰化,移置干燥器内;3.2.1.4放冷至室温,精密称定后,再在800℃炽灼至恒重。
4.2.2结果判定:遗留残渣不得过45%,判为合格。
3.3澄清度与颜色:3.3.1操作步骤:取供试液10.0ml置纳氏比色管中。
4.3.2结果判定:溶液应澄清无色。
如显浑浊,与2号浊度标准液比较,不得更浓;如显色,与黄绿色5号标准比色液比较,不得更浓,判为合格。
3.3.3注:试验液S1和空白液S0的制备按《丁基橡胶药用瓶塞质量标准》进行。
3.4酸碱度3.4.1操作步骤3.4.1.1用量筒分别量取试验液S1和空白液S0各20ml,置两个小烧杯中。
3.4.1.2向两个小烧杯中各加入氯化钾溶液(1→1000)1.0ml。
胶塞质量控制及常见质量问题[1]
![胶塞质量控制及常见质量问题[1]](https://img.taocdn.com/s3/m/b42509f29e3143323968935d.png)
胶塞常见质量问题
·微粒: 微粒超标是经常困扰输液生产质量的难
题,微粒超标的原因较多,有胶丝、胶屑、杂质、控制 悬浮物、纤维、毛边等,按形成原因分为内源性微粒污 染、外源性污染、摩擦产生的微粒污染和生物微粒污染 四方面。要减少胶塞微粒污染,生产胶塞的配方应考虑 内源性不溶性微粒要少;加强生产过程控制及符合规范 的环境;优异的清洗工艺;生产环境必须通过GMP认证。 在具备以上条件后,胶塞微粒才可以得到很好的控制
生物特性:溶血 热源 急性全身毒性
胶塞与药物相容性试验
• 胶塞与药物相互作用的方式 吸收—药物分散在胶塞内 吸附—药物被吸附在胶塞的表面 浸出—胶塞内可萃取物浸入药剂内 渗透—水分、空气以渗透方式从胶塞传输至药剂内
胶塞与药物相容性试验
• 胶塞与药物相容性出现问题的基本迹象 药液PH值变化 药液产生沉淀或浑浊 药液颜色改变 药物有效成分降解、含量降低 辅料(如防腐剂等)含量降低 胶塞膨胀变形
加速
长期
考察包装对药物 有效期的支持
5 - 8 个 , 0 、 3、 6、9、12、18、 12、18、 24、36月 24、36月
胶塞与药物相容性试验
丁基胶塞与头孢粉针剂相容性简易试验
国食药监办【2008】 号文件(2008.12)要求: SFDA 国食药监办【2008】7号文件(2008.12)要求: 注射用头孢曲松钠,用丁基胶塞的,每批成品出厂前, 做相容性简易试验,合格后方可出厂;其他头孢类注射剂参 照此法,建立适宜试验方法 。
胶塞和瓶口尺寸及公差对比
• YY0169.2-94《丁基橡胶抗生素瓶塞》1994-07-02 丁基橡胶抗生素瓶塞》 丁基橡胶抗生素瓶塞 发布 1994-11-01实施 本标准等效采用国际标准 ISO8362-2—1998《医用注射器具—第二部分:注 射瓶瓶塞》 • 塞颈直径 A型 B1型 B2型 • 13.2 13.0 13.0±0.1 • 对应的瓶口内径 12.6 12.5 12.5±0.2 • YBB00062005-2 YBB00292005-1 YBB00292005-2
药用丁基胶塞质量标准[指南]
![药用丁基胶塞质量标准[指南]](https://img.taocdn.com/s3/m/b89569fa77232f60dccca1ad.png)
药用丁基胶塞质量标准[指南]药用丁基胶塞质量标准药用氯化丁基橡胶塞标准(试行) YBB 00042002本标准适用于直接与注射剂接触的氯化丁基橡胶塞。
【外观】取本品数个,目视检测,表面色泽应均匀,不得有污点、杂质、气泡、裂纹、缺胶、粗糙、胶丝、胶屑、海绵状、毛边;不得有除边造成的残缺或锯齿现象;不得有模具造成的明显痕迹。
【鉴别】(1)称取本品5,20g,置于干燥的试管中,将长约4毫米的钠片一片置于固定并倾斜的试管中,使其恰好位于试样之上,用火焰的尖端加热试管,将钠融化在试样上,继续加热2分钟,使呈深红色,冷却后加入乙醇,将过剩的钠醇化,加水约10ml溶解,过滤,滤液备用。
A:取滤液1.5ml置于试管中,加硝酸酸化,煮沸1,2分钟,加入硝酸银1滴,应产生白色沉淀。
B:取滤液0.2ml,置于微量试管中,加氯仿1滴,加稀硫酸1滴,加薪配置的氨水1滴(或3,H2O2溶液2,3滴),经振荡混匀后,静止5分钟,氯仿层应不显色。
(2)红外光谱取本品约3g切成3mm×3mm小块置索氏抽提器中用丙酮或适宜的溶剂回流浸提8小时,取残渣80?烘干,取0.1,0.2g置于裂解管的底部,然后用试管夹水平的将裂解管移到酒精灯上加热,当出现裂解产物冷凝在裂解管冷端时,再继续加热至裂解基本完全但没碳化为止,取少许裂解物滴在溴化钾片上,在80?烘干,照分光光度法(《中华人民共和国药典》2000年版二部?C)测定,应与对照图谱基本一致。
【穿刺落屑】输液瓶用胶塞:取10只被测胶塞和10只已知穿落屑数的胶塞分别装在与其相配的输液瓶上,每只瓶中注入半瓶水。
加上铝盖,用手动封盖机封口,打开铝盖穿刺部位。
按先被测胶塞再已知穿刺落屑数胶塞的顺序交替穿刺胶塞。
穿刺时,胶塞保持直立,握持金属穿刺器(见图1)垂直向胶塞标记区域内穿刺,晃动数秒后拨出穿刺器。
每次穿刺前用丙酮或甲基—异丁基酮擦拭穿刺器。
穿刺器不得有损坏,并保持锋利(如穿器损坏,须换用新的)。
药包材标准中丁基胶塞质量要求及测试方法介绍

药包材标准中丁基胶塞质量要求及测试方法介绍药包材标准中丁基胶塞质量要求及测试方法介绍丁基橡胶塞在洁净度、化学稳定性、气密性等方面的性能都非常好,所以广泛应用于输液、口服液等药品包装中。
由于丁基胶塞在药品行业应用广泛,而且胶塞产品直接接触药品,所以国家出台了很多相关标准来控制丁基胶塞的产品质量。
以下我们结合国家药包材标准的要求来介绍丁基胶塞产品的性能要求和测试方法。
一、参考标准(部分):YBB00042005-2015注射液用卤化丁基橡胶塞YBB00052005-2015注射用无菌粉末用卤化丁基橡胶塞YBB00322004-2015注射剂用胶塞垫片穿刺力测定法YBB00332004-2015注射用胶塞垫片穿刺落屑测定法1.穿刺落屑取样品10个,照注射剂用胶塞、垫片穿刺落屑测定法YBB00332004-2015测定,落屑数应不得过20粒。
测定法:选择20个注射剂瓶,每个瓶内加1/2公称容量的水。
取10个被测胶塞和10个已知穿刺落屑胶塞分别装在注射剂瓶上,盖上铝盖或铝塑组合盖,封口后进行预处理。
预处理完成后用丙酮或其他适当的有机溶剂擦拭金属穿刺器,然后手持穿刺器,垂直穿刺被测试胶塞上的标记部位,刺入后晃动注射剂瓶数秒后拔出穿刺器。
将注射剂瓶中水全部通过一张滤纸过滤,在人眼距离滤纸20cm 的位置,用肉眼观察滤纸上的落屑数,必要时可以通过显微镜进一步证实落屑大小和数量。
附金属穿刺器标准要求1.穿刺力取样品10个。
照注射剂用胶塞、垫片穿刺力测定法YBB00322004-2015第二法测定,穿刺瓶塞所需的力均不得超过10N。
测定法:依据标准要求对胶塞样品进行预处理。
取10个胶塞配套的注射剂瓶,分别加入公称容量的水,装上预处理过的被测胶塞,加上铝盖或者铝塑组合盖,封口。
将一只注射针置于材料试验机上固定,将注射剂瓶放入材料试验机,打开铝盖或铝塑组合盖,漏出胶塞标记部位,穿刺器以200mm/min的速度进行垂直穿刺,记录胶塞穿刺所施加的最大力值。
药用丁基胶塞质量标准

药用丁基胶塞质量标准一、引言药用丁基胶塞是药品包装的重要组成部分,其质量直接关系到药品的质量和安全性。
为了规范药用丁基胶塞的生产和质量控制,本质量标准应运而生。
本标准主要包含以下方面:外观、尺寸、物理性能、化学性能和微生物限度。
二、外观药用丁基胶塞的外观应平整、光滑,无明显瑕疵、气泡和颜色不均匀。
胶塞的表面应无残留物、油腻或手印等污染。
三、尺寸药用丁基胶塞的尺寸应符合设计要求,包括直径、高度和厚度等参数。
胶塞的直径应在规定范围内,以确保与药瓶的配合良好;高度和厚度应符合标准,以保证足够的机械强度和密封性能。
四、物理性能1. 拉伸强度:药用丁基胶塞应具有一定的拉伸强度,以保证在正常包装和运输过程中不易破损。
2. 撕裂强度:药用丁基胶塞应具有足够的撕裂强度,以防止在开启药瓶时出现撕裂现象。
3. 压缩永久变形:药用丁基胶塞应具有较小的压缩永久变形,以保证在使用过程中保持良好密封性能。
4. 密度:药用丁基胶塞的密度应均匀一致,且符合标准要求。
5. 热稳定性:药用丁基胶塞应能在药品包装的温度条件下保持稳定,不出现软化、变形等现象。
五、化学性能1. 耐化学腐蚀性:药用丁基胶塞应能耐受药品、消毒剂等化学物质的腐蚀,以确保药品包装的密封性和安全性。
2. 溶出物:药用丁基胶塞在浸泡于药品或其他溶剂中时,应不溶出对人体有害的物质。
3. 蒸发残渣:药用丁基胶塞在高温烘烤或暴露于空气中时,应不产生对人体有害的蒸发残渣。
4. 微生物限度:药用丁基胶塞中不得检出对人体有害的微生物(如细菌、霉菌等)。
5. 无毒无害性:药用丁基胶塞应无毒无害,不会对人体健康造成影响。
6. 透明度:药用丁基胶塞应具有较高的透明度,以便于观察药品包装内部的情况。
7. 耐磨性:药用丁基胶塞应具有较好的耐磨性,以防止在重复使用过程中出现磨损现象。
8. 抗老化性:药用丁基胶塞应具有较好的抗老化性能,以防止在储存和使用过程中出现性能下降现象。
注射液用卤化丁基橡胶塞

YBB30062012注射液用卤化丁基橡胶塞Zhusheyeyong Luhuadingji XiangjiaosaiHalogenated Butyl Rubber Stopper for Injection本标准适用于直接与注射液接触的氯化或溴化丁基橡胶塞。
【外观】取本品数个,照表1 依法检查,应符合规定。
【规格尺寸】取本品数个,照表2 和表3 依法检查,应符合规定。
【鉴别】*(1)称取本品2.0g,剪成小颗粒,置坩锅中,加碳酸氢钠2.0g 均匀覆盖试样,置电炉上,缓缓加热至炭化,放冷,置马弗炉300℃加热至完全灰化,取出后,冷却至室温,加水10ml 使溶解,滤过,取续滤液1.5ml,置于试管中,加硝酸酸化,加入硝酸银试液1 滴,应产生白色或淡黄色沉淀。
(2)取本品适量,照包装材料红外光谱测定法(YBB60012012)第四法测定,应与对照图谱基本一致。
【穿刺落屑】取本品10 个,照注射剂用胶塞、垫片穿刺落屑测定法(YBB60082012)第一法测定,落屑数应不得过20 粒。
【穿刺力】取本品10 个,照注射剂用胶塞、垫片穿刺力测定法(YBB60072012)第一法测定,平均穿刺力不得过75N,且每个胶塞的穿刺力均不得过80N,穿刺过程中不应有胶塞被推入瓶内。
【密封性与穿刺器保持性】取本品10 个,置高压蒸汽灭菌器中(不浸水),121℃±2℃,保持30 分钟,冷却至室温,另取10 个与之配套的玻璃注射液瓶加水至标示容量,用上述胶塞,塞紧,再加上与之配套铝盖,压盖。
用符合注射剂用胶塞、垫片穿刺力测定法(YBB60072012)中图1 所示的穿刺器,向胶塞穿刺部位垂直穿刺,穿刺器刺穿胶塞,倒挂瓶,穿刺器悬挂0.5kg 重物,穿刺器应保持4 小时不被拔出,且瓶塞穿刺部位应无泄漏。
【灰分】取本品1.0g,照橡胶灰分测定法(YBB600212012)测定,不得过45%。
【挥发性硫化物】* 取本品,照挥发性硫化物测定法(YBB60052012)测定,应符合规定。
药用瓶塞和密封件用卤化丁基橡胶

1wt% 硫酸肼溶液和等量 10wt % 六甲基四胺溶 液混合制成储备液, 其浊度值在 4 000NTU 左右 ( 本研 究的实际测量值是 4 077NTU) 。基本的 浊度标 准液是 把 15. 0 ml 储备液 用水 稀释到 1 000 ml, 稀释后的浊度标准是 60NTU ( 本研究 的实际测量值是 60. 1NTU) 。
4 影响药品浊度的因素
药品浊度取决于贮存药粉的表面积( 吸附 力) 、挥发物质的特性、化学性质、贮存的环境和 贮存的时间。
表 2 表明, 药品浊度( 用浊度计测量 NTU) 随药粉表面积的增加而增加, 这表明药物的表 面积越大, 药粉的吸附力越强。
表 2 不同抗生素暴露 18 h 后所测得的浊度值
抗生素名称
第 34 卷第 9 期 2007 年 9 月
世界橡胶工业 World Rubber Industry
Vol. 34 No. 9: 6~ 9 Sep. 2007
药用瓶塞和密封件用卤化丁基橡胶
王备战 ( 郑州翱翔医药包装有限公司, 河南 开封 452483) 编译
摘要: 卤化丁基橡胶被广泛用于医药瓶塞 和密封件的制造, 它的高度不透气、不透水性、化学 惰性
阿帕西林钠( 青霉素) 美洛西林钠( 青霉素) 头孢噻 钠( 头孢菌素) 头孢地嗪钠( 头孢菌素)
状态
冻干 粉剂 粉剂 粉剂
表面积 m2 g- 1
0. 16 5. 6 9. 0 > 100
浊度值 NTU
62 49 6 58 10 96 12
表 3 表明, 极性挥发物比非极性挥发物更 易诱发药品混浊, 这表明极性物质更易吸附在 所用抗生素的表面。
- 1、下载文档前请自行甄别文档内容的完整性,平台不提供额外的编辑、内容补充、找答案等附加服务。
- 2、"仅部分预览"的文档,不可在线预览部分如存在完整性等问题,可反馈申请退款(可完整预览的文档不适用该条件!)。
- 3、如文档侵犯您的权益,请联系客服反馈,我们会尽快为您处理(人工客服工作时间:9:00-18:30)。
药用丁基胶塞质量标准[指南]药用丁基胶塞质量标准药用氯化丁基橡胶塞标准(试行) YBB 00042002本标准适用于直接与注射剂接触的氯化丁基橡胶塞。
【外观】取本品数个,目视检测,表面色泽应均匀,不得有污点、杂质、气泡、裂纹、缺胶、粗糙、胶丝、胶屑、海绵状、毛边;不得有除边造成的残缺或锯齿现象;不得有模具造成的明显痕迹。
【鉴别】(1)称取本品5,20g,置于干燥的试管中,将长约4毫米的钠片一片置于固定并倾斜的试管中,使其恰好位于试样之上,用火焰的尖端加热试管,将钠融化在试样上,继续加热2分钟,使呈深红色,冷却后加入乙醇,将过剩的钠醇化,加水约10ml溶解,过滤,滤液备用。
A:取滤液1.5ml置于试管中,加硝酸酸化,煮沸1,2分钟,加入硝酸银1滴,应产生白色沉淀。
B:取滤液0.2ml,置于微量试管中,加氯仿1滴,加稀硫酸1滴,加薪配置的氨水1滴(或3,H2O2溶液2,3滴),经振荡混匀后,静止5分钟,氯仿层应不显色。
(2)红外光谱取本品约3g切成3mm×3mm小块置索氏抽提器中用丙酮或适宜的溶剂回流浸提8小时,取残渣80?烘干,取0.1,0.2g置于裂解管的底部,然后用试管夹水平的将裂解管移到酒精灯上加热,当出现裂解产物冷凝在裂解管冷端时,再继续加热至裂解基本完全但没碳化为止,取少许裂解物滴在溴化钾片上,在80?烘干,照分光光度法(《中华人民共和国药典》2000年版二部?C)测定,应与对照图谱基本一致。
【穿刺落屑】输液瓶用胶塞:取10只被测胶塞和10只已知穿落屑数的胶塞分别装在与其相配的输液瓶上,每只瓶中注入半瓶水。
加上铝盖,用手动封盖机封口,打开铝盖穿刺部位。
按先被测胶塞再已知穿刺落屑数胶塞的顺序交替穿刺胶塞。
穿刺时,胶塞保持直立,握持金属穿刺器(见图1)垂直向胶塞标记区域内穿刺,晃动数秒后拨出穿刺器。
每次穿刺前用丙酮或甲基—异丁基酮擦拭穿刺器。
穿刺器不得有损坏,并保持锋利(如穿器损坏,须换用新的)。
直至所有胶塞胶被穿刺一次。
取下被测胶塞,将瓶中水全部通过快速滤纸过滤,确保瓶中不残留落屑。
在一般条件下,眼与滤纸距离为25cm,用肉眼观察快速滤纸上的穿刺落屑数。
对已知穿刺落屑数的胶塞同法操作。
被测胶塞落屑总数不得过20粒(注:如果已知穿刺落屑数胶塞的结果与先前测得的结果具有一致性,则应判被测胶塞测得的结果有效。
反之,则无效)。
抗生素瓶用胶塞:胶塞预处理:取适量胶塞加二倍胶塞总表面积(Acm2)的水(2Aml)。
煮沸5min,用水冲洗5次,将胶塞放入三角烧瓶中,加2Aml水,用铝箔或一只硅硼酸盐烧杯将烧杯瓶口盖住,放入高压蒸汽消毒器中加热,在30分钟内升温至121??2?,保持30分钟,于20,30分钟内冷却至室温,取出,在60?条件下烘60min,贮存于密封的玻璃容器中备用。
选择50只与被测胶塞相配的注射剂瓶,每只瓶中注入半瓶水。
将被测胶塞装在25只瓶上,将25只已知穿刺落屑数的胶塞装在另25只瓶上,胶塞均预处理过。
加上铝盖,用手动封盖机封口,打开铝盖穿刺部位。
按先被测胶塞再已知穿刺落屑数胶塞的顺序交替穿刺胶塞。
穿刺时,胶塞保持直立,将注射器充水并除去注射针头(外径0.8mm)上的水,垂直向胶塞标记区域内穿刺,再重复三次,最后一次拔出针头前,将1ml水注入瓶内。
每次穿刺前用丙酮或甲基—异丁基酮擦拭注射针。
每针刺20次后,更换一只注射针。
直至所有胶塞被穿刺四次。
取下被测胶塞,将瓶中水全部通过快速滤纸过滤,确保瓶中不残留落屑。
在一般条件下,眼与滤纸距离为25cm,用肉眼观察快速滤纸上的落屑数。
对已知穿刺落屑数的胶塞同法操作。
被测胶塞落屑总数:不得过5粒。
(注:如果已知穿刺落屑数的胶塞的结果与先前测得的结果具有一致性,则应判测胶塞测得的结果有效。
反之,则无效)。
【穿刺力】输液瓶用胶塞:取10只被测胶塞和10已知穿刺力的胶塞分别装在与其相配的输液瓶上,每只瓶中注入半瓶水。
盖上铝盖,用手动封盖机封口,放入高压蒸汽消毒器中在121??2?下保持20min,降至室温,取出。
用丙酮擦拭穿刺器,不能破坏针尖锋利度,将穿刺器装在穿刺装置上,将瓶放入穿刺装置中,使胶塞中心能受到垂直穿刺,用符合图1规定的穿刺器以(200?50)mm/min的速度,按先被测胶塞再已知穿刺力胶塞的顺序交替穿刺胶塞。
记录刺透胶塞所施加的力。
穿刺器刺10次后,更换一只穿刺器。
直至所有胶塞被穿刺一次。
(注:如果已知穿刺力胶塞的结果与先前测得的结果具有一致性,则应判被测胶塞测得的结果有效。
反之,则无效)。
穿刺被测胶塞所需的力最大不得过80N,平均值不得过75N,穿刺过程中不应有胶塞被推入瓶内。
抗生素瓶用胶塞:将10只被测胶塞(胶塞均照穿刺落屑项下预处理方法预处理过)装在与其相配的注射剂瓶上。
加上铝盖,用手动封盖机封口,打开铝盖穿刺部位,将瓶放入穿刺装置中,使胶塞中心能受到垂直穿刺,用注射针(外径0.8mm)以(200?50)mm/min的速度进行穿刺。
记录刺透胶塞所施加的力,重复穿刺步骤,直至所有胶塞被刺一次。
穿刺被测胶塞所需的力不得过10N。
【密封性与穿刺器保持性】输液瓶用胶塞:取10只胶塞在不浸水条件下放入高压蒸汽消毒器中于121??2?下保持20min,降至室温,取出。
取10只输液瓶加水至标示容量,用被测胶塞盖上,加上铝盖,用手动封盖机封口,打开铝盖穿刺部位。
手握一只符合图1规定的穿刺器垂直于瓶上方,对准胶塞穿刺部位对穿刺器垂直用力,直至刺到底或手刺不动为止。
将刺穿胶塞的瓶子,瓶底向上,固定。
在穿刺器挂钩上施加0.5Kg重物,保持4h,观察。
用穿刺器穿刺胶塞时,穿刺器应能刺到底:穿刺器在0.5Kg重物作用,能保持4小时不被拔出,且胶塞穿刺部位应无水泄漏。
【自密封性】抗生素瓶用胶塞:试验前2h内对被测胶塞进行预处理:将10只被测胶塞放入水中煮沸5min后取出,在70?恒温干燥箱中干燥1h。
在10只瓶中,每只加半瓶水,将胶塞和铝盖用手动封盖机封口后,放入高压蒸汽消毒器中,121??2?,保持30分钟,取出后冷却,放置24小时。
打开铝盖穿刺部位,将瓶放入穿刺装置中,用注射针(外径0.8mm)以(200?50)mm/min的速度在胶塞标记区域内不同位置穿刺3次。
重复穿刺步骤,直至所有胶塞被穿刺3次。
每穿刺10次后换一只新注射针。
将穿刺过的胶塞放进装有10g/L亚甲蓝溶液的烧杯中,使其完全浸没。
将烧杯放入真空箱中,抽真空至真空度为75KPa,维持30min,真空箱恢复至常压,再维持30min。
取出,用水冲洗瓶外,以目力观察。
亚甲基蓝溶液不得渗入瓶内。
【胶塞与容器密合性】抗生素瓶用胶塞:试验前2h内对被测胶塞进得预处理:将10只被测胶塞放入水中煮沸5min后取出,在70?恒温干燥箱中干燥1h。
在10只瓶中,每只加半瓶水,将胶塞和铝盖用手动封盖机封口后,放入高压蒸汽消毒器中,121??2?,保持30分钟,取出后冷却,放置24h。
然后将胶塞放进装有10g/L 亚甲蓝溶液的烧杯中,使其完全浸没。
将烧杯放入真空箱中,抽真空至真空度为75kPa,维持30min,真空箱恢复至常压,再维持30min。
取出,用水冲洗瓶外,以目力观察。
亚甲基蓝溶液不得渗入瓶内。
【炽灼残渣】取本品0.2g,依法检查(中华人民共和国药典2000年版二部附录?N)遗留残渣不得过45.0,。
【挥发性硫化物】取被测胶塞表面积20cm2(必要时可切割)放入加有2.0,枸橼酸液50ml的100ml锥形瓶中,将一张醋酸铅试纸(将白色滤纸(80g/m2)裁成15mm×40mm的纸条,浸入0.25mol/L乙酸铅溶液中,1小时后取出滤纸,置于空气中晾干,装入密封容器备用。
也可采用市售乙酸铅试纸)放在锥形瓶口上,用烧杯反扣其上。
另取一个100ml的锥形瓶,加入标准硫化钠溶液(1.0×10-5g/ml)5ml,枸橼酸液(8.0,)12.5ml和水32.5ml。
将一张醋酸铅试纸放在锥形瓶口上,用烧杯反扣其上。
将上述两个锥形瓶放入高压灭菌器内,121??2?,保持30min。
供试液的醋酸铅试纸不得显色。
如显色,与标准醋酸铅试纸的颜色比较不得更深(50µg/20cm2)。
【不溶性微粒】取被测胶塞表面积100cm2,置于锥形瓶中,加入50ml注射用水至振荡器中(振荡频率300,350次/分钟)振荡20秒。
取上述溶液,照不溶性微粒检查法测定(中华人民共和国药典2000年版二部附录?C),应符合表1规定。
【化学性能】试验液制备取被测胶塞200cm2,放在烧杯中,加入400ml水浸没,煮沸5min,然后每次用400ml水冲洗,共冲洗5次。
再置于锥形瓶中,加水400ml,在高压灭菌器中,在30min内升温至121??2?,保持30min,于20,30分钟内冷却至室温,即得试验液,备用,同时制备空白液。
做以下试验。
澄清度与颜色取试验液10ml,依法检查(中华人民共和国药典2000年版二部附录?B),溶液应澄清;如显浑浊,输液瓶用胶塞与2号浊度标准液比较,不得更浓;抗生素瓶用胶塞,与3号浊度标准液比较,不得更浓;如显色,依法检查(中华人民共和国药典2000年版二部附录?A)与黄绿色5号标准比色液比较,不得更深。
pH变化值取试验液和空白液各20ml,分别加入氯化钾液(1?1000)1ml,依法检查(中华人民共和国药典2000年版二部附录?H),两者之差不得大于1.0。
紫外吸收度取试验液,用孔径0.45µm的滤膜过滤,以空白液为对照,照分光光度法(中华人民共和国药典2000年版二部附录?A)测定,在波长220,360范围内进行扫描。
220,360nm间最大吸收度,输液瓶用胶塞不得过0.1,抗生素瓶用胶塞不得过0.2。
不挥发物精密量取试验液及空白液100ml,置于已恒重的蒸发皿中,水浴蒸干,在105?干燥至恒重,两者之差不得过4.0mg。
易氧化物精密量取试验液20ml,精密加入高锰酸钾滴定液(0.002mol/L)20ml与稀硫酸2ml,煮沸3分钟,迅速冷却。
加0.1g碘化钾至试验液中,用硫代硫酸钠滴定液(0.01mol/L)滴定至浅棕色,再加入5滴淀粉指示液后滴定至无色。
另取水空白液同法操作,二者消耗滴定液之差:输液瓶用胶塞不得过3.0ml,抗生素瓶用胶塞不得过7.0ml。
重金属精密量取试验液10ml,加醋酸盐缓冲液(pH3.5)2ml,依法检查(中华人民共和国药典2000年版二部附录?H第一法),含重金属不得过百万分之一。
铵离子精密量取试验液10ml,加碱性碘化汞钾试液2ml,放置15分钟;如显色,与氯化铵溶液(取氯化铵31.5mg加无氯水适量使溶解并稀释至1000ml)2.0ml,加空白提取液8ml与碱性碘化汞钾试液2ml制成的对照液比较,不得更深(0.0002,)。
